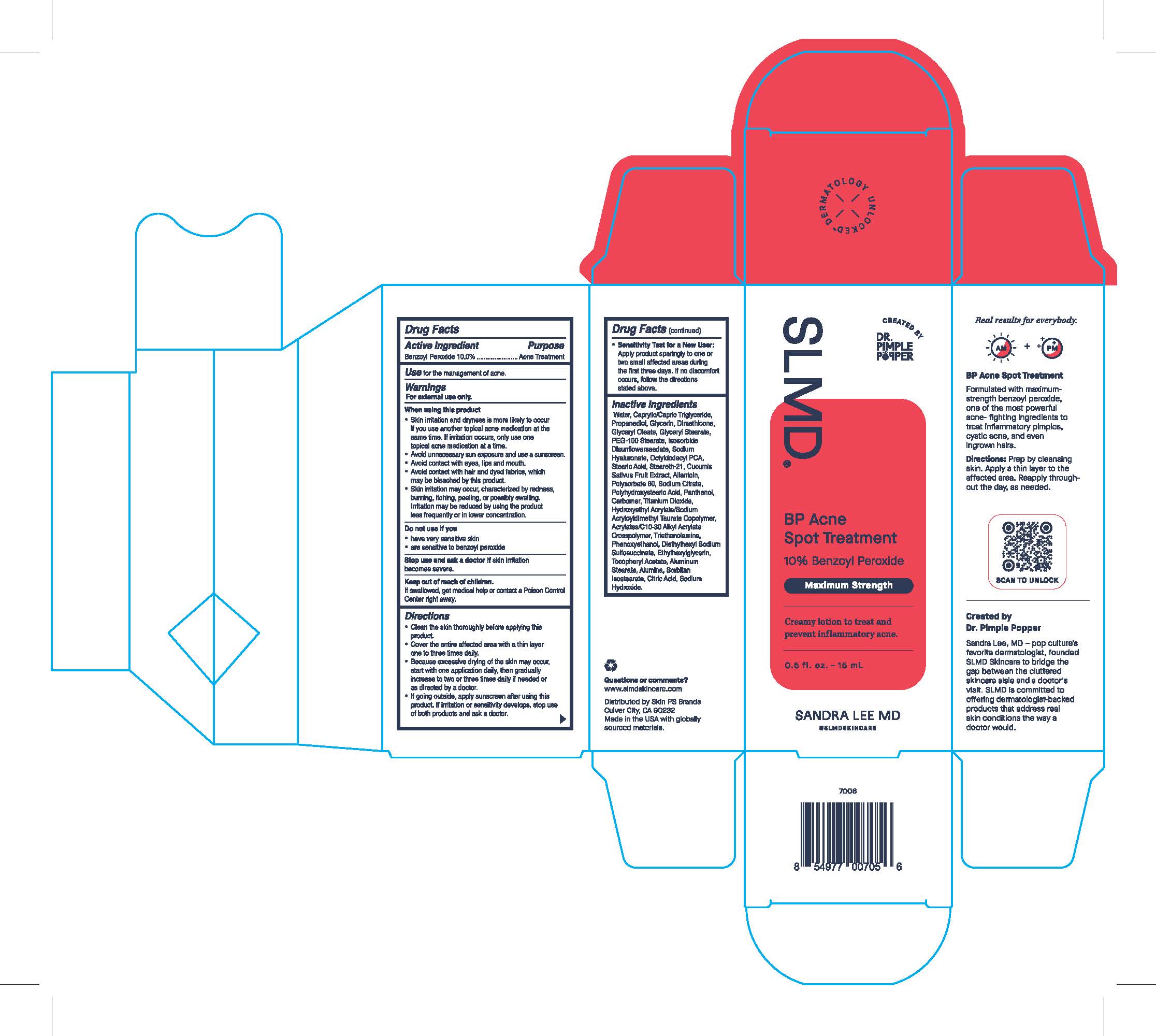
Label: BP ACNE SPOT TREATMENT- acne spot treatment lotion
- NDC Code(s): 73318-7002-5
- Packager: Skin PS Brands
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
-
When using this product
- Skin irritation and dryness is more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- avoid unnecessary sun exposure and use a sunscreen.
- avoid contact with eyes, lips and mouth.
- avoid contact with hair and dyed fabrics, which may be bleached by this product.
- Skin irritation may occur, characterized by redness, burning, itching, peeling, or possibly swelling. Irritation may be reduced by using the product less frequently or in a lower concentration.
- Do not use if you
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- Clean the skin thoroughly before applying this product.
- Cover the entire affected area with a thin layer one to three times daily.
- Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three daily if needed or as directed by a doctor.
- If going outside, apply sunscreen after using this product. If irritation or sensitivity develops, stop use of both products and ask a doctor.
- Sensitivity Test for a New User:
- Apply product sparingly to one or two small affected areas during the first three days. if no discomfort occurs, follow the directions stated above.
-
Inactive Ingredients
Water, Caprylic/Capric Triglyceride, Propanediol, Glycerin, Dimethicone, Glyceryl Oleate, Glyceryl Stearate, PEG-100 Stearate, Isosorbide Disunflowerseedate, Sodium Hyaluronate, Octyldodecyl PCA, Stearic Acid, Steareth-21, Cucumis Sativus Fruit Extract, Allantoin, Polysorbate 60, Sodium Citrate, Polyhydroxystearic Acid, Panethol, Carbomer, Titanium Dioxide, Hydroxyethyl Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Acrylates/C10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Phenoxyethanol, Diethylhexyl Sodium Sulfosuccinate, Ethylhexylglycerin, Tocopheryl Acetate, Aluminum Stearate, Alumina, Sorbitan Isostearate, Citric Acid, Sodium Hydroxide.
- Questions or comments?
-
SLMD
Sandra Lee MD
Skin Care by Dr. Pimple Popper
BP Acne Spot Treatment
10% Benzoyl Peroxide
Maximum Strength
Creamy lotion to treat and prevent inflammatory acne.
0.5 fl. oz. - 15 mL
SANDRA LEE MD
@SLMDSKINCARE
Unit Carton:
Real results for everybody.
AM + PM
BP Acne Spot Treatment
Formulated with maximum-strength benzoyl peroxide, one of the mose powerful acne-fighting ingredients to treat inflammatory pimples, cystic acne, and even ingrown hairs.
Directions: Prep by cleansing skin. Apply a thin layer to the affected area. Reapply throughout the day, as needed.
Created by Dr. Pimple Popper
Sandra Lee, MD - pop culture's favorite dermatologist, founded SLMD Skincare to bridge the gap between the cluttered skincare aisle and a doctor's visit. SLMD is committed to offering dermatologist-backed products that address real skin conditions the way a doctor would.
-
INGREDIENTS AND APPEARANCE
BP ACNE SPOT TREATMENT
acne spot treatment lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73318-7002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 100 mg in 1 g Inactive Ingredients Ingredient Name Strength MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) PROPANEDIOL (UNII: 5965N8W85T) TROLAMINE (UNII: 9O3K93S3TK) POLYSORBATE 60 (UNII: CAL22UVI4M) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL OLEATE (UNII: 4PC054V79P) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) PEG-100 STEARATE (UNII: YD01N1999R) WATER (UNII: 059QF0KO0R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) 2-OCTYLDODECYL 5-OXO-L-PROLINATE (UNII: E25TY46YTD) STEARIC ACID (UNII: 4ELV7Z65AP) STEARETH-21 (UNII: 53J3F32P58) CUCUMBER (UNII: YY7C30VXJT) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ALUMINUM OXIDE (UNII: LMI26O6933) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITAN ISOSTEARATE (UNII: 01S2G2C1E4) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) PANTHENOL (UNII: WV9CM0O67Z) HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER (45000 MPA.S AT 1%) (UNII: 86FQE96TZ4) PHENOXYETHANOL (UNII: HIE492ZZ3T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SODIUM CITRATE (UNII: 1Q73Q2JULR) DOCUSATE SODIUM (UNII: F05Q2T2JA0) ALUMINUM STEARATE (UNII: U6XF9NP8HM) CARBOMER INTERPOLYMER TYPE A (55000 CPS) (UNII: 59TL3WG5CO) DIMETHICONE (UNII: 92RU3N3Y1O) ALLANTOIN (UNII: 344S277G0Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73318-7002-5 1 in 1 CARTON 06/22/2023 1 15 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M006 06/22/2023 Labeler - Skin PS Brands (081085221) Registrant - Skin PS Brands (081085221) Establishment Name Address ID/FEI Business Operations Owen Biosciences 790003045 manufacture(73318-7002)