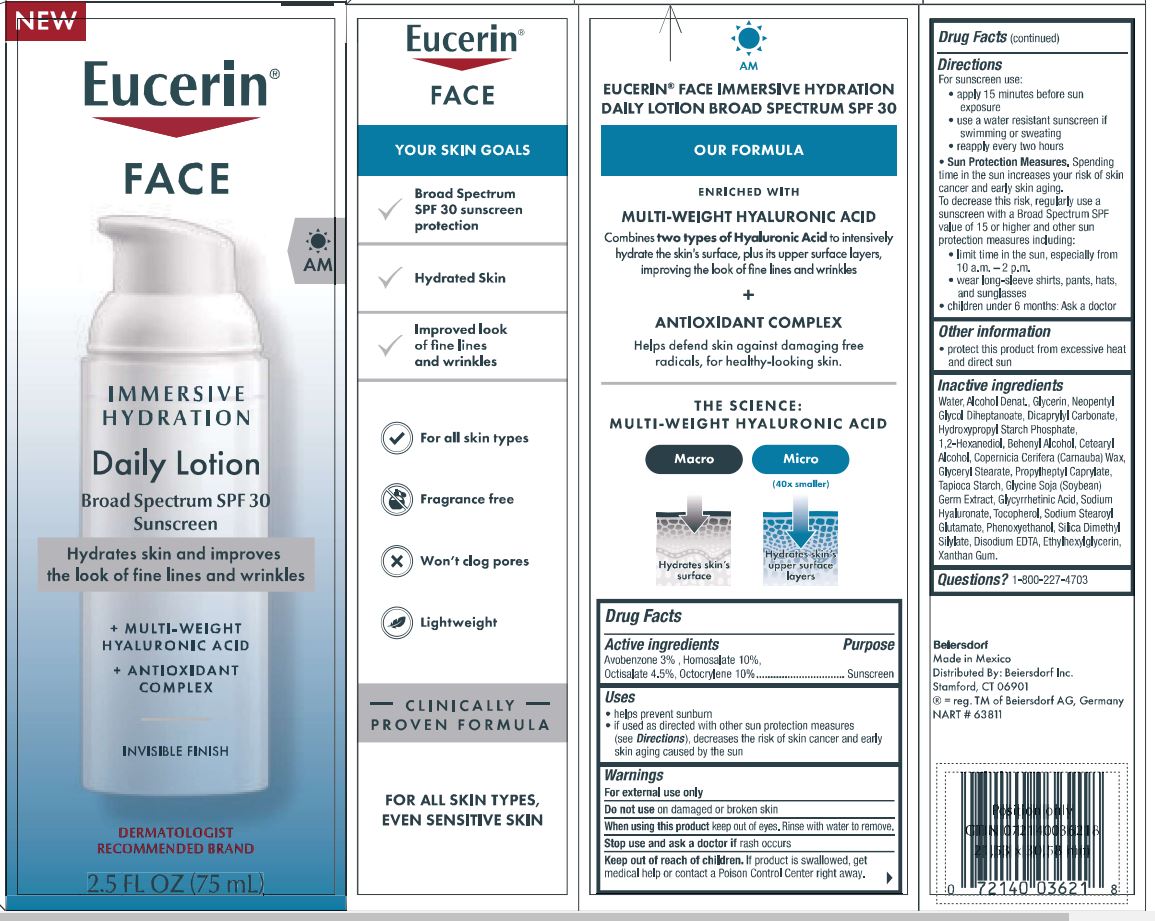
Label: EUCERIN FACE IMMERSIVE HYDRATION- avobenzone, homosalate, octisalate, octocrylene lotion
- NDC Code(s): 10356-385-02, 10356-385-16
- Packager: Beiersdorf Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
For sunscreen use:
• apply liberally 15 minutes before sun exposure
• use a water resistant sunscreen if swimming or sweating
• reapply at least every 2 hours
• children under 6 months of age: Ask a doctor
• Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and
early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF
value of 15 or higher, and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses.
• children under 6 months of age: Ask a doctor. - OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive ingredients
Water, Alcohol Denat., Glycerin, Neopentyl
Glycol Diheptanoate, Dicaprylyl Carbonate,
Hydroxypropyl Starch Phosphate,
1,2-Hexanediol, Behenyl Alcohol, Cetearyl
Alcohol, Copernicia Cerifera (Carnauba) Wax,
Glyceryl Stearate, Propylheptyl Caprylate,
Tapioca Starch, Glycine Soja (Soybean)
Germ Extract, Glycyrrhetinic Acid, Sodium
Hyaluronate, Tocopherol, Sodium Stearoyl
Glutamate, Phenoxyethanol, Silica Dimethyl
Silylate, Disodium EDTA, Ethylhexylglycerin,
Xanthan Gum.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EUCERIN FACE IMMERSIVE HYDRATION
avobenzone, homosalate, octisalate, octocrylene lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10356-385 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 10 g in 100 g HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 100 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 3 g in 100 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 4.5 g in 100 g Inactive Ingredients Ingredient Name Strength NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) PROPYLHEPTYL CAPRYLATE (UNII: 991Z19V2OD) SOYBEAN GERM (UNII: JBW2VHD14M) ALCOHOL (UNII: 3K9958V90M) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) EDETATE DISODIUM (UNII: 7FLD91C86K) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) CARNAUBA WAX (UNII: R12CBM0EIZ) HYALURONATE SODIUM (UNII: YSE9PPT4TH) SODIUM STEAROYL GLUTAMATE (UNII: 65A9F4P024) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) STARCH, TAPIOCA (UNII: 24SC3U704I) ENOXOLONE (UNII: P540XA09DR) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) TOCOPHEROL (UNII: R0ZB2556P8) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) SILICA DIMETHYL SILYLATE (UNII: EU2PSP0G0W) PHENOXYETHANOL (UNII: HIE492ZZ3T) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10356-385-16 1 in 1 CARTON 06/15/2023 1 75 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 2 NDC:10356-385-02 7 g in 1 TUBE; Type 0: Not a Combination Product 01/15/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 06/15/2023 Labeler - Beiersdorf Inc (001177906)