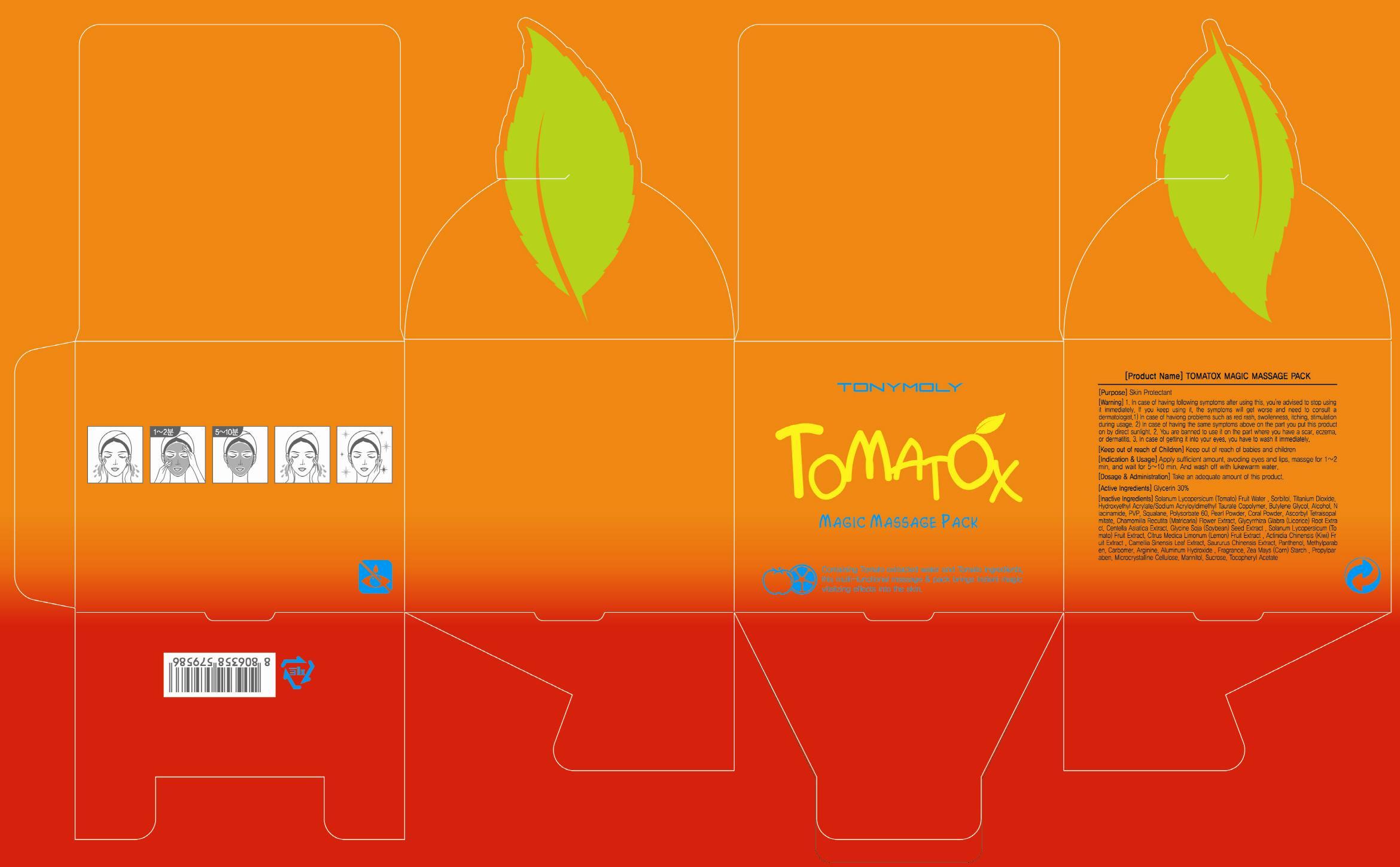
Label: TOMATOX MAGIC MASSAGE PACK- glycerin cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 59078-034-01 - Packager: TONYMOLY CO., LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 29, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive Ingredients:
Solanum Lycopersicum (Tomato) Fruit Water, Sorbitol, Titanium Dioxide, Hydroxyethyl Acrylate/Sodium Acryloyldimethyl Taurate Copolymer, Butylene Glycol, Alcohol, Niacinamide, PVP, Squalane, Polysorbate 60, Pearl Powder, Coral Powder, Ascorbyl Tetraisopalmitate, Chamomilla Recutita (Matricaria) Flower Extract, Glycyrrhiza Glabra (Licorice) Root Extract, Centella Asiatica Extract, Glycine Soja (Soybean) Seed Extract , Solanum Lycopersicum (Tomato) Fruit Extract, Citrus Medica Limonum (Lemon) Fruit Extract , Actinidia Chinensis (Kiwi) Fruit Extract , Camellia Sinensis Leaf Extract, Saururus Chinensis Extract, Panthenol, Methylparaben, Carbomer, Arginine, Aluminum Hydroxide , Fragrance, Zea Mays (Corn) Starch , Propylparaben, Microcrystalline Cellulose, Mannitol, Sucrose, Tocopheryl Acetate - PURPOSE
-
WARNINGS
Warnings:
1. In case of having following symptoms after using this, you're advised to stop using it immediately. If you keep using it, the symptoms will get worse and need to consult a dermatologist.
1) In case of having problems such as red rash, swollenness, itching, stimulation during usage.
2) In case of having the same symptoms above on the part you put this product on by direct sunlight.
2. You are banned to use it on the part where you have a scar, eczema, or dermatitis.
3. In case of getting it into your eyes, you have to wash it immediately. - KEEP OUT OF REACH OF CHILDREN
- INDICATIONS AND USAGE
- DOSAGE AND ADMINISTRATION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOMATOX MAGIC MASSAGE PACK
glycerin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59078-034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Glycerin (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) Glycerin 24 g in 80 g Inactive Ingredients Ingredient Name Strength TOMATO (UNII: Z4KHF2C175) Sorbitol (UNII: 506T60A25R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59078-034-01 80 g in 1 CARTON Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 10/01/2013 Labeler - TONYMOLY CO., LTD. (688216798) Registrant - TONYMOLY CO., LTD. (688216798) Establishment Name Address ID/FEI Business Operations TONYMOLY CO., LTD. 688216798 manufacture(59078-034)