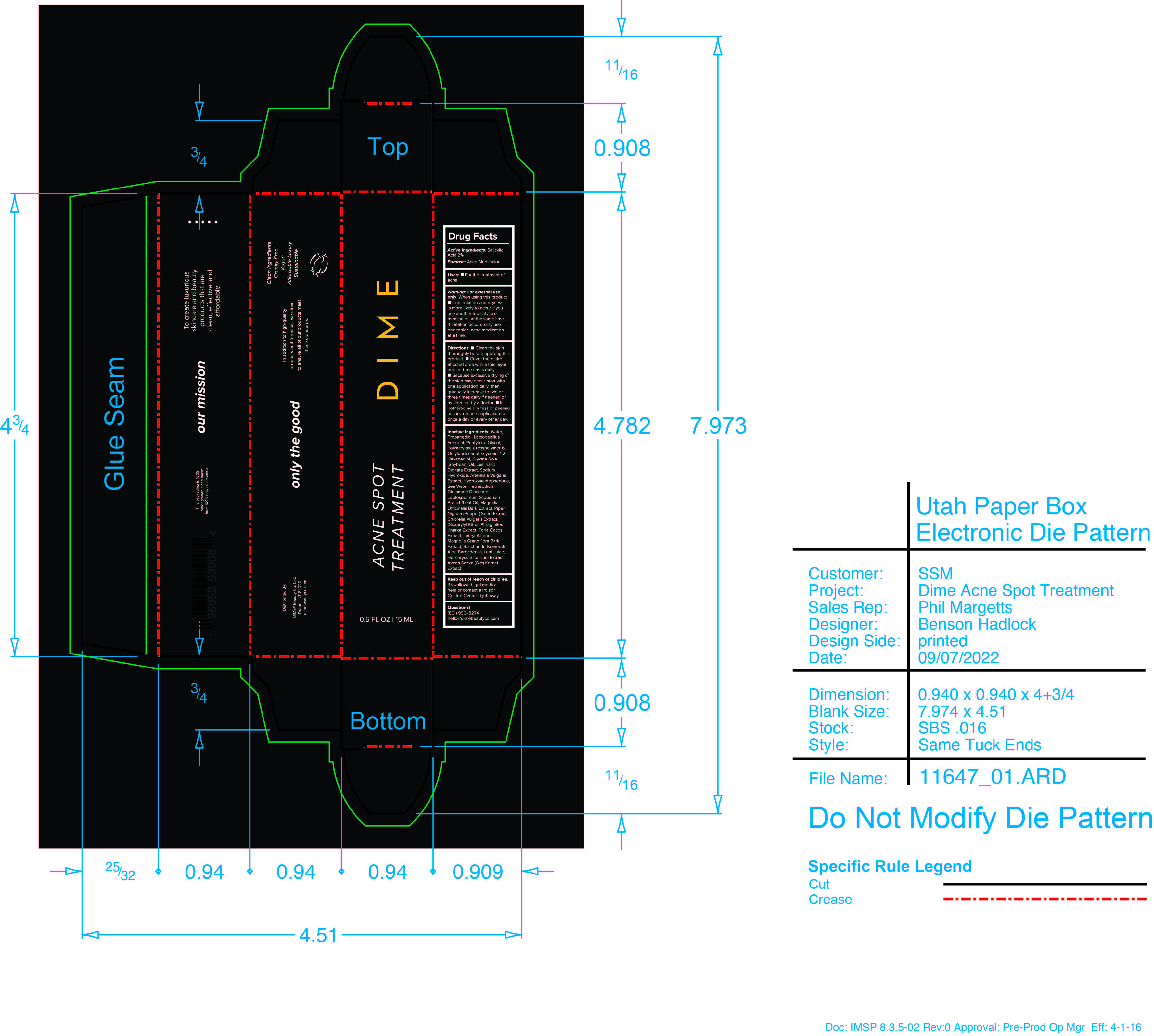
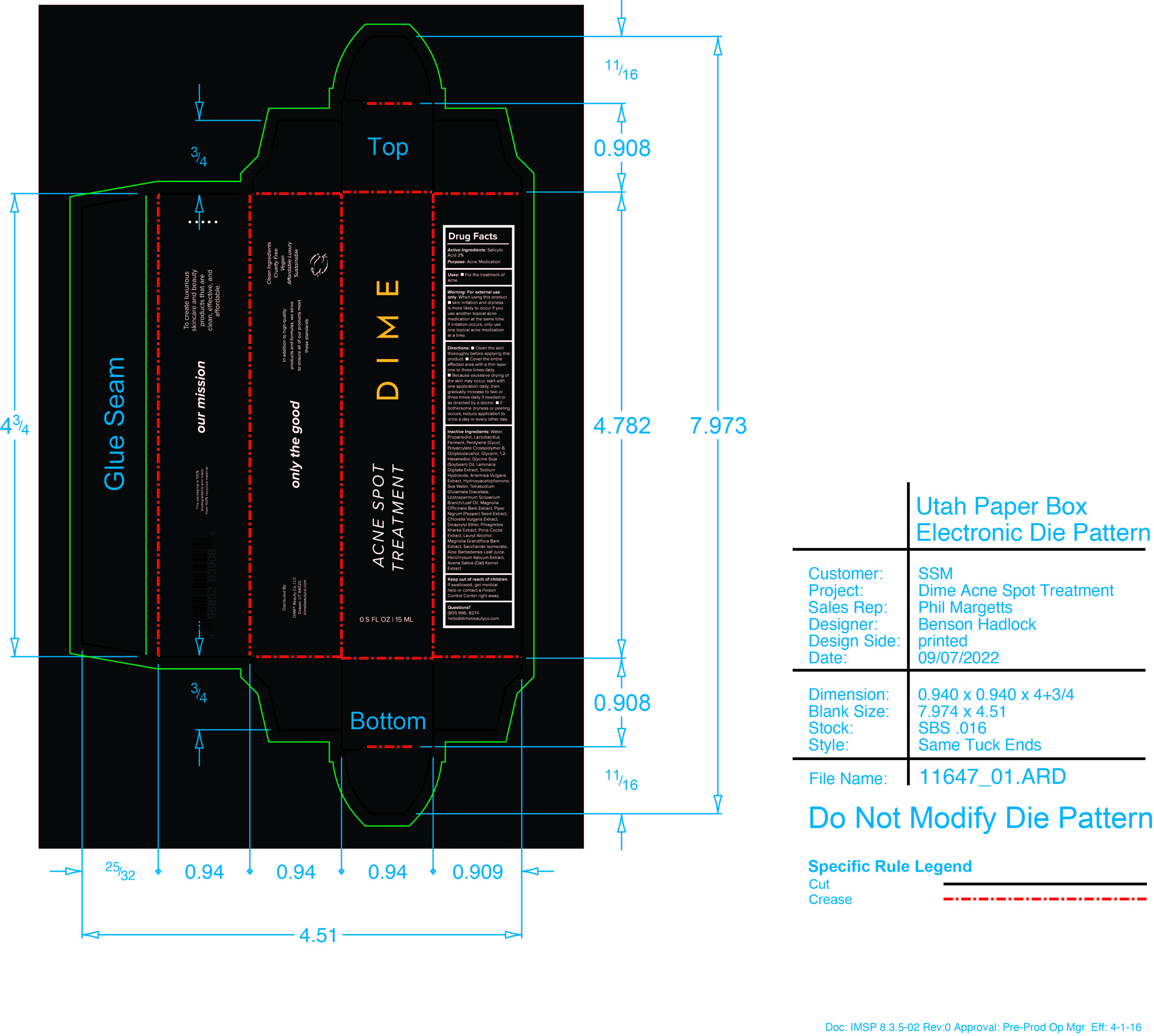
Label: ACNE SPOT TREATMENT- salicylic acid cream
- NDC Code(s): 82870-006-01
- Packager: Dime Beauty Co, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions:
- Clean the skin thoroughly before applying this product
- Cover the entire affected area with a thin layer one to three times daily.
- Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily or as directed by a doctor
- If bothersome dryness or peeling occurs reduce application to once a day or every other day
-
INACTIVE INGREDIENT
Inactive ingredients: Water, Propanediol, Lactobacillus Ferment, Pentylene Glycol, Polyacrylate Crosspolymer-6, Octyldodecanol, Glycerin, 1,2-Hexanediol, Glycine Soja (Soybean) Oil, Laminaria Digitata Extract, Sodium Hydroxide, Artemisia Vulgaris Extract, Hydroxyacetophenone, Sea Water, Tetrasodium Glutamate Diacetate, Leptospermum Scoparium Branch/Leaf Oil, Magnolia Officinalis Bark Extract, Piper Nigrum (Pepper) Seed Extract, Chlorella Vulgaris Extract, Dicaprylyl Ether, Phragmites Kharka Extract, Poria Cocos Extract, Lauryl Alcohol, Magnolia Grandiflora Bark Extract, Saccharide Isomerate, Aloe Barbadensis Leaf Juice, Helichrysum Italicum Extract, Avena Sativa (Oat) Kernel Extract
- OTHER SAFETY INFORMATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACNE SPOT TREATMENT
salicylic acid creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82870-006 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 2 g in 100 mL Inactive Ingredients Ingredient Name Strength ARTEMISIA VULGARIS ROOT (UNII: 32MP823R8S) MANUKA OIL (UNII: M6QU9ZUH2X) MAGNOLIA OFFICINALIS BARK (UNII: 5M609NV974) MAGNOLIA GRANDIFLORA BARK (UNII: J4XF5T6418) SODIUM HYDROXIDE (UNII: 55X04QC32I) FU LING (UNII: XH37TWY5O4) PROPANEDIOL (UNII: 5965N8W85T) CHLORELLA VULGARIS (UNII: RYQ4R60M02) HELICHRYSUM ITALICUM FLOWER (UNII: P62Y550X24) WHITE PEPPER (UNII: M29DW54Q9E) LIMOSILACTOBACILLUS FERMENTUM (UNII: 2C1F12C6AP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) AMMONIUM ACRYLOYLDIMETHYLTAURATE, DIMETHYLACRYLAMIDE, LAURYL METHACRYLATE AND LAURETH-4 METHACRYLATE COPOLYMER, TRIMETHYLOLPROPANE TRIACRYLATE CROSSLINKED (45000 MPA.S) (UNII: Q7UI015FF9) SODIUM CHLORIDE (UNII: 451W47IQ8X) DICAPRYLYL ETHER (UNII: 77JZM5516Z) WATER (UNII: 059QF0KO0R) OAT (UNII: Z6J799EAJK) ALOE VERA LEAF (UNII: ZY81Z83H0X) SACCHARIDE ISOMERATE (UNII: W8K377W98I) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) SOYBEAN OIL (UNII: 241ATL177A) LAMINARIA DIGITATA (UNII: 15E7C67EE8) LAURYL ALCOHOL (UNII: 178A96NLP2) OCTYLDODECANOL (UNII: 461N1O614Y) GLYCERIN (UNII: PDC6A3C0OX) TETRASODIUM GLUTAMATE DIACETATE (UNII: 5EHL50I4MY) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82870-006-01 15 mL in 1 CARTON; Type 0: Not a Combination Product 03/27/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M006 03/27/2023 Labeler - Dime Beauty Co, LLC (058459261)