Label: NAPROXEN tablet
- NDC Code(s): 21130-062-05, 21130-062-09, 21130-062-20
- Packager: Safeway, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 22, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
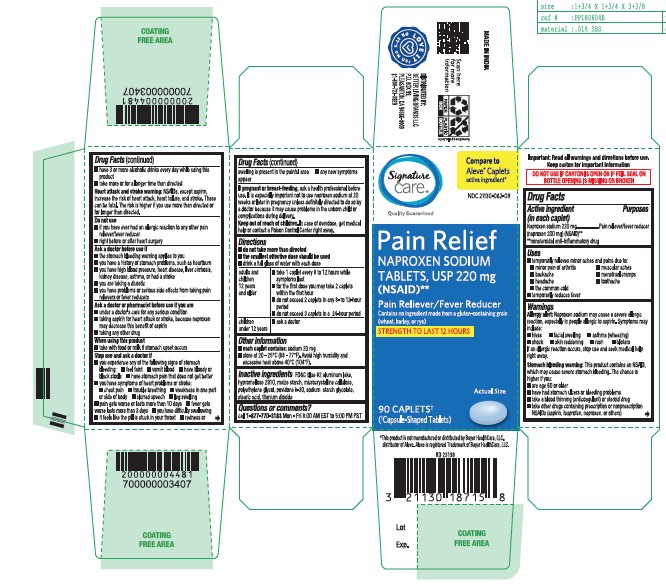
- Active Ingredient (in each caplet)
- Purposes
- Uses
- Allergy Allert
-
Stomach bleeding warning
This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you:
■ are age 60 or older
■ have had stomach ulcers or bleeding problems
■ take a blood thinning (anticoagulant) or steroid drug
■ take other drugs containing prescription or nonprescription NSAIDs (aspirin, ibuprofen, naproxen, or others)
■ have 3 or more alcoholic drinks every day while using this product
■ take more or for a longer time than directed
- Heart attack and stroke warning
- Do not use
-
Ask a doctor before use if
■ the stomach bleeding warning applies to you
■ you have a history of stomach problems, such as heartburn■ you have high blood pressure, heart disease, liver cirrhosis, kidney disease, asthma, or had a stroke
■ you are taking a diuretic
■ you have problems or serious side effects from taking pain relievers or fever reducers
- Ask a doctor or pharmacist before use if you are
- When using this product
-
Stop use and ask a doctor if
■ you experience any of the following signs of stomach bleeding:
■ feel faint ■ vomit blood ■ have bloody or black stools ■ have stomach pain that does not get better
■ you have symptoms of heart problems or stroke:
■ chest pain ■ trouble breathing ■ weakness in one part or side of body ■ slurred speech ■ leg swelling
■ pain gets worse or lasts more than 10 days ■ fever gets worse or lasts more than 3 days ■ you have difficulty swallowing ■ it feels like the pill is stuck in your throat ■ redness or swelling is present in the painful area
■ any new symptoms appear - If pregnant or breast-feeding,
- Keep out of reach of children
-
Directions
■ do not take more than directed
■ the smallest effective dose should be used
■ drink a full glass of water with each dose
Adults and children 12 years and older ■ take 1 caplet every 8 to 12 hours while symptoms last
■ for the first dose you may take 2 caplets within the first hour
■ do not exceed 2 caplets in any 8- to12-hour period
■ do not exceed 3 caplets in a 24-hour periodChildren under 12 years ■ ask a doctor
- Other Information
- Inactive Ingredients
- Questions or Comments?
- Principal Display Panel
- Principal Display Panel
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
NAPROXEN
naproxen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-062 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NAPROXEN SODIUM (UNII: 9TN87S3A3C) (NAPROXEN - UNII:57Y76R9ATQ) NAPROXEN SODIUM 220 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) POVIDONE K30 (UNII: U725QWY32X) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) STARCH, CORN (UNII: O8232NY3SJ) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) Product Characteristics Color blue (light blue) Score no score Shape OVAL ((Caplet -Shaped)) Size 12mm Flavor Imprint Code 220 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-062-20 200 in 1 BOTTLE; Type 0: Not a Combination Product 09/30/2023 2 NDC:21130-062-09 90 in 1 BOTTLE; Type 0: Not a Combination Product 08/30/2023 3 NDC:21130-062-05 50 in 1 BOTTLE; Type 0: Not a Combination Product 08/30/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091353 08/30/2023 Labeler - Safeway, Inc (009137209)