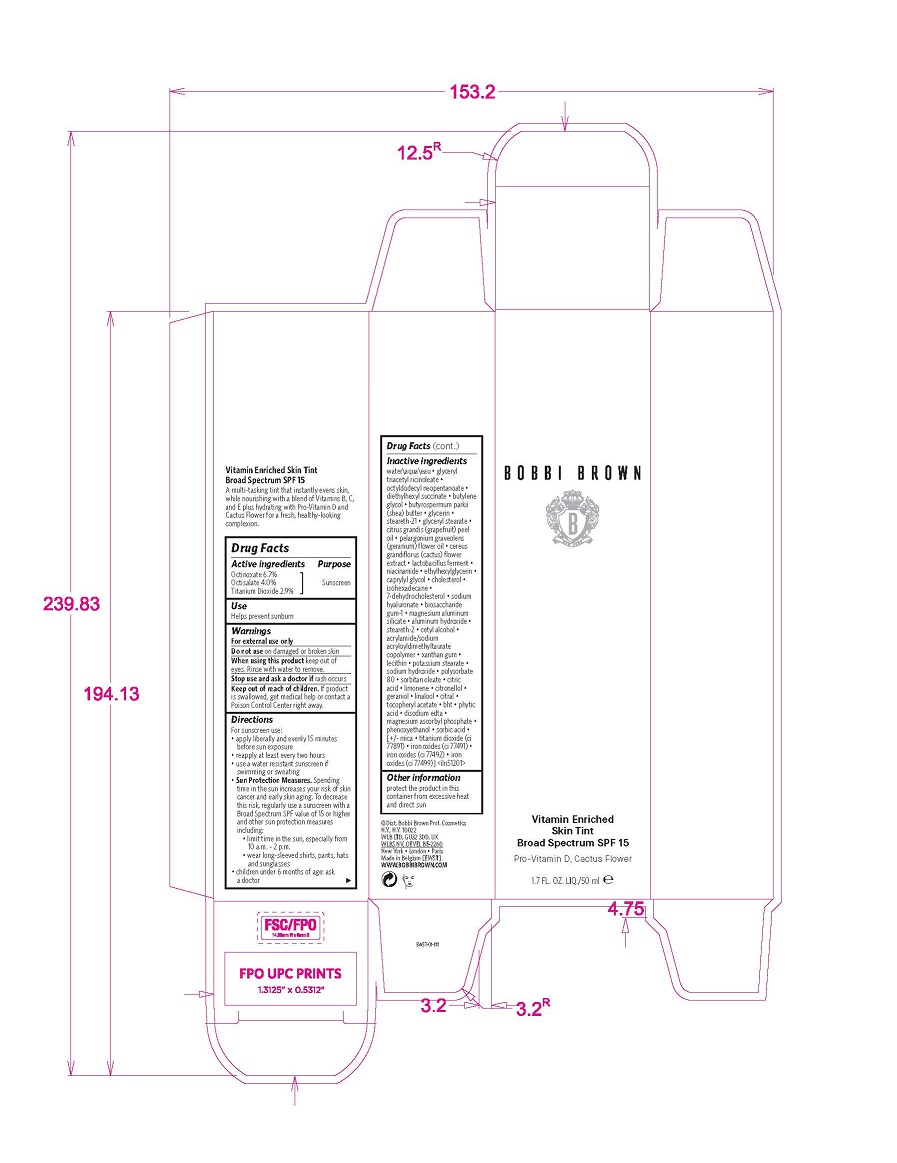
Label: VITAMIN ENRICHED SKIN TINT BROAD SPECTRUM SPF 15 liquid
- NDC Code(s): 64141-033-01, 64141-033-02
- Packager: Bobbi Brown Professional Cosmetics Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Use
- Warnings
-
Directions
For sunscreen use:
• apply liberally and evenly 15 minutes
before sun exposure
• reapply at least every two hours
• use a water resistant sunscreen if
swimming or sweatingSun Protection Measures. Spending
time in the sun increases your risk of skin
cancer and early skin aging. To decrease
this risk, regularly use a sunscreen with a
Broad Spectrum SPF value of 15 or higher
and other sun protection measures
including:
• limit time in the sun, especially fr om
10 a.m. – 2 p.m.
• wear long-sleeved shirts, pants, hats
and sunglasses
• children under 6 months of age: ask
a doctor -
Inactive ingredients
water\aqua\eau • glyceryl
triacetyl ricinoleate •
octyldodecyl neopentanoate •
diethylhexyl succinate • butylene
glycol • butyrospermum parkii
(shea) butter • glycerin •
steareth-21 • glyceryl stearate •
citrus grandis (grapefruit) peel
oil • pelargonium graveolens
(geranium) flower oil • cereus
grandiflorus (cactus) flower
extract • lactobacillus ferment •
niacinamide • ethylhexylglycerin •
caprylyl glycol • cholesterol •
isohexadecane •
7-dehydrocholesterol • sodium
hyaluronate • biosaccharide
gum-1 • magnesium aluminum
silicate • aluminum hydroxide •
steareth-2 • cetyl alcohol •
acrylamide/sodium
acryloyldimethyltaurate
copolymer • xanthan gum •
lecithin • potassium stearate •
sodium hydroxide • polysorbate
80 • sorbitan oleate • citric
acid • limonene • citronellol •
geraniol • linalool • citral •
tocopheryl acetate • bht • phytic
acid • disodium edta •
magnesium ascorbyl phosphate •
phenoxyethanol • sorbic acid •
[+/- mica • titanium dioxide (ci
77891) • iron oxides (ci 77491) •
iron oxides (ci 77492) • iron
oxides (ci 77499)] - Other Information
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
VITAMIN ENRICHED SKIN TINT BROAD SPECTRUM SPF 15
vitamin enriched skin tint broad spectrum spf 15 liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64141-033 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 67 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 40 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 29 mg in 1 mL Inactive Ingredients Ingredient Name Strength ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ISOHEXADECANE (UNII: 918X1OUF1E) HYALURONATE SODIUM (UNII: YSE9PPT4TH) POTASSIUM STEARATE (UNII: 17V812XK50) .BETA.-CITRONELLOL, (R)- (UNII: P01OUT964K) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYSORBATE 80 (UNII: 6OZP39ZG8H) GLYCERYL TRIACETYL RICINOLEATE (UNII: 6TX6N2LEXN) SHEA BUTTER (UNII: K49155WL9Y) GLYCERIN (UNII: PDC6A3C0OX) LIMOSILACTOBACILLUS FERMENTUM (UNII: 2C1F12C6AP) NIACINAMIDE (UNII: 25X51I8RD4) CAPRYLYL GLYCOL (UNII: 00YIU5438U) CHOLESTEROL (UNII: 97C5T2UQ7J) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) DIETHYLHEXYL SUCCINATE (UNII: 69W9UMG3P8) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) SELENICEREUS GRANDIFLORUS FLOWER (UNII: II877K4UNR) LIMONENE, (+)- (UNII: GFD7C86Q1W) GERANIOL (UNII: L837108USY) LINALOOL, (+/-)- (UNII: D81QY6I88E) CITRAL (UNII: T7EU0O9VPP) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) FYTIC ACID (UNII: 7IGF0S7R8I) MAGNESIUM ASCORBYL PHOSPHATE (UNII: 0R822556M5) PHENOXYETHANOL (UNII: HIE492ZZ3T) SORBIC ACID (UNII: X045WJ989B) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) CETYL ALCOHOL (UNII: 936JST6JCN) XANTHAN GUM (UNII: TTV12P4NEE) SODIUM HYDROXIDE (UNII: 55X04QC32I) 7-DEHYDROCHOLESTEROL (UNII: BK1IU07GKF) BIOSACCHARIDE GUM-1 (UNII: BB4PU4V09H) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) STEARETH-21 (UNII: 53J3F32P58) CITRUS MAXIMA FRUIT RIND OIL (UNII: 8U3877WD44) PELARGONIUM GRAVEOLENS FLOWER OIL (UNII: 3K0J1S7QGC) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) WATER (UNII: 059QF0KO0R) STEARETH-2 (UNII: V56DFE46J5) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) MICA (UNII: V8A1AW0880) SODIUM ACRYLOYLDIMETHYLTAURATE-ACRYLAMIDE COPOLYMER (1:1; 90000-150000 MPA.S) (UNII: 5F4963KLHS) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64141-033-01 1 in 1 CARTON 05/01/2023 1 50 mL in 1 TUBE; Type 0: Not a Combination Product 2 NDC:64141-033-02 1 in 1 CARTON 05/01/2023 2 7 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M020 05/01/2023 Labeler - Bobbi Brown Professional Cosmetics Inc. (627131279) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations Estee Lauder N.V. 370151326 manufacture(64141-033) , label(64141-033) , pack(64141-033)