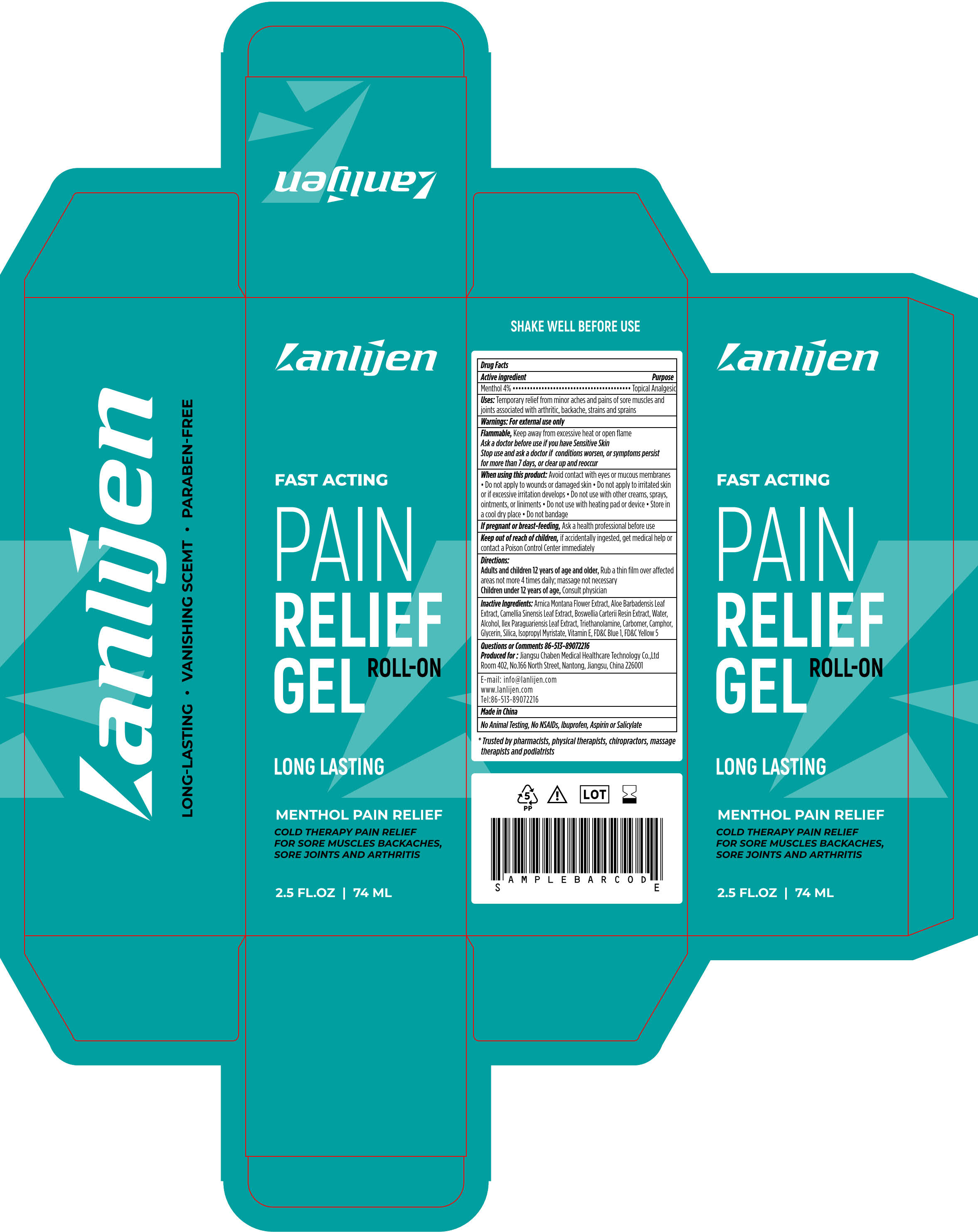
Label: LANLIJEN MENTHOL PAIN RELIEF- menthol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 83050-231-25 - Packager: Jiangsu Chaben Medical Healthcare Technology Co.,Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 18, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- ASK DOCTOR
- STOP USE
-
When using this product
Avoid contact with eyes or mucous membranes • Do not apply to wounds or damaged skin • Do not apply to irritated skin or if excessive irritation develops • Do not use with other creams, sprays, ointments, or liniments • Do not use with heating pad or device • Store in a cool dry place • Do not bandage
- DO NOT USE
- STORAGE AND HANDLING
- If pregnant or breast-feeding
- Keep out of reach of children
- Directions
- ASK DOCTOR/PHARMACIST
- Inactive Ingredients
- Questions or Comments
- OTHER SAFETY INFORMATION
- Package label. Principal display panel
-
INGREDIENTS AND APPEARANCE
LANLIJEN MENTHOL PAIN RELIEF
menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83050-231 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.04 g in 1 g Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) WATER (UNII: 059QF0KO0R) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) ILEX PARAGUARIENSIS LEAF (UNII: 1Q953B4O4F) .ALPHA.-TOCOPHEROL, D- (UNII: N9PR3490H9) ALOE VERA LEAF (UNII: ZY81Z83H0X) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) GLYCERIN (UNII: PDC6A3C0OX) FRANKINCENSE (UNII: R9XLF1R1WM) ALCOHOL (UNII: 3K9958V90M) GREEN TEA LEAF (UNII: W2ZU1RY8B0) TROLAMINE (UNII: 9O3K93S3TK) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83050-231-25 75 g in 1 BOTTLE; Type 0: Not a Combination Product 10/18/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/18/2022 Labeler - Jiangsu Chaben Medical Healthcare Technology Co.,Ltd (615936219) Registrant - Jiangsu Chaben Medical Healthcare Technology Co.,Ltd (615936219) Establishment Name Address ID/FEI Business Operations Shanghai Chuangshi Medical Technology (Group) Co., Ltd. 546872672 manufacture(83050-231)