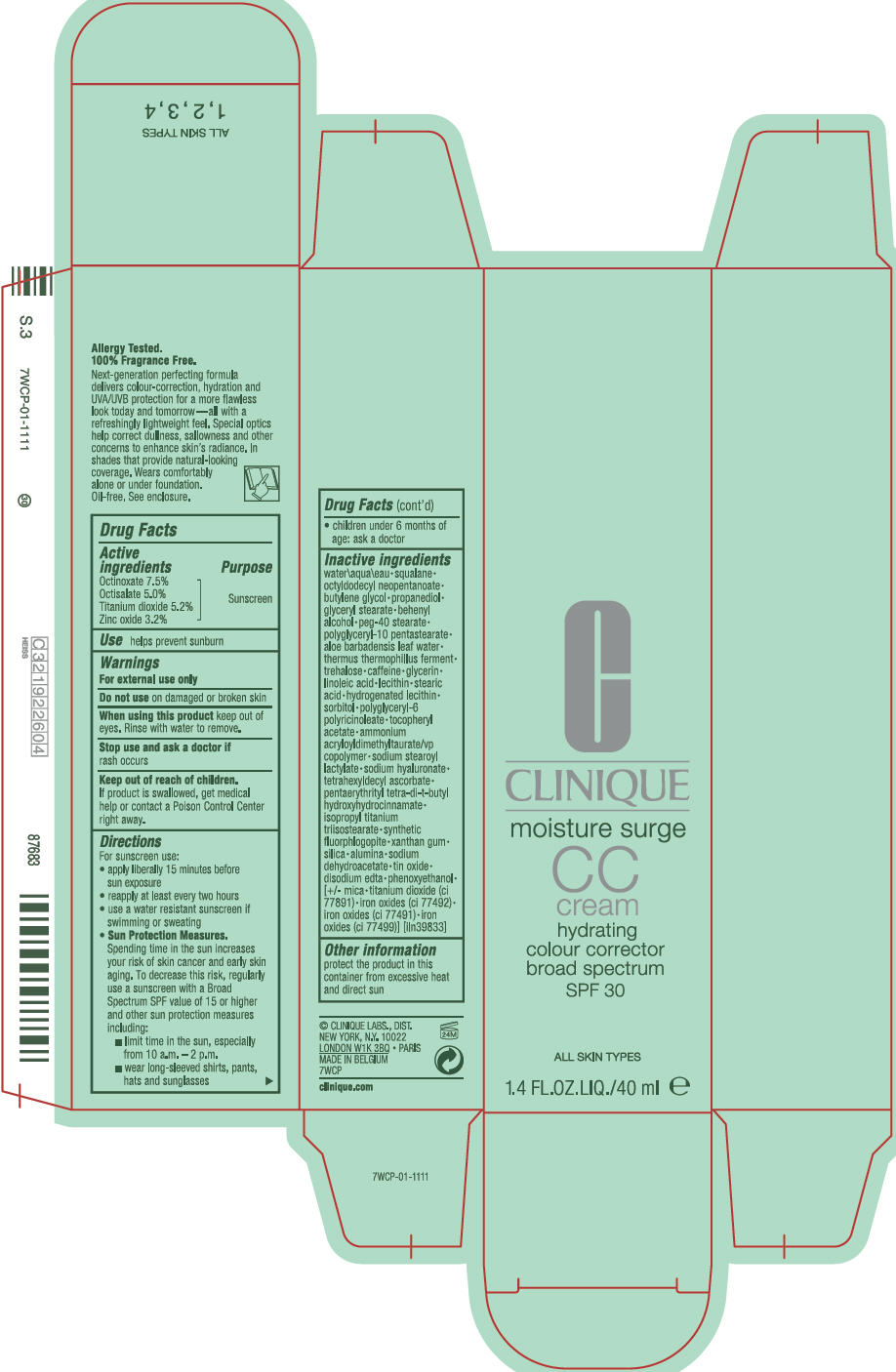
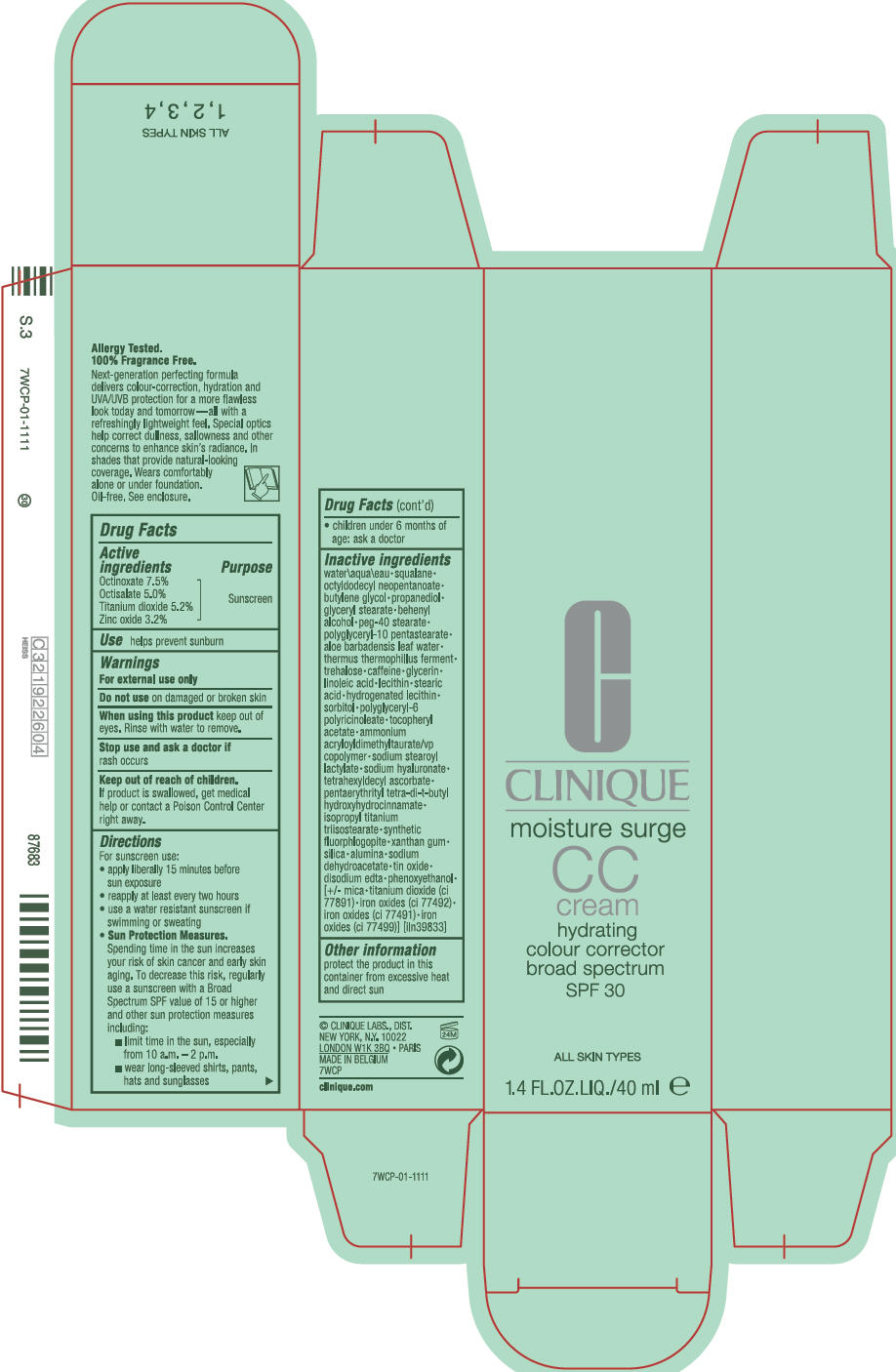
Label: MOISTURE SURGE CC BROAD SPECTRUM SPF 30 HYDRATING COLOUR CORRECTOR- octinoxate, octisalate, titanium dioxide, and zinc oxide cream
- NDC Code(s): 49527-029-01, 49527-029-02, 49527-029-03
- Packager: CLINIQUE LABORATORIES LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 9, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau • squalane • octyldodecyl neopentanoate • butylene glycol • propanediol • glyceryl stearate • behenyl alcohol • peg-40 stearate • polyglyceryl-10 pentastearate • aloe barbadensis leaf water • thermus thermophillus ferment • trehalose • caffeine • glycerin • linoleic acid • lecithin • stearic acid • hydrogenated lecithin • sorbitol • polyglyceryl-6 polyricinoleate • tocopheryl acetate • ammonium acryloyldimethyltaurate/vp copolymer • sodium stearoyl lactylate • sodium hyaluronate • tetrahexyldecyl ascorbate • pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate • isopropyl titanium triisostearate • synthetic fluorphlogopite • xanthan gum • silica • alumina • sodium dehydroacetate • tin oxide • disodium edta • phenoxyethanol • [+/- mica • titanium dioxide (ci 77891) • iron oxides (ci 77492) • iron oxides (ci 77491) • iron oxides (ci 7799)] [iln39833]
- Other information
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 40 ml Bottle Carton
-
INGREDIENTS AND APPEARANCE
MOISTURE SURGE CC BROAD SPECTRUM SPF 30 HYDRATING COLOUR CORRECTOR
octinoxate, octisalate, titanium dioxide, and zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-029 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 52 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 32 mg in 1 mL Inactive Ingredients Ingredient Name Strength THERMUS THERMOPHILUS LYSATE (UNII: 775R692494) POLYGLYCERYL-10 PENTASTEARATE (UNII: PMX5872701) WATER (UNII: 059QF0KO0R) SQUALANE (UNII: GW89575KF9) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PROPANEDIOL (UNII: 5965N8W85T) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) DOCOSANOL (UNII: 9G1OE216XY) PEG-40 STEARATE (UNII: ECU18C66Q7) ALOE VERA LEAF (UNII: ZY81Z83H0X) TREHALOSE (UNII: B8WCK70T7I) CAFFEINE (UNII: 3G6A5W338E) GLYCERIN (UNII: PDC6A3C0OX) LINOLEIC ACID (UNII: 9KJL21T0QJ) STEARIC ACID (UNII: 4ELV7Z65AP) SORBITOL (UNII: 506T60A25R) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) AMMONIUM ACRYLOYLDIMETHYLTAURATE/VP COPOLYMER (UNII: W59H9296ZG) SODIUM STEAROYL LACTYLATE (UNII: IN99IT31LN) HYALURONATE SODIUM (UNII: YSE9PPT4TH) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) PENTAERYTHRITOL TETRAKIS(3-(3,5-DI-TERT-BUTYL-4-HYDROXYPHENYL)PROPIONATE) (UNII: 255PIF62MS) ISOPROPYL TITANIUM TRIISOSTEARATE (UNII: 949E3KBJ1I) XANTHAN GUM (UNII: TTV12P4NEE) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ALUMINUM OXIDE (UNII: LMI26O6933) SODIUM DEHYDROACETATE (UNII: 8W46YN971G) STANNIC OXIDE (UNII: KM7N50LOS6) EDETATE DISODIUM (UNII: 7FLD91C86K) PHENOXYETHANOL (UNII: HIE492ZZ3T) MICA (UNII: V8A1AW0880) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) POLYGLYCERYL-6 POLYRICINOLEATE (UNII: YPM0ZOC2HR) MAGNESIUM POTASSIUM ALUMINOSILICATE FLUORIDE (UNII: YK3DC63Y5M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-029-01 1 in 1 CARTON 10/05/2022 1 40 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:49527-029-02 15 mL in 1 TUBE; Type 0: Not a Combination Product 10/05/2022 12/31/2023 3 NDC:49527-029-03 7 mL in 1 TUBE; Type 0: Not a Combination Product 10/05/2022 12/31/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/05/2022 Labeler - CLINIQUE LABORATORIES LLC (044475127) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd 202952982 manufacture(49527-029) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd. 204132062 pack(49527-029) , label(49527-029) , manufacture(49527-029) Establishment Name Address ID/FEI Business Operations The Estee Lauder Inc 802599436 manufacture(49527-029) , label(49527-029) , pack(49527-029) Establishment Name Address ID/FEI Business Operations PADC 949264774 label(49527-029) , pack(49527-029)