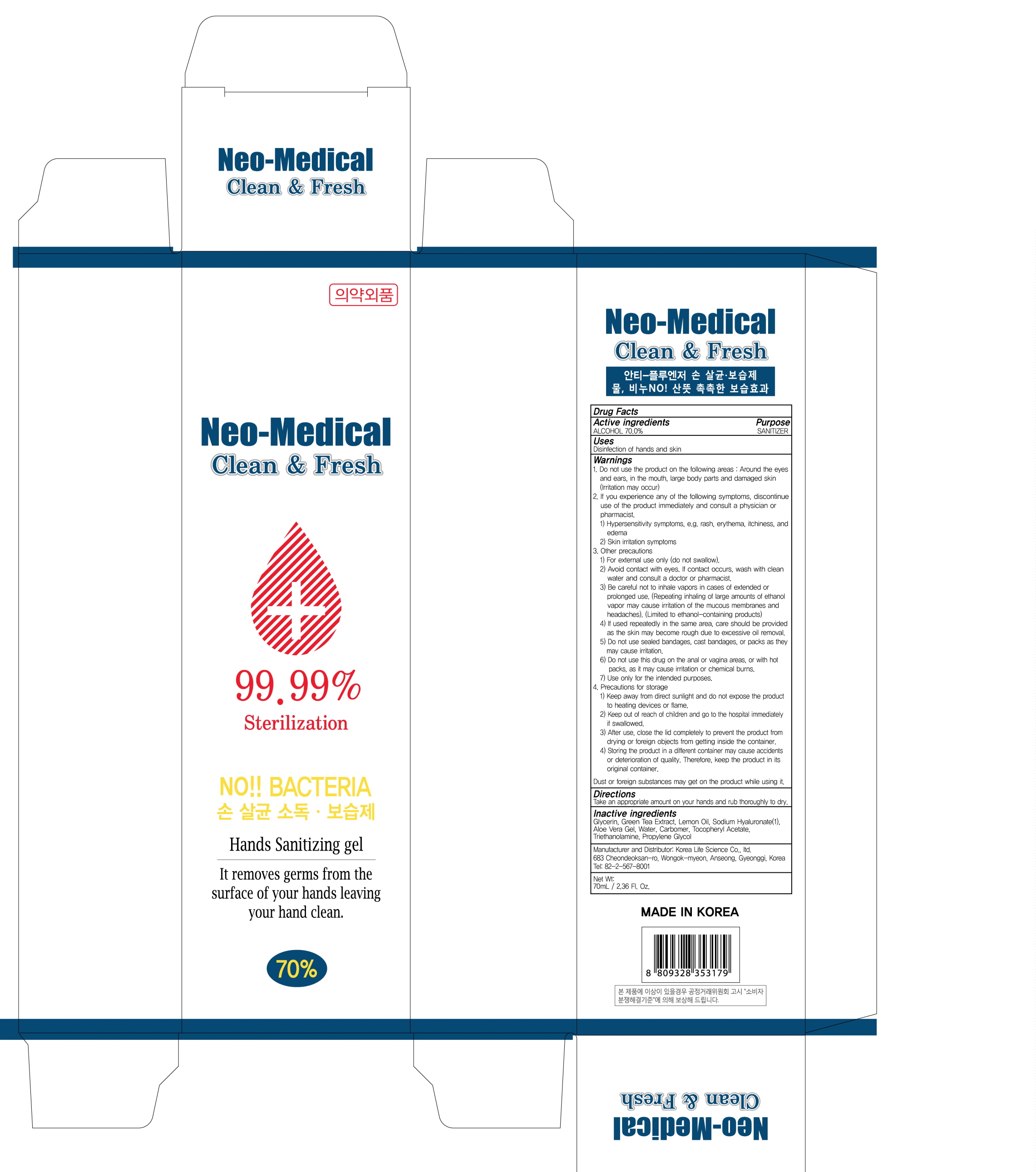
Label: CLEAN AND FRESH S- alcohol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 74406-070-01 - Packager: Korea Life Science Co.,ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 23, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
Warnings:
1.Do not use the product on the following areas:
Around the eyes and ears, in the mouth, large body parts and damaged skin(Irritation may occur)2.If you experience any of the following symptoms, discontinue use of the product immediately and consult a physician or pharmacist.
1) Hypersensitivity symptoms, e.g. rash, erythema, itchiness, and edema
2)Skin irritation symptoms3.Other precautions
1) For external use only (do not swallow).
2) Avoid contact with eyes. If contact occurs, wash with clean water and consult a doctor or pharmacist.
3)Be careful not to inhale vapors in cases of extended or prolonged use. (Repeating inhaling of large amounts of ethanol vapor may cause irritation of the mucous membranes and headaches).
(Limited to ethanol-containing products)
4) If used repeatedly in the same area, care should be provided as the skin may become rough due to excessive oil removal.
5) Do not use sealed bandages, cast bandages, or packs as they may cause irritation.
6) Do not use this drug on the anal or vagina areas, or with hot packs, as it may cause irritation or chemical burns.
7) Use only for the intended purposes.4.Precautions for storage
1) Keep away from direct sunlight and do not expose the product to heating devices or flame.
2) Keep out of reach of children and go to the hospital immediately if swallowed.
3) After use, close the lid completely to prevent the product from drying or foreign objects from getting inside the container.
4) Storing the product in a different container may cause accidents or deterioration of quality. Therefore, keep the product in its original container.Dust or foreign substances may get on the product while using it.
- KEEP OUT OF REACH OF CHILDREN
- Uses
- Directions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLEAN AND FRESH S
alcohol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74406-070 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 49 mL in 70 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) GREEN TEA LEAF (UNII: W2ZU1RY8B0) Lemon Oil (UNII: I9GRO824LL) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TROLAMINE (UNII: 9O3K93S3TK) Propylene Glycol (UNII: 6DC9Q167V3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74406-070-01 70 mL in 1 TUBE; Type 0: Not a Combination Product 04/01/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 04/01/2020 Labeler - Korea Life Science Co.,ltd (694914835) Registrant - Korea Life Science Co.,ltd (694914835) Establishment Name Address ID/FEI Business Operations Korea Life Science Co.,ltd 694914835 manufacture(74406-070)