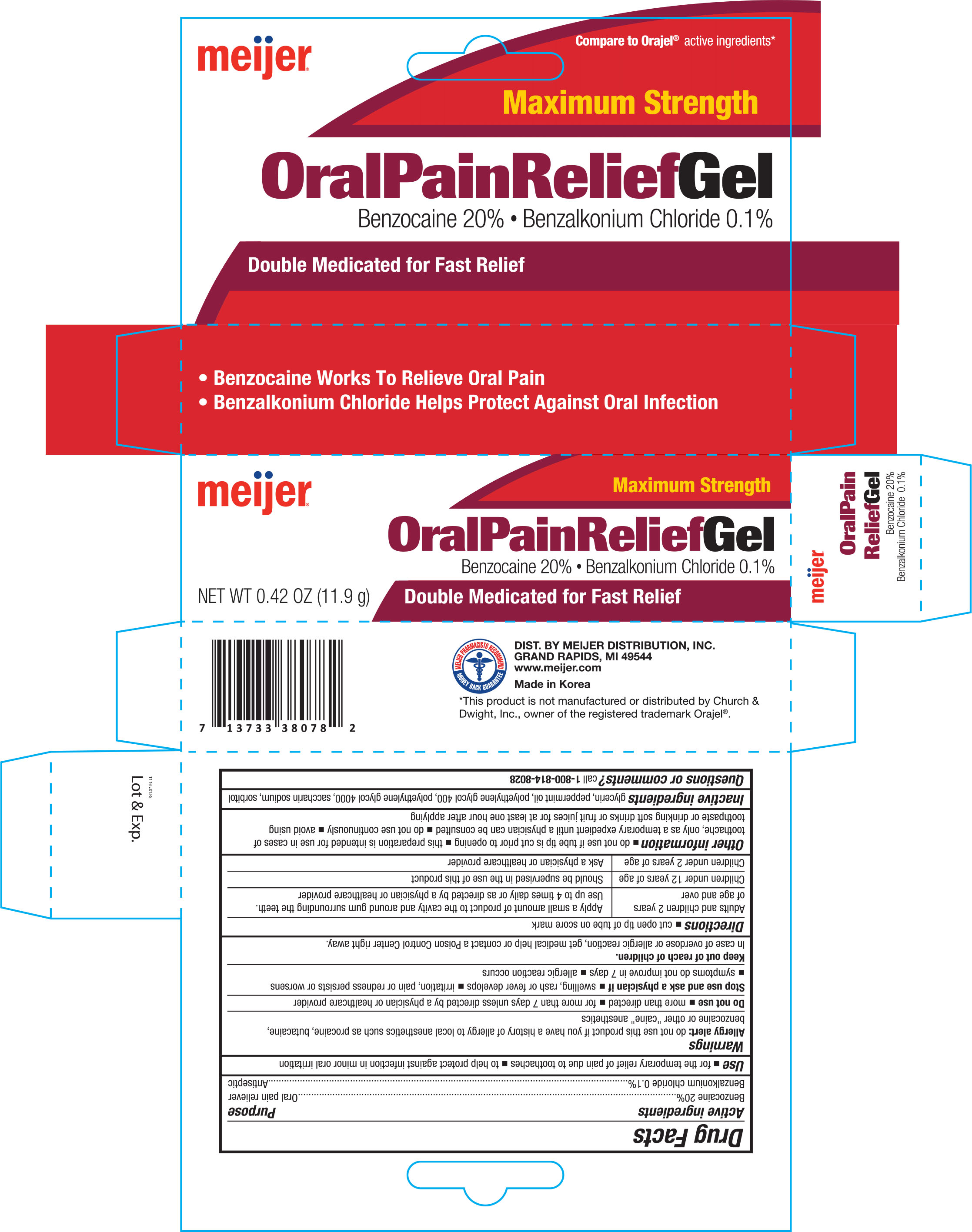
Label: MEIJER MAXIMUM STRENGTH ORAL PAIN RELIEF- benzocaine, and benzalkonium chloride gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 41250-782-42 - Packager: MEIJER DISTRIBUTION INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 8, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- DO NOT USE
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Directions
- cut open tip of tube on score mark
Adults and children 2 years of age and over Apply a small amount of product to the cavity around gum surrounding the teeth. Children under 12 years of age Should be supervised in the use of this product Children under 2 years of age Ask a physician or healthcare provider
- cut open tip of tube on score mark
-
STORAGE AND HANDLING
Other information
- do not use if tube tip is cut prior to opening
- this preparation is intended for use in cases of toothache, only as a temporary expedient until a physician can be consulted
- do not use continuously
- avoid using toothpaste or drinking soft drinks or fruit juices for at least one hour after applying
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MEIJER MAXIMUM STRENGTH ORAL PAIN RELIEF
benzocaine, and benzalkonium chloride gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41250-782 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOCAINE (UNII: U3RSY48JW5) (BENZOCAINE - UNII:U3RSY48JW5) BENZOCAINE 200 mg in 1 g BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) PEPPERMINT OIL (UNII: AV092KU4JH) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYETHYLENE GLYCOL 4000 (UNII: 4R4HFI6D95) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SORBITOL (UNII: 506T60A25R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41250-782-42 1 in 1 BOX 12/08/2016 1 11.9 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 12/08/2016 Labeler - MEIJER DISTRIBUTION INC (006959555)