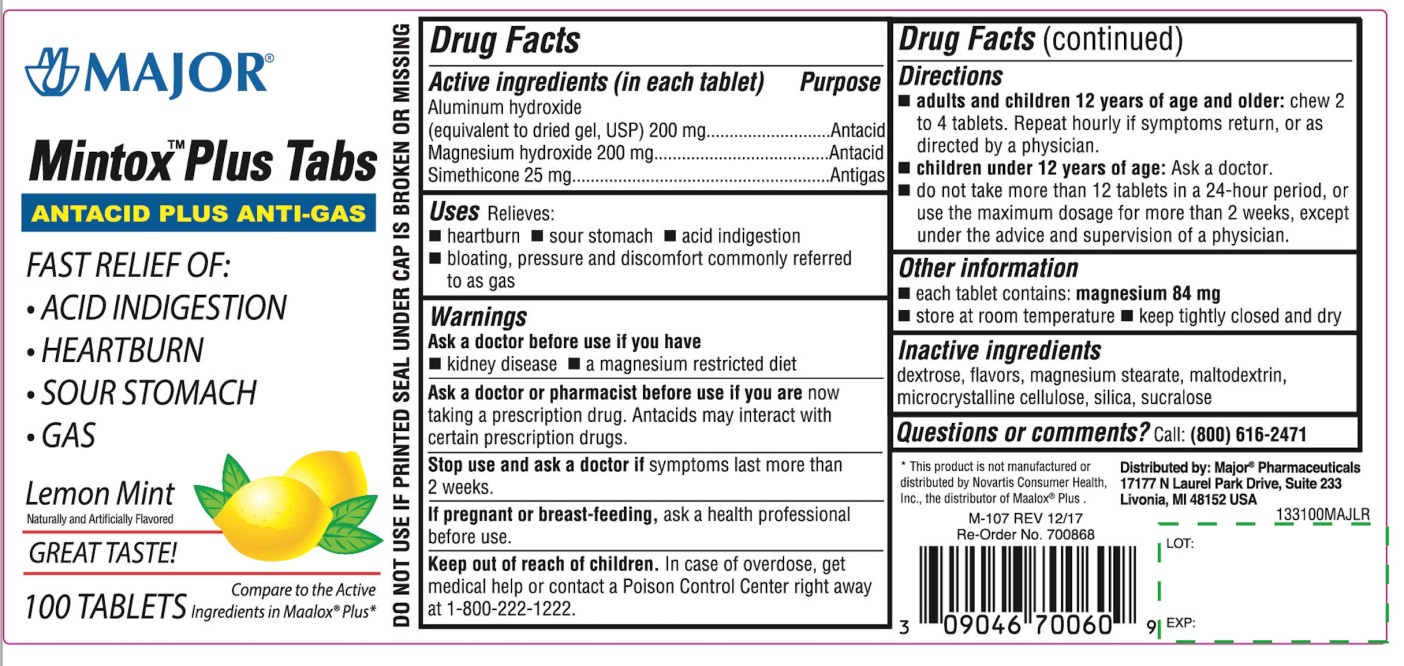
Label: MINTOX PLUS TABS- aluminum hydroxide, magnesium hydroxide, simethicone tablet, chewable
- NDC Code(s): 0904-6700-60
- Packager: Major Pharmaceuticals
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each tablet)
- Purpose
- Uses
- Warnings
-
Directions
- ▪
- adults and children 12 years of age and older: chew 2 to 4 tablets. Repeat hourly if symptoms return, or as directed by a physician .
- ▪
- children under 12 years of age : Ask a doctor.
- ▪
- do not take more than 12 tablets in a 24- hour period, or use the maximum dosage for more than 2 weeks ,except under the advice and supervision of a physician.
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
MAJOR®
Mintox™ Plus Tabs
ANTACID PLUS ANTI-GAS
FAST RELIEF OF:
- •
- ACID INDIGESTION
- •
- HEARTBURN
- •
- SOUR STOMACH
- •
- GAS
Lemon Mint
Naturally and Artificially Flavored
GREAT TASTE!
100 TABLETS
Compare to the Active Ingredients in Maalox ®Plus*
Distributed by:
Major® Pharmaceuticals
17177N Laurel park drive, Suite 233
Livonia, MI 48152 USA
*This product is not manufactured or distributed by Novartis Consumer Health Inc, the distributor of Maalox® plus.
-
INGREDIENTS AND APPEARANCE
MINTOX PLUS TABS
aluminum hydroxide, magnesium hydroxide, simethicone tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0904-6700 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) (ALUMINUM HYDROXIDE - UNII:5QB0T2IUN0) ALUMINUM HYDROXIDE 200 mg MAGNESIUM HYDROXIDE (UNII: NBZ3QY004S) (MAGNESIUM CATION - UNII:T6V3LHY838, HYDROXIDE ION - UNII:9159UV381P) MAGNESIUM HYDROXIDE 200 mg DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 25 mg Inactive Ingredients Ingredient Name Strength DEXTROSE, UNSPECIFIED FORM (UNII: IY9XDZ35W2) MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color WHITE Score no score Shape ROUND Size 16mm Flavor LEMON (MINT) Imprint Code RP133 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0904-6700-60 100 in 1 BOTTLE; Type 0: Not a Combination Product 08/02/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 08/02/2018 Labeler - Major Pharmaceuticals (191427277)