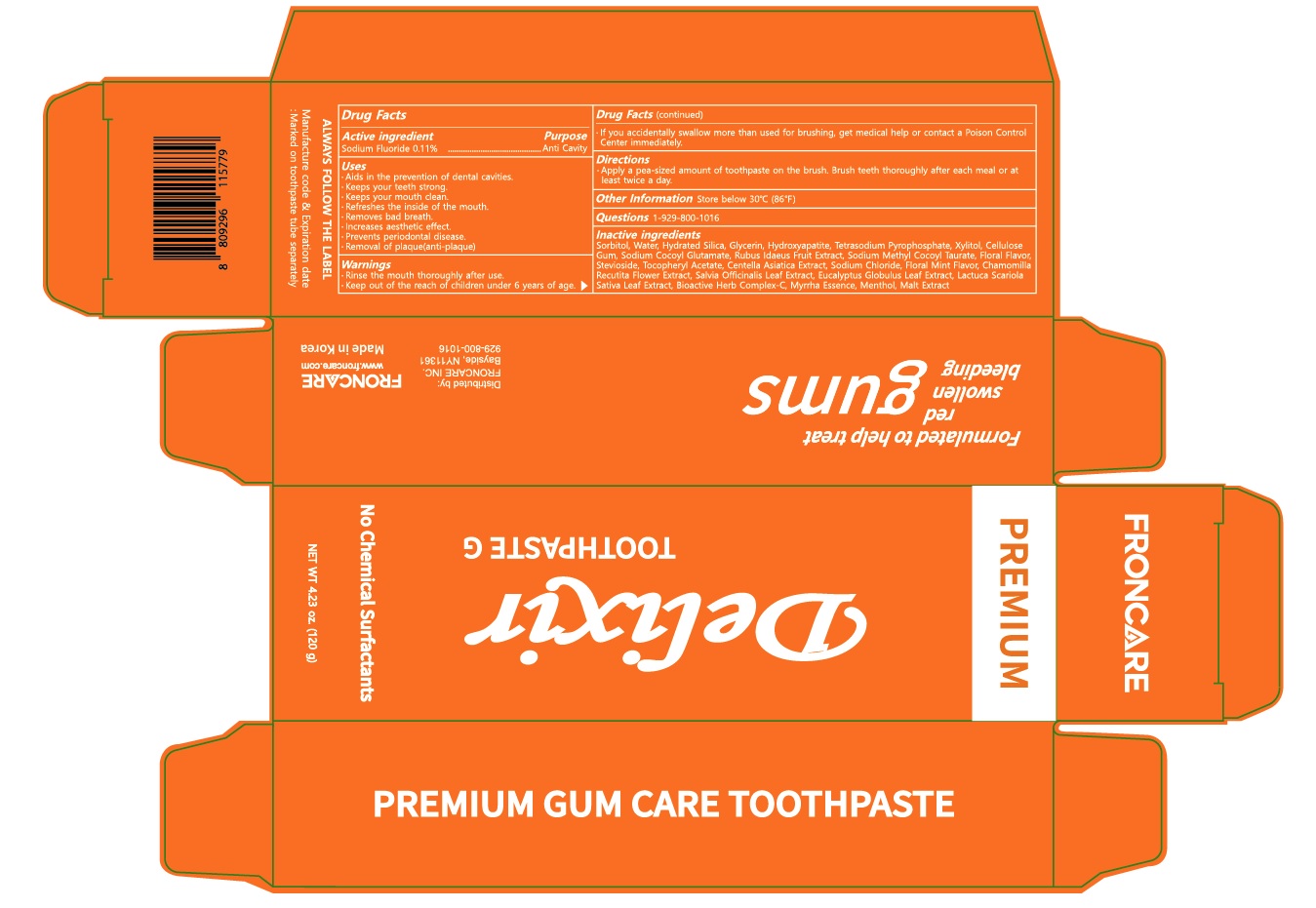
Label: DELIXIR G TOOTHPASTE- sodium fluoride paste, dentifrice
- NDC Code(s): 83466-010-01, 83466-010-02
- Packager: Froncare, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated May 17, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
-
Inactive ingredients
Sorbitol, Water, Hydrated Silica, Glycerin, Hydroxyapatite, Tetrasodium Pyrophosphate, Xylitol, Cellulose Gum, Sodium Cocoyl Glutamate, Rubus Idaeus Fruit Extract, Sodium Methyl Cocoyl Taurate, Floral Flavor, Stevioside, Tocopheryl Acetate, Centella Asiatica Extract, Sodium Chloride, Floral Mint Flavor, Chamomilla Recutita Flower Extract, Salvia Officinalis Leaf Extract, Eucalyptus Globulus Leaf Extract, Lactuca Scariola Sativa Leaf Extract, Bioactive Herb Complex-C, Myrrha Essence, Menthol, Malt Extract
- Purpose
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Use(s)
- Directions
- Other Information
- Questions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DELIXIR G TOOTHPASTE
sodium fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83466-010 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Sodium Fluoride (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 0.11 g in 100 g Inactive Ingredients Ingredient Name Strength Sorbitol (UNII: 506T60A25R) Water (UNII: 059QF0KO0R) Hydrated Silica (UNII: Y6O7T4G8P9) Glycerin (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83466-010-02 1 in 1 CARTON 05/01/2023 1 NDC:83466-010-01 120 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/01/2023 Labeler - Froncare, Inc. (040424783) Registrant - Froncare, Inc. (040424783) Establishment Name Address ID/FEI Business Operations DONG IL PHARMS CO., LTD. 557810721 manufacture(83466-010)