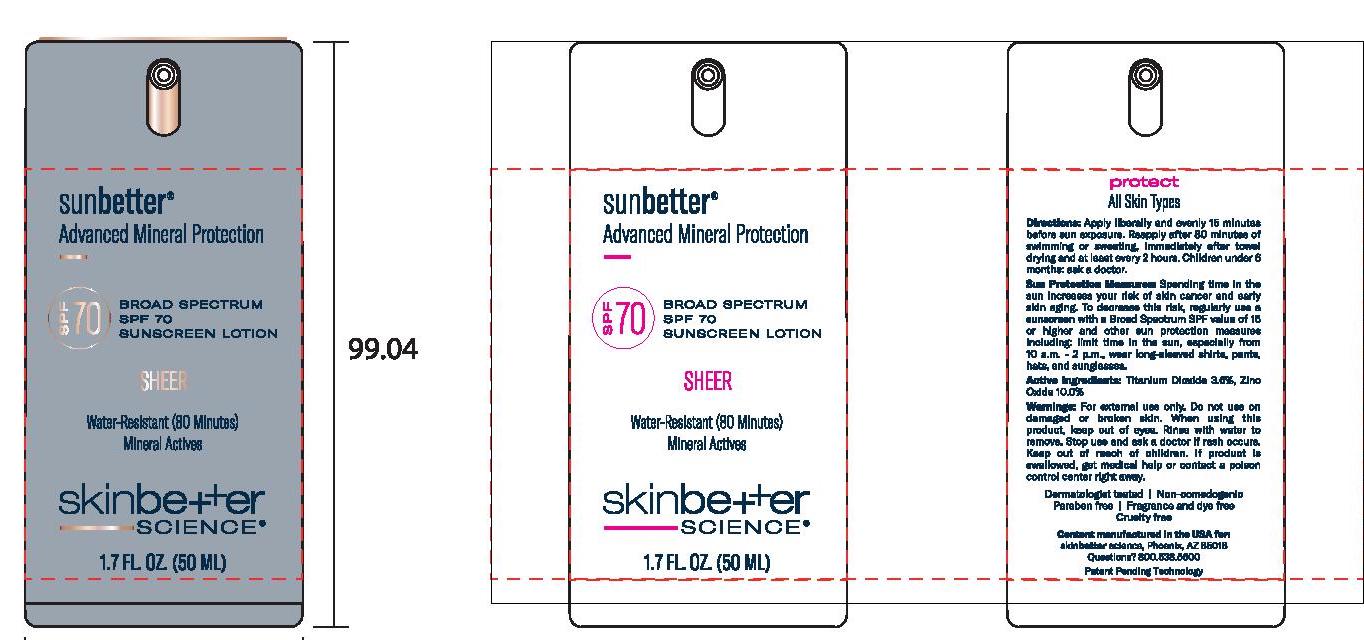
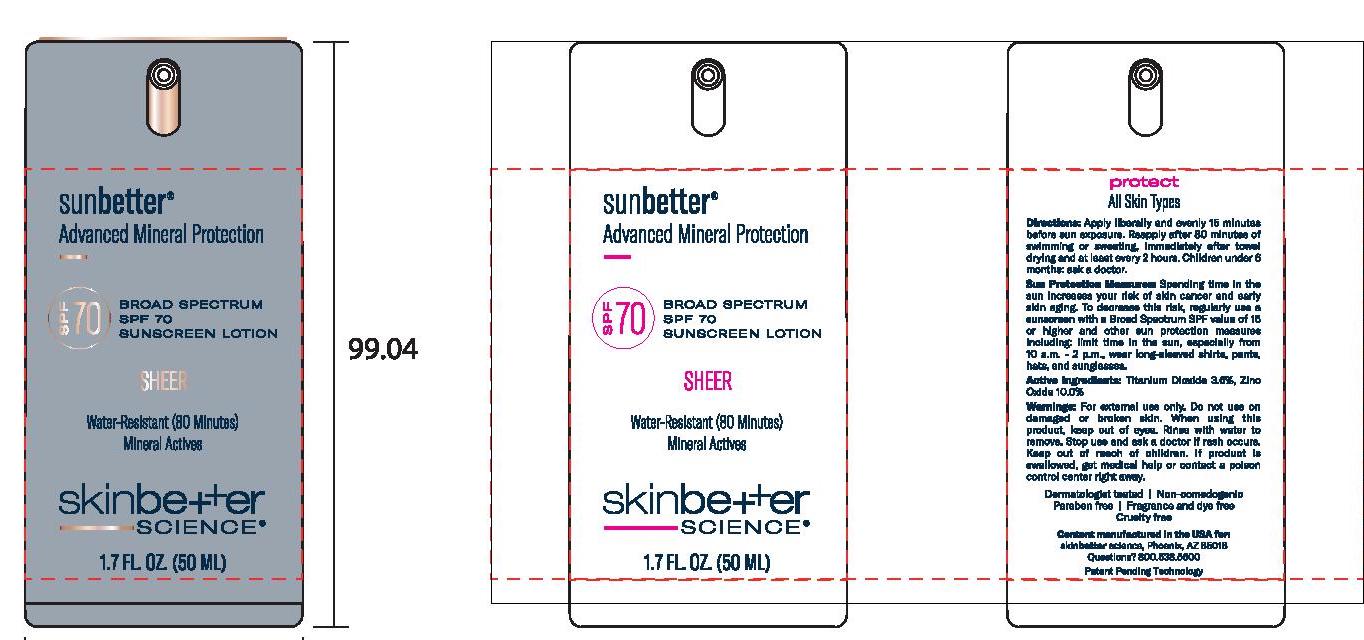
Label: SUNBETTER BROAD SPECTRUM SPF 70 SUNSCREEN SHEER- zinc oxide, titanium dioxide lotion
- NDC Code(s): 62282-377-01, 62282-377-02, 62282-377-03
- Packager: Northwest Cosmetic Laboratories, LLC D.B.A Elevation Labs Idaho
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 3, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
SUNBETTER BROAD SPECTRUM SPF 70 SUNSCREEN SHEER
zinc oxide, titanium dioxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62282-377 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 100 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 36 mg in 1 mL Inactive Ingredients Ingredient Name Strength MAGNESIUM POTASSIUM ALUMINOSILICATE FLUORIDE (UNII: YK3DC63Y5M) NIACINAMIDE (UNII: 25X51I8RD4) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) POLYGLYCERYL-4 OLEATE (UNII: 15B05TY4GX) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) ALUMINUM OXIDE (UNII: LMI26O6933) HIMANTHALIA ELONGATA (UNII: 21RND18XRR) PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) CERIC OXIDE (UNII: 619G5K328Y) STEARIC ACID (UNII: 4ELV7Z65AP) WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) CETYL DIMETHICONE 45 (UNII: IK315POC44) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CAPRYLYL GLYCOL (UNII: 00YIU5438U) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) POLY(METHYL METHACRYLATE; 450000 MW) (UNII: Z47NNT4J11) DODECAMETHYLPENTASILOXANE (UNII: 0QDQ2VQ5YJ) STYRENE/ACRYLAMIDE COPOLYMER (500000 MW) (UNII: 5Z4DPO246A) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) SQUALANE (UNII: GW89575KF9) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) PHENYL TRIMETHICONE (UNII: DR0K5NOJ4R) GLYCERIN (UNII: PDC6A3C0OX) SODIUM CHLORIDE (UNII: 451W47IQ8X) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) .ALPHA.-BISABOLOL, (+/-)- (UNII: 36HQN158VC) PANTHENOL (UNII: WV9CM0O67Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62282-377-01 1 in 1 CARTON 02/02/2021 1 50 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 2 NDC:62282-377-02 15 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 02/01/2021 3 NDC:62282-377-03 1 mL in 1 PACKET; Type 0: Not a Combination Product 02/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 02/01/2021 Labeler - Northwest Cosmetic Laboratories, LLC D.B.A Elevation Labs Idaho (929572014)