Label: ALCAFTADINE solution/ drops
- NDC Code(s): 68083-649-01, 68083-649-02
- Packager: Gland Pharma Limited
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Use
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
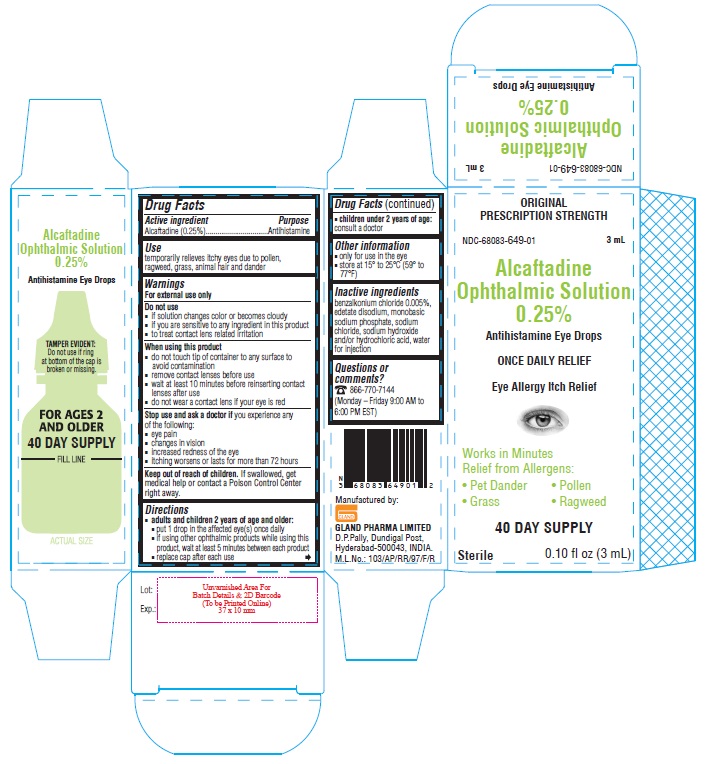
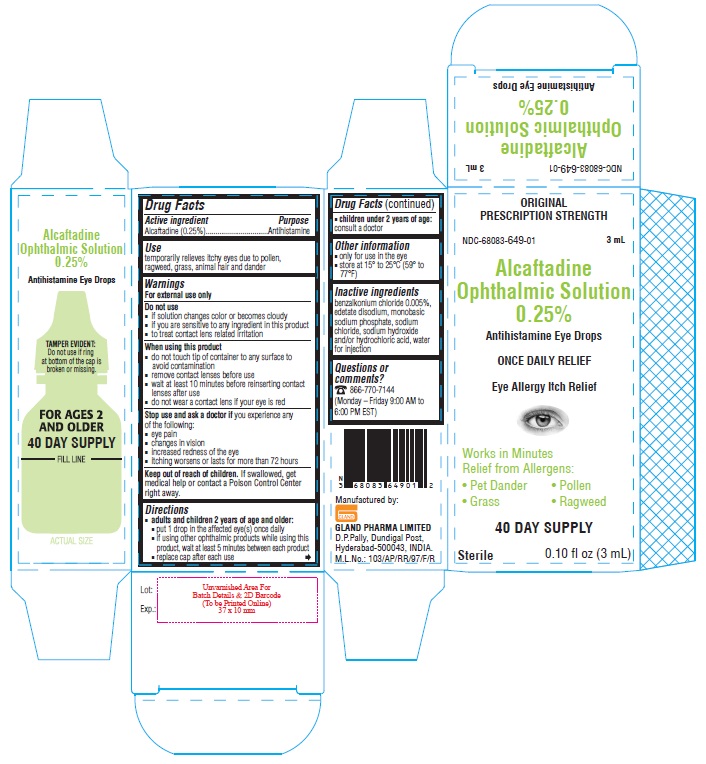
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL
Container Label - 0.25% (3 mL)
NDC-68083-649-01
Alcaftadine
Ophthalmic Solution
0.25%
Antihistamine Eye Drops
0.10 fl oz (3 mL) Sterile
Container Carton - 0.25% (3 mL)
ORIGINAL PRESCRIPTION STRENGTH
NDC-68083-649-01 3 mL
Alcaftadine
Ophthalmic Solution
0.25%
Antihistamine Eye Drops
ONCE DAILY RELIEF
Eye Allergy Itch Relief
Works in Minutes
Relief from Allergens:
• Pet Dander • Pollen
• Grass • Ragweed
40 DAY SUPPLYSterile 0.10 fl oz (3 mL)
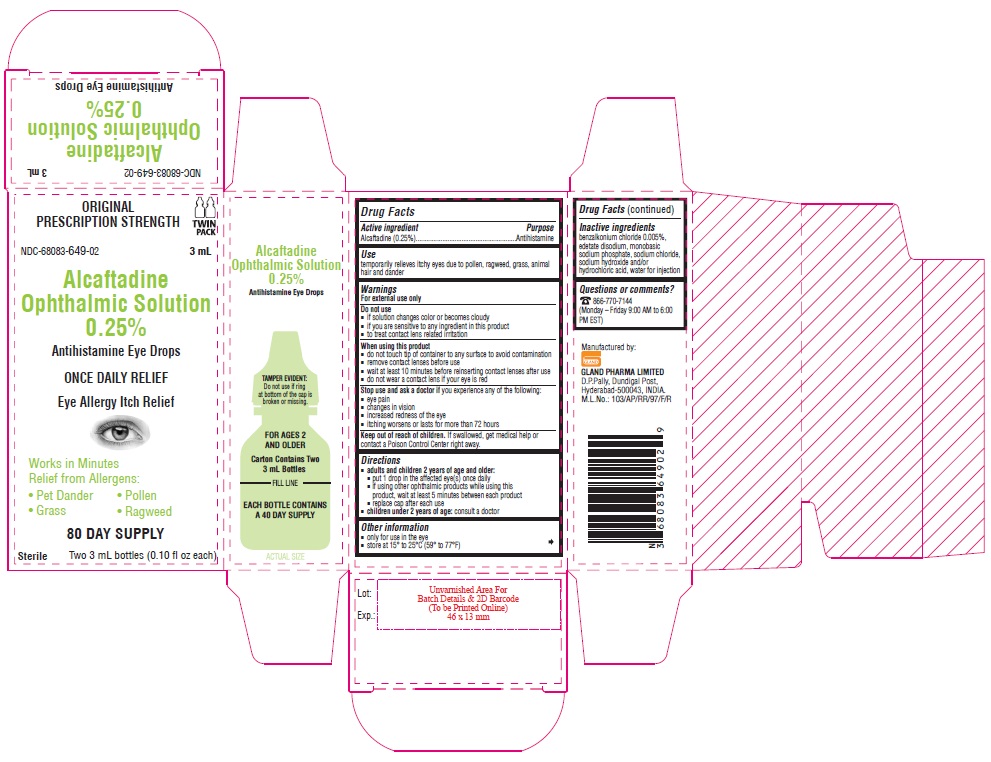
Container Carton - 0.25% (3 mL) - Twin Pack
ORIGINAL PRESCRIPTION STRENGTH
TWIN PACK
NDC-68083-649-02 3 mL
Alcaftadine
Ophthalmic Solution
0.25%
Antihistamine Eye Drops
ONCE DAILY RELIEF
Eye Allergy Itch Relief
Works in Minutes
Relief from Allergens:
• Pet Dander • Pollen
• Grass • Ragweed
80 DAY SUPPLY
Sterile Two 3 mL bottles (0.10 fl oz each)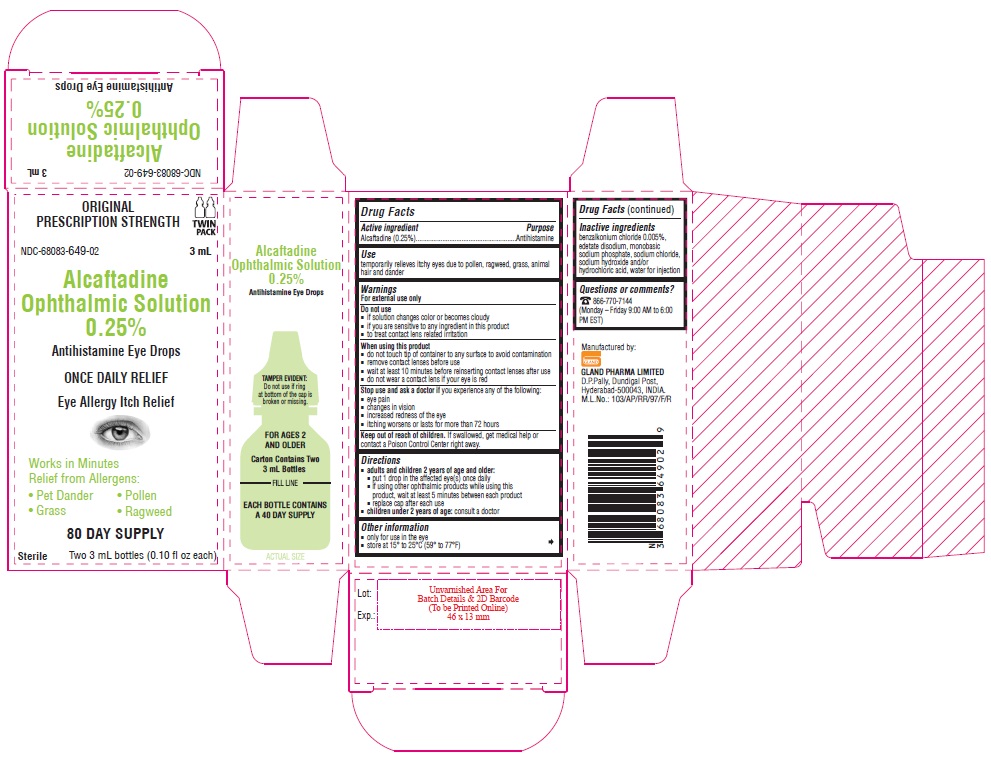
-
INGREDIENTS AND APPEARANCE
ALCAFTADINE
alcaftadine solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68083-649 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCAFTADINE (UNII: 7Z8O94ECSX) (ALCAFTADINE - UNII:7Z8O94ECSX) ALCAFTADINE 2.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) EDETATE DISODIUM (UNII: 7FLD91C86K) HYDROCHLORIC ACID (UNII: QTT17582CB) SODIUM PHOSPHATE, MONOBASIC (UNII: 3980JIH2SW) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68083-649-01 1 in 1 CARTON 03/01/2024 1 3 mL in 1 BOTTLE; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) 2 NDC:68083-649-02 2 in 1 CARTON 03/01/2024 2 3 mL in 1 BOTTLE; Type 9: Other Type of Part 3 Combination Product (e.g., Drug/Device/Biological Product) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209706 03/01/2024 Labeler - Gland Pharma Limited (918601238) Establishment Name Address ID/FEI Business Operations GLAND PHARMA LIMITED 918601238 ANALYSIS(68083-649) , LABEL(68083-649) , MANUFACTURE(68083-649) , PACK(68083-649)