Label: FLAVOBAC HEALTH GUARD COMPLETE- mouth spray against sore throat pain irritation spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 77982-111-11 - Packager: Laboratoires MSP Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 29, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
-
Warnings
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, swelling, nausea or vomiting, consult a doctor promptly
Stop use and ask a doctor or dentist if sore mouth symptoms do not improve in 7 days
If pregnant or breast-feeding, ask a health professional before useKeep out of the reach of children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Do not exceed the recommended dosage. When using this product, try to avoid swallowing spray - WHEN USING
- PREGNANCY
- OTHER SAFETY INFORMATION
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
Directions
adults and children 2 years of age and older: apply 4 sprays to the affected area. Gargle, swish around, or allow to remain in place at least 1 minute and then spit out. Use up to 4 times daily or as directed by a dentist or doctor
children under 12 years of age: should be supervised in the use of this product
children under 2 years of age: not recommended
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
FLAVOBAC HEALTH GUARD COMPLETE
mouth spray against sore throat pain irritation sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:77982-111 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 10 mg in 100 mL MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.04 mg in 100 mL Inactive Ingredients Ingredient Name Strength POTASSIUM SORBATE (UNII: 1VPU26JZZ4) XYLITOL (UNII: VCQ006KQ1E) POLYSORBATE 20 (UNII: 7T1F30V5YH) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) STEVIA LEAF (UNII: 6TC6NN0876) ALCOHOL (UNII: 3K9958V90M) MINT (UNII: FV98Z8GITP) CITRUS BIOFLAVONOIDS (UNII: BD70459I50) WATER (UNII: 059QF0KO0R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:77982-111-11 1 in 1 BOX 01/15/2021 1 59 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 01/15/2021 Labeler - Laboratoires MSP Inc (251046124)

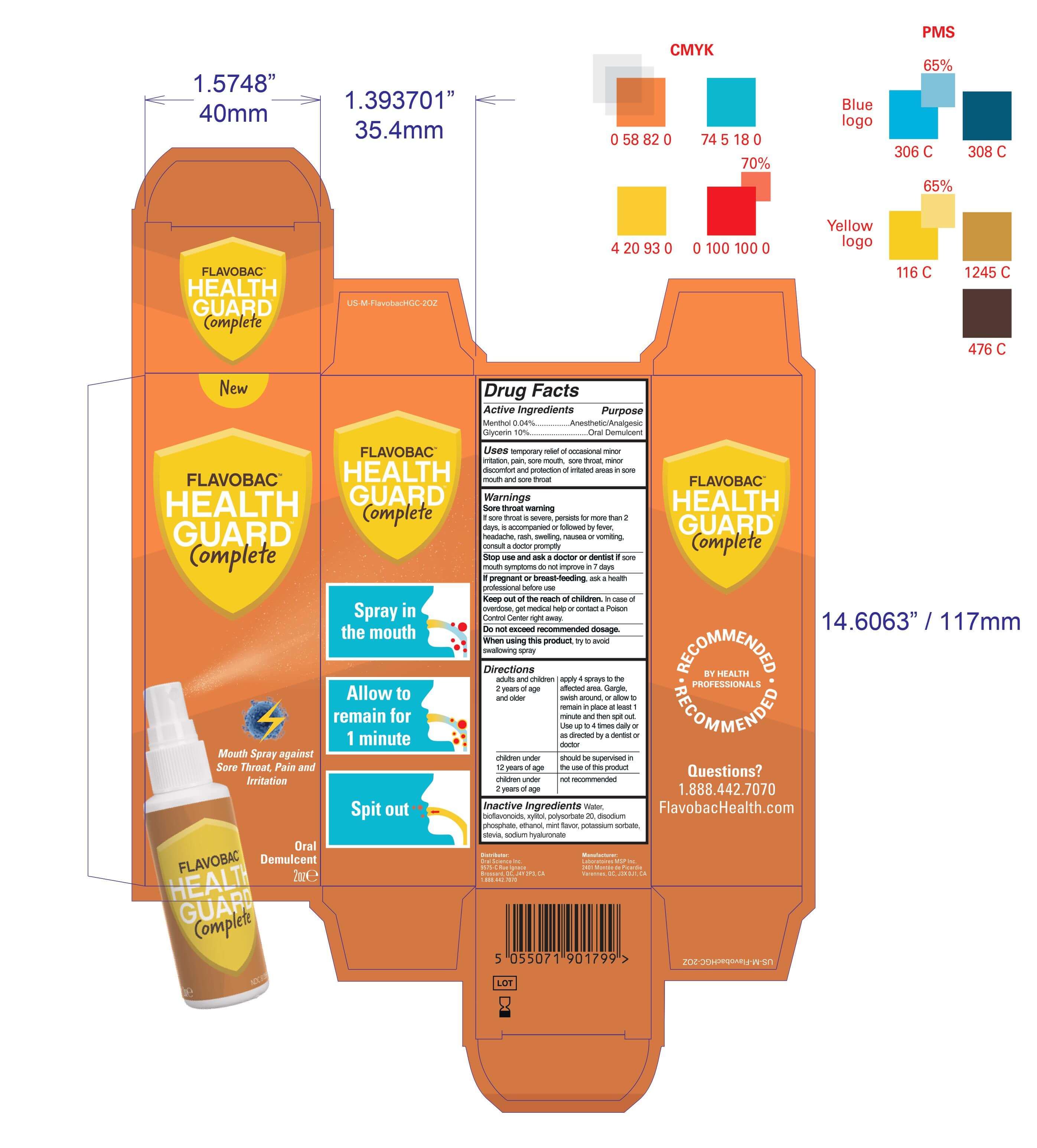
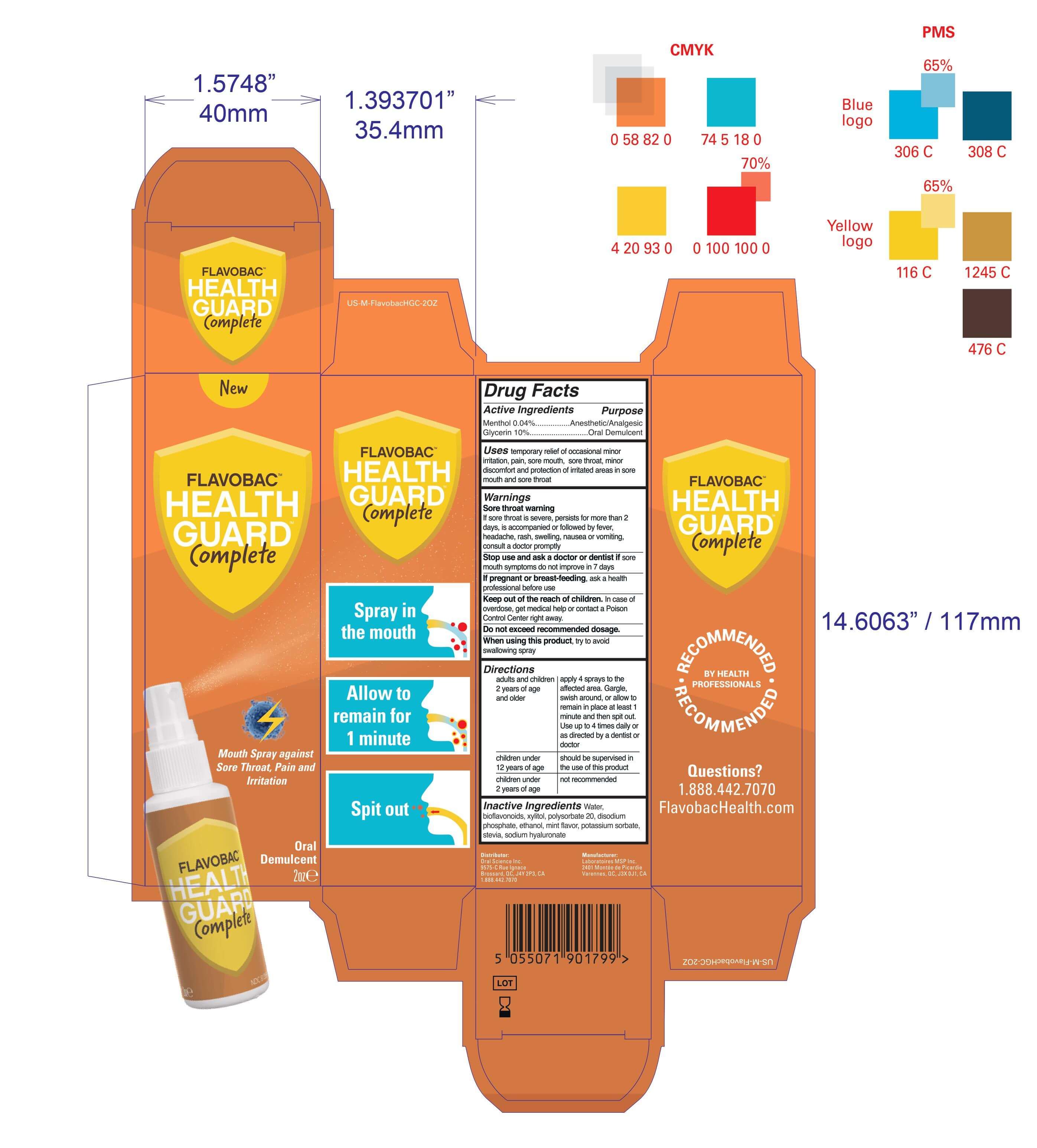 59 mL NDC: 81359-111-11
59 mL NDC: 81359-111-11