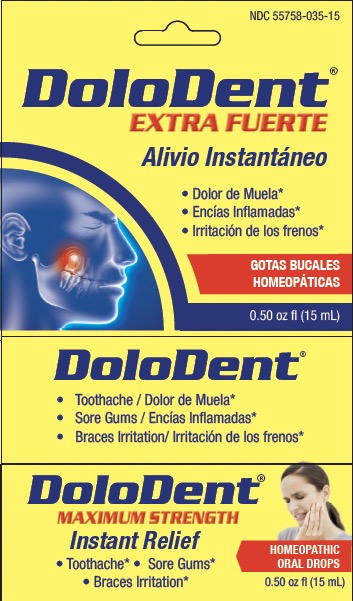
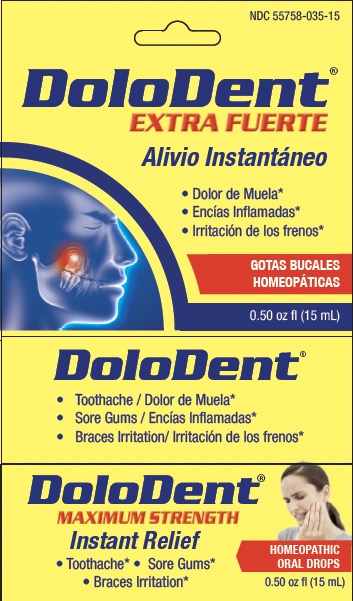
Label: DOLODENT- arnica montana, chamomilla, hecla lava, hypericum, asafoedida liquid
- NDC Code(s): 55758-324-15
- Packager: Pharmadel LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated September 7, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
-
Active ingredients & Purposes
Active ingredients** (HPUS) Purposes* Arnica montana 30X Pain in the tooth, with inflammation Asafoetida 6X Cavity pain Chamomilla vulgaris 30X Irritation, toothache, sore gums Hekla lava 30X Tooth sensitive to pressure, with swollen gums Hypericum perforatum 30X Sharp shooting pain, due to toothache
**The letters HPUS indicate that this ingredient is officially included in the Homeopathic Pharmacopeia of the United States.
- Uses*
- Warnings
- Directions
- Inactive ingredients
- Question & comments?
- Distributed by:/ Distribuido por:
- Homeopathic Statements
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
DOLODENT
arnica montana, chamomilla, hecla lava, hypericum, asafoedida liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55758-324 Route of Administration BUCCAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ARNICA MONTANA (UNII: O80TY208ZW) (ARNICA MONTANA - UNII:O80TY208ZW) ARNICA MONTANA 30 [hp_X] in 1 mL CHAMOMILE (UNII: FGL3685T2X) (CHAMOMILE - UNII:FGL3685T2X) CHAMOMILE 30 [hp_X] in 1 mL FERULA ASSA-FOETIDA RESIN (UNII: W9FZA51AS1) (FERULA ASSA-FOETIDA RESIN - UNII:W9FZA51AS1) FERULA ASSA-FOETIDA RESIN 6 [hp_X] in 1 mL HYPERICUM PERFORATUM (UNII: XK4IUX8MNB) (HYPERICUM PERFORATUM - UNII:XK4IUX8MNB) HYPERICUM PERFORATUM 30 [hp_X] in 1 mL HEKLA LAVA (UNII: C21158IIRK) (HEKLA LAVA - UNII:C21158IIRK) HEKLA LAVA 30 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength CLOVE OIL (UNII: 578389D6D0) GLYCERIN (UNII: PDC6A3C0OX) PROPOLIS WAX (UNII: 6Y8XYV2NOF) POLYSORBATE 20 (UNII: 7T1F30V5YH) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55758-324-15 1 in 1 BOX 09/09/2023 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 11/01/2017 Labeler - Pharmadel LLC (030129680)