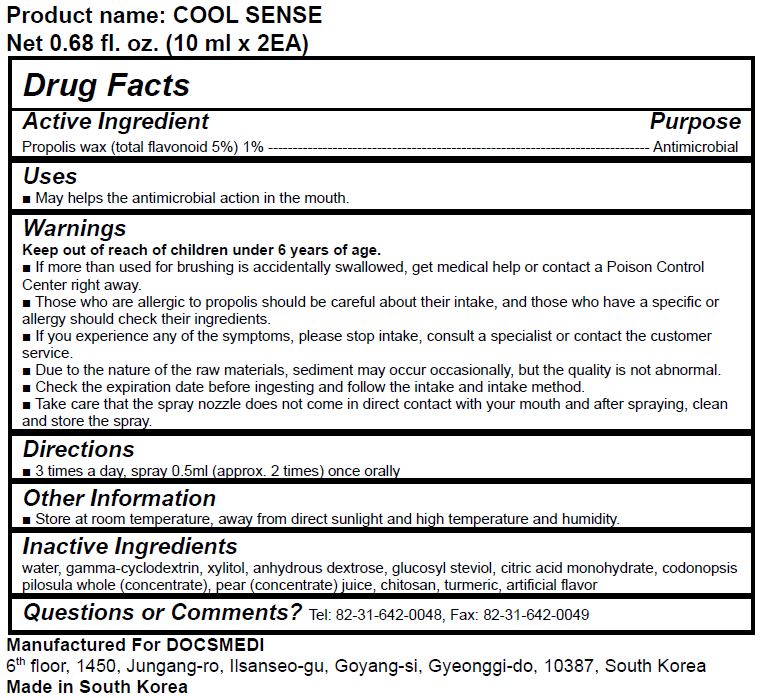
Label: COOL SENSE- propolis wax liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 73706-0001-1, 73706-0001-2, 73706-0001-3 - Packager: Bncare Agriculture Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated March 19, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
- Those who are allergic to propolis should be careful about their intake, and those who have a specific or allergy should check their ingredients.
- If you experience any of the symptoms, please stop intake, consult a specialist or contact the customer service.
- Due to the nature of the raw materials, sediment may occur occasionally, but the quality is not abnormal.
- Check the expiration date before ingesting and follow the intake and intake method.
- Take care that the spray nozzle does not come in direct contact with your mouth and after spraying, clean and store the spray.
- INACTIVE INGREDIENT
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
COOL SENSE
propolis wax liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73706-0001 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PROPOLIS WAX (UNII: 6Y8XYV2NOF) (PROPOLIS WAX - UNII:6Y8XYV2NOF) PROPOLIS WAX 1 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73706-0001-2 2 in 1 CARTON 03/17/2020 1 NDC:73706-0001-1 10 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 2 NDC:73706-0001-3 2 in 1 BLISTER PACK 03/17/2020 2 NDC:73706-0001-1 10 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/17/2020 Labeler - Bncare Agriculture Co., Ltd. (688206563) Registrant - Bncare Agriculture Co., Ltd. (688206563) Establishment Name Address ID/FEI Business Operations Bncare Agriculture Co., Ltd. 688206563 manufacture(73706-0001)