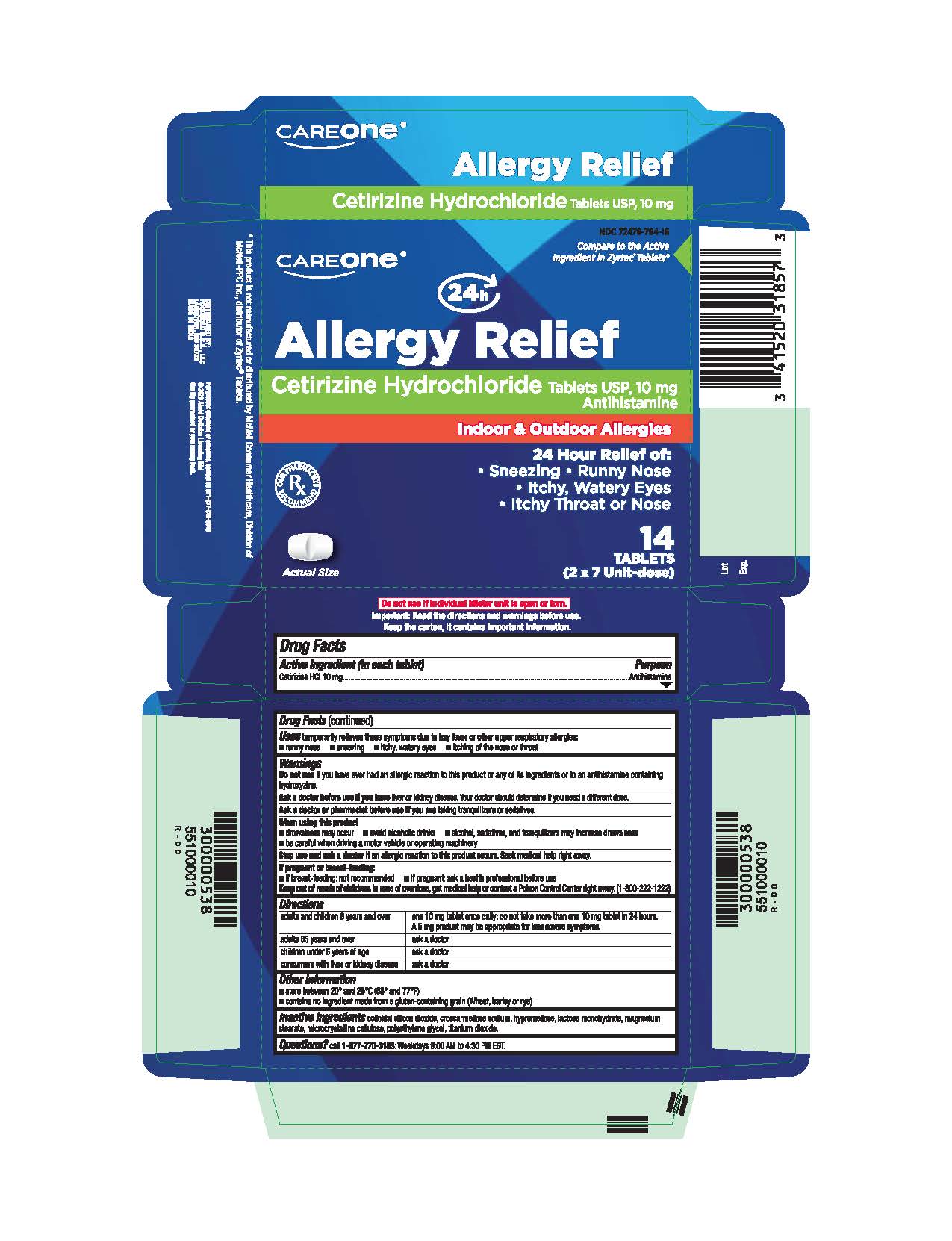
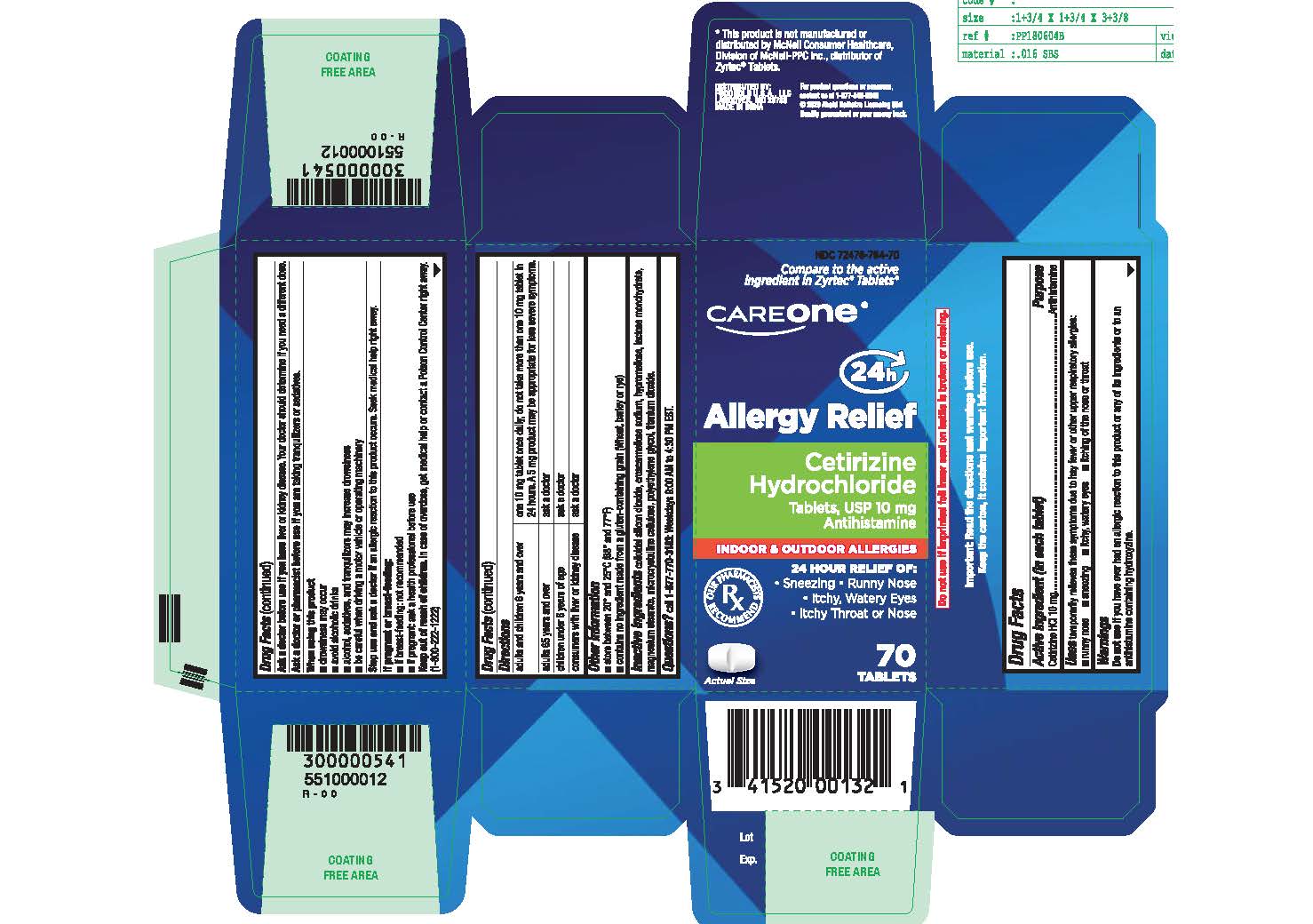
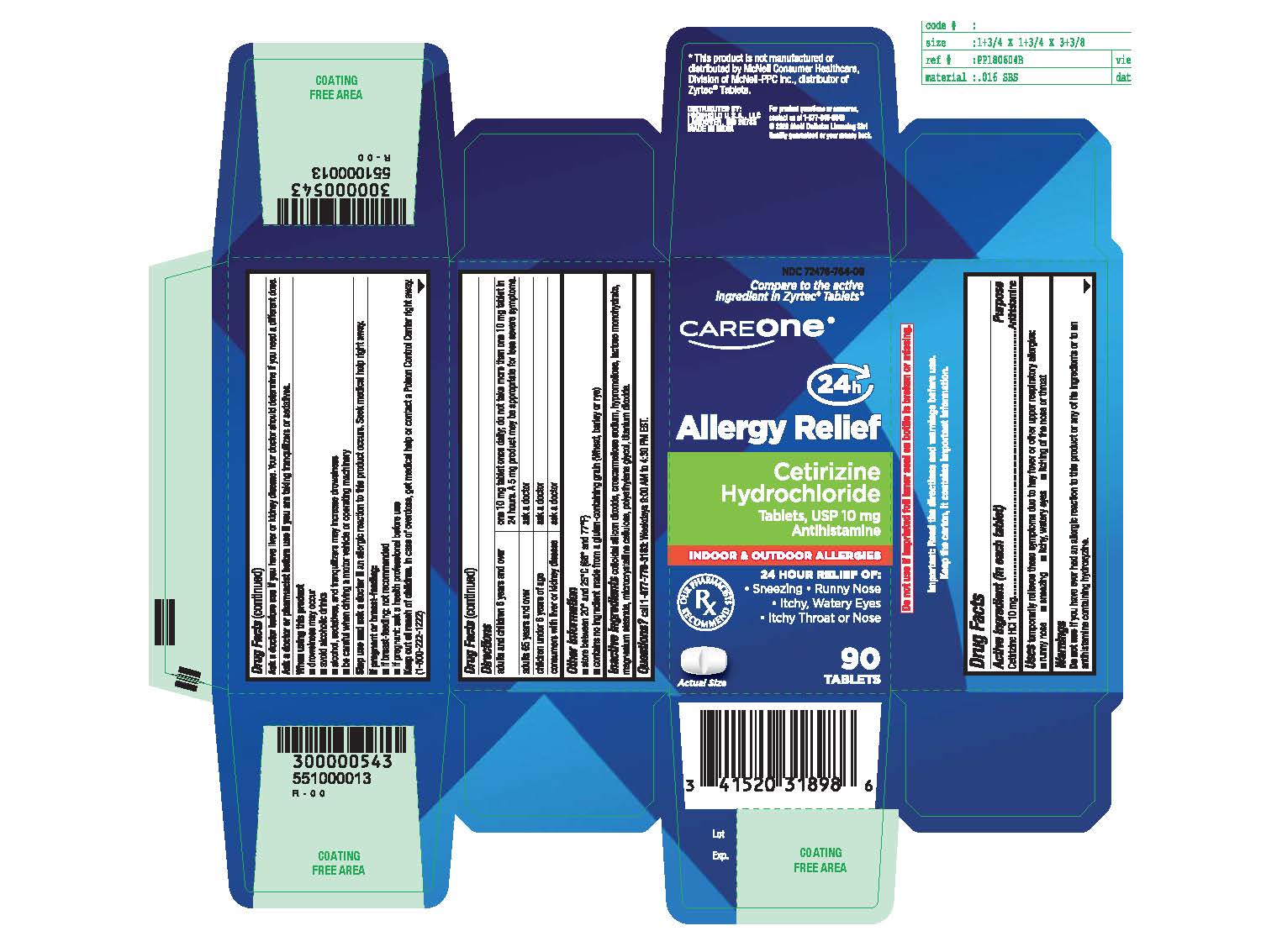
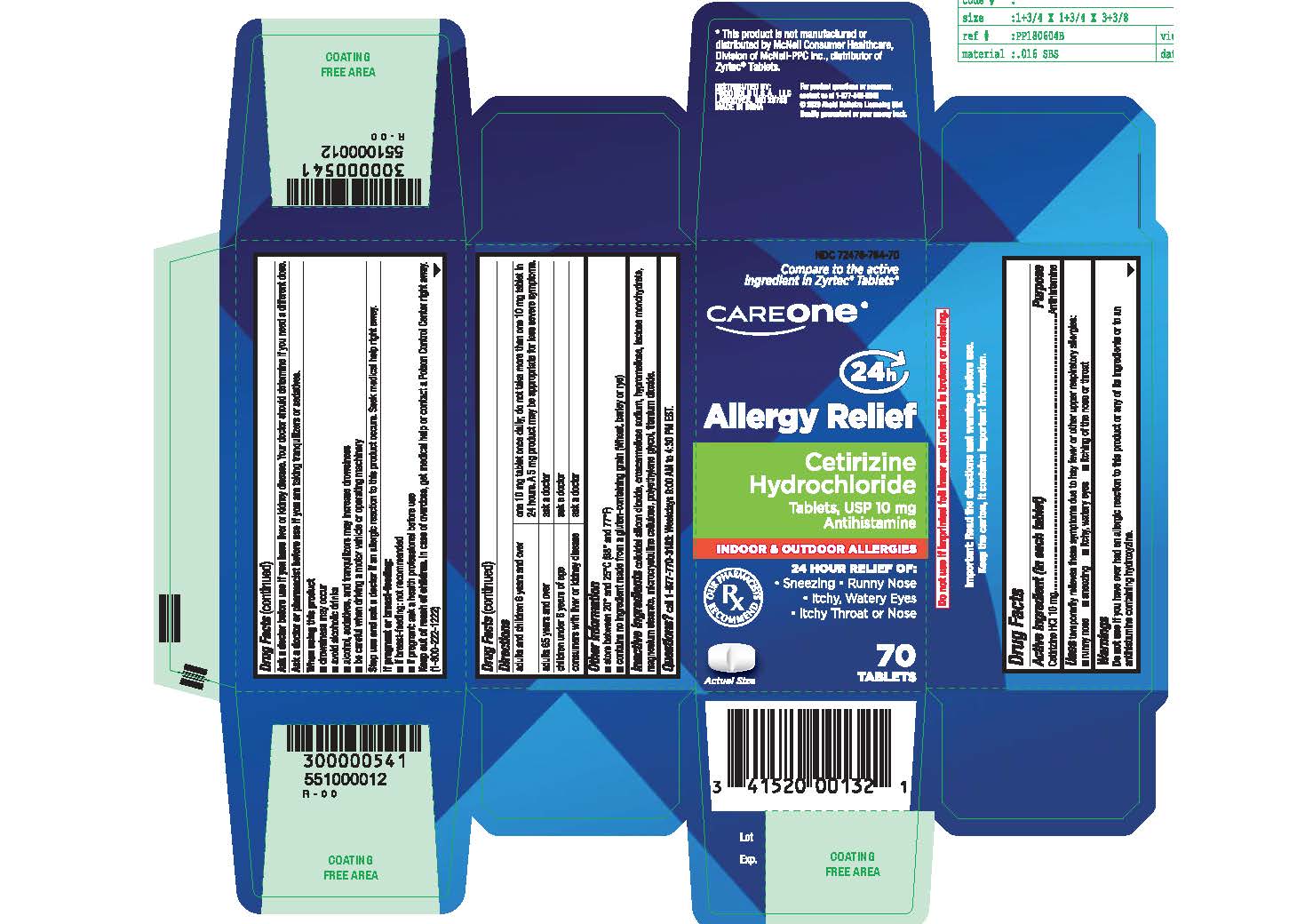
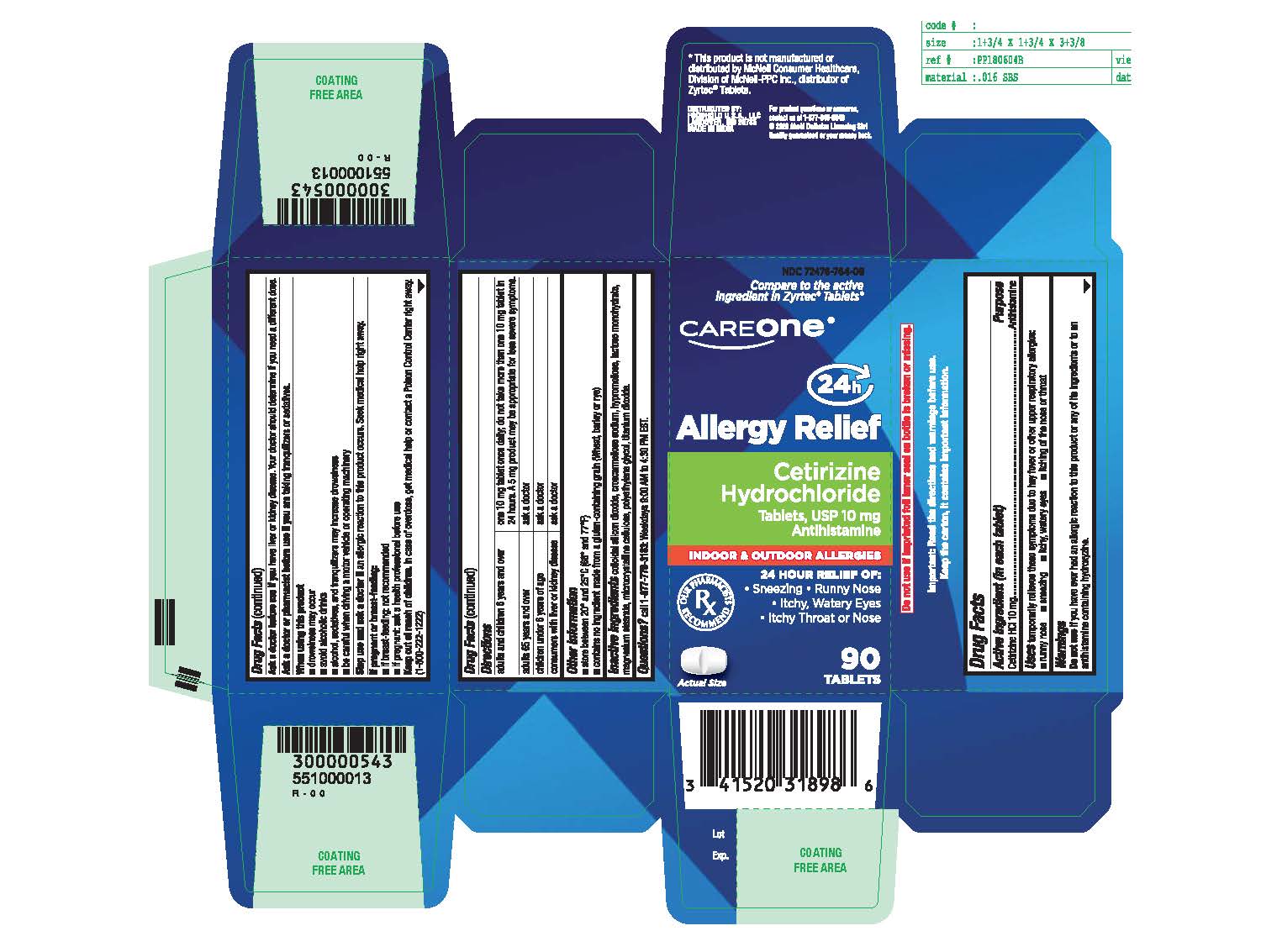
Label: CETIRIZINE HYDROCHLORIDE tablet
-
NDC Code(s):
72476-764-03,
72476-764-09,
72476-764-16,
72476-764-23, view more72476-764-70
- Packager: RETAIL BUSINESS SERVICES,LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 31, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each tablet)
- Purpose
- Uses
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding:
- Keep out of reach of children
-
Directions
adults and children 6 years and over
- one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours.
- A 5 mg product may be appropriate for less severe symptoms.
adults 65 years and over
- ask a doctor
children under 6 years of age
- ask a doctor
consumers with liver or kidney disease
- ask a doctor - Other information
- Inactive ingredients
- Questions?
- PDP
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72476-764 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (White to off-white) Score 2 pieces Shape RECTANGLE (round-off rectangular shaped tablets) Size 9mm Flavor Imprint Code G;4 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72476-764-16 14 in 1 BLISTER PACK; Type 0: Not a Combination Product 04/21/2020 2 NDC:72476-764-09 90 in 1 BOTTLE; Type 0: Not a Combination Product 04/21/2020 12/31/2024 3 NDC:72476-764-23 120 in 1 BOTTLE; Type 0: Not a Combination Product 04/21/2020 4 NDC:72476-764-03 30 in 1 BOTTLE; Type 0: Not a Combination Product 04/21/2020 5 NDC:72476-764-70 70 in 1 BOTTLE; Type 0: Not a Combination Product 04/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209274 04/21/2020 Labeler - RETAIL BUSINESS SERVICES,LLC. (967989935)