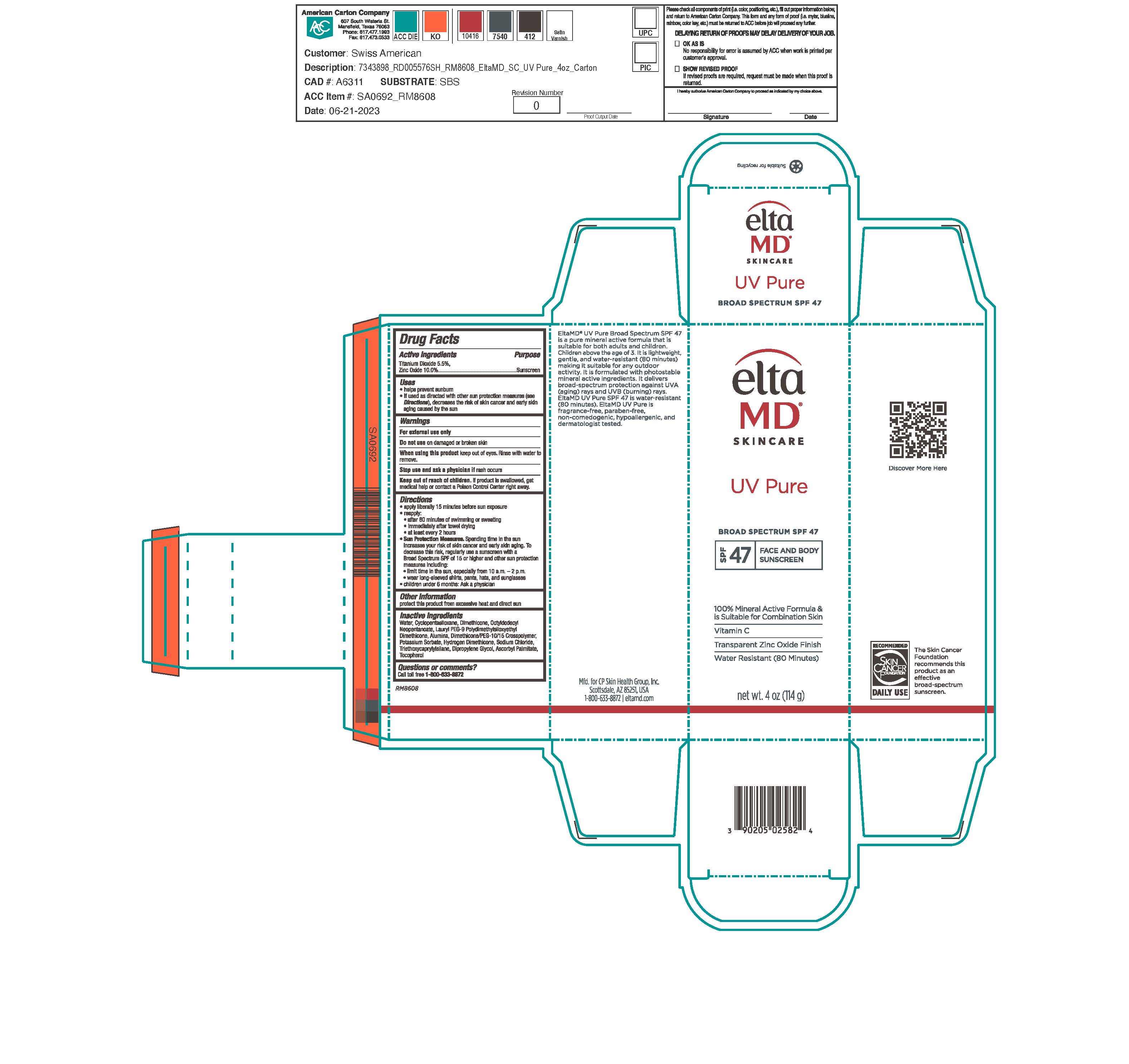
Label: ELTAMD UV PURE SPF47- zinc oxide and titanium dioxide sunscreen cream
- NDC Code(s): 72043-2582-2, 72043-2582-4
- Packager: CP Skin Health Group, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 27, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Warnings
- Active Ingredients
- Uses
- Uses
-
Directions
Apply liberally 15 minutes before sun exposure Reapply after 80 minutes of swimming or sweating immediately after towel drying at least every 2 hours Sun Protection Measures. Spending time int he sun increases your risk of skin cancer and early skin aging. To decrease this risk, egularly use a sunscreen with a broad-spectrum SPF of 15 or higher and other sun protection measures including: limit time in the sun, especially from 10 a.m. - 2 p.m. wear long-sleeve shirts, pants, hats and sunglasses Children under 6 months: Ask a physician.
- Inactive Ingredients
- KEEP OUT OF REACH OF CHILDREN
- Labeling
-
INGREDIENTS AND APPEARANCE
ELTAMD UV PURE SPF47
zinc oxide and titanium dioxide sunscreen creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72043-2582 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 100 g in 1000 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 55 g in 1000 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM CHLORIDE (UNII: 451W47IQ8X) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) DIMETHICONE (UNII: 92RU3N3Y1O) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) ASCORBYL PALMITATE (UNII: QN83US2B0N) LAURYL PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: 25G622K2RA) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) HYDROGEN DIMETHICONE (13 CST) (UNII: 4QGR4P2YOI) ALUMINUM OXIDE (UNII: LMI26O6933) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72043-2582-4 114 g in 1 BOTTLE; Type 0: Not a Combination Product 01/10/2018 2 NDC:72043-2582-2 2 g in 1 PACKET; Type 0: Not a Combination Product 07/06/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/10/2018 Labeler - CP Skin Health Group, Inc. (611921669) Registrant - Swiss-American CDMO, LLC (080170933) Establishment Name Address ID/FEI Business Operations Swiss-American CDMO, LLC 080170933 manufacture(72043-2582)