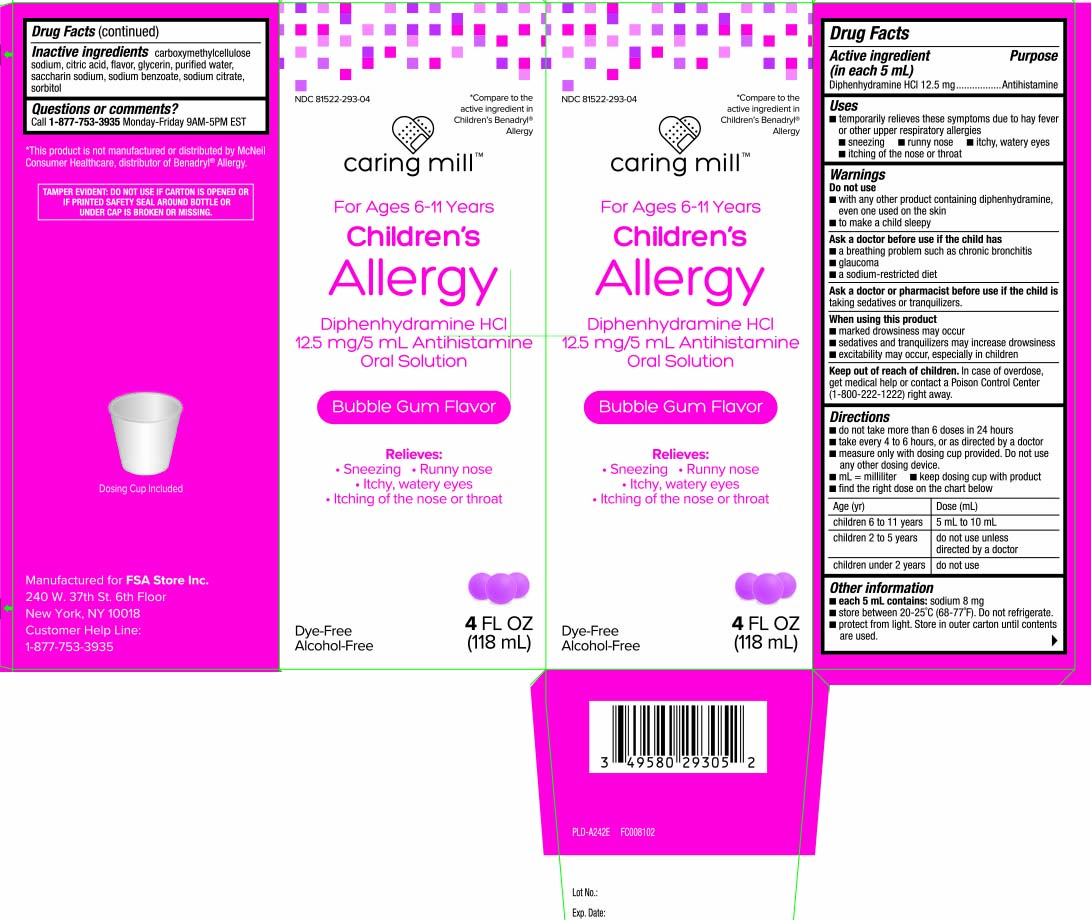
Label: CHILDRENS ALLERGY RELIEF DYE FREE BUBBLE GUM- diphenhydramine hydrochloride liquid
- NDC Code(s): 81522-293-04
- Packager: FSA Store Inc. (Caring Mills)
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 5 mL)
- Purpose
- Uses
-
Warnings
Do not use
- with any other product containing diphenhydramine, even one used on skin
- to make a child sleepy
Ask a doctor before use if the child has
- a breathing problem such as chronic bronchitis
- glaucoma
- a sodium-restricted diet
-
Directions
- do not take more than 6 doses in 24 hours
- take every 4 to 6 hours, or as directed by a doctor
- measure only with dosing cup provided. Do not use any other dosing device.
- mL = milliliter
- keep dosing cup with product
- find the right dose on the chart below
age (yr) dose (mL) children 6 to 11 years 5 mL to 10 mL children 2 to 5 years do not use unless directed by a doctor children under 2 years do not use
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
*Compare to the active ingredient in Children's Benadryl® Allergy
For Ages 6-11 Years
Children's Allergy Relief
Diphenhydramine HCI 125 mg/5 mL
Antihistamine Oral Solution
Bubble Gum Flavor
Dye-Free
Alcohol-Free
Relieves:
- Sneezing
- Runny nose
- Itchy, watery eyes
- Itching of the nose or throat
Dosing Cup included
FL OZ (mL)
*This product is not manufactured or distributed by McNeil Consumer Healthcare, distributor of Benadryl® Allergy.
TAMPER EVIDENT: DO NOT USE IF CARTON IS OPENED OR IF PRINTED SAFETY SEAL AROUND BOTTLE OR UNDER CAP IS BROKEN OR MISSING.
Manufactured for FSA Store Inc.
240 W. 37th St., 6th Floor
New York, NY 10018
- Packaging Label
-
INGREDIENTS AND APPEARANCE
CHILDRENS ALLERGY RELIEF DYE FREE BUBBLE GUM
diphenhydramine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81522-293 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SACCHARIN SODIUM (UNII: SB8ZUX40TY) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) SORBITOL (UNII: 506T60A25R) Product Characteristics Color Score Shape Size Flavor BUBBLE GUM Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81522-293-04 1 in 1 CARTON 04/18/2022 1 118 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/18/2022 Labeler - FSA Store Inc. (Caring Mills) (049283340)