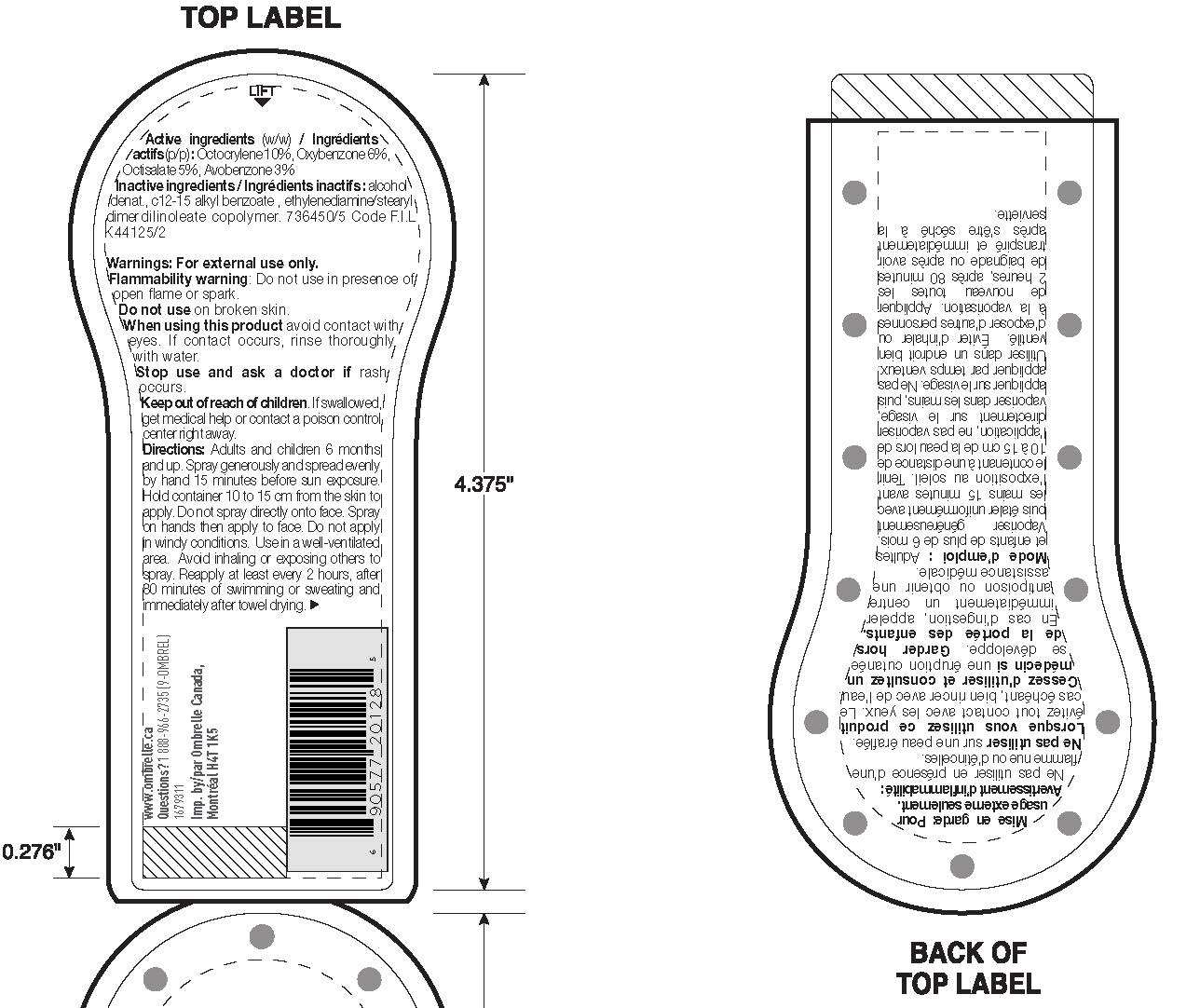
Label: GARNIER OMBRELLE SPORT VAPORISATEUR BROAD SPECTRUM SPF 30 SWEAT RESISTANT 80 MINUTES- avobenzone, octisalate, octocrylene and oxybenzone spray
- NDC Code(s): 49967-772-01
- Packager: L'OREAL USA PRODUCTS INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Export only
Drug Label Information
Updated January 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
-
Warnings
For external use only. Flammability Warning. Do not use in presence of open flame or spark. Do not use on broken skin. When using this product avoid contact with eyes. If contact occurs, rinse thoroughlly with water. Stop use and ask a doctor if rash occurs. Keep out of reach of children. If swallowed get medical help or contact a poison control center right away.
-
Directions
Adults and children over 6 months and up. Spray generously and spread evenly by hand 15 minutes before sun exposure. Hold container 10 to 15 cm from the skin to apply. Do not spray directly onto face. Spray on hands then apply to face. Do not apply in windy conditions. Use in a well-ventilated area. Reapply at least every 2 hours, after 80 minutes of swimming or sweating, and immediately after towel drying.
- Others
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
GARNIER OMBRELLE SPORT VAPORISATEUR BROAD SPECTRUM SPF 30 SWEAT RESISTANT 80 MINUTES
avobenzone, octisalate, octocrylene and oxybenzone sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49967-772 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 100 mg in 1 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 60 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49967-772-01 145 mL in 1 CONTAINER; Type 0: Not a Combination Product 01/01/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date export only 01/01/2021 Labeler - L'OREAL USA PRODUCTS INC (002136794) Establishment Name Address ID/FEI Business Operations Dimensional Merchandising Inc. 076693183 manufacture(49967-772)