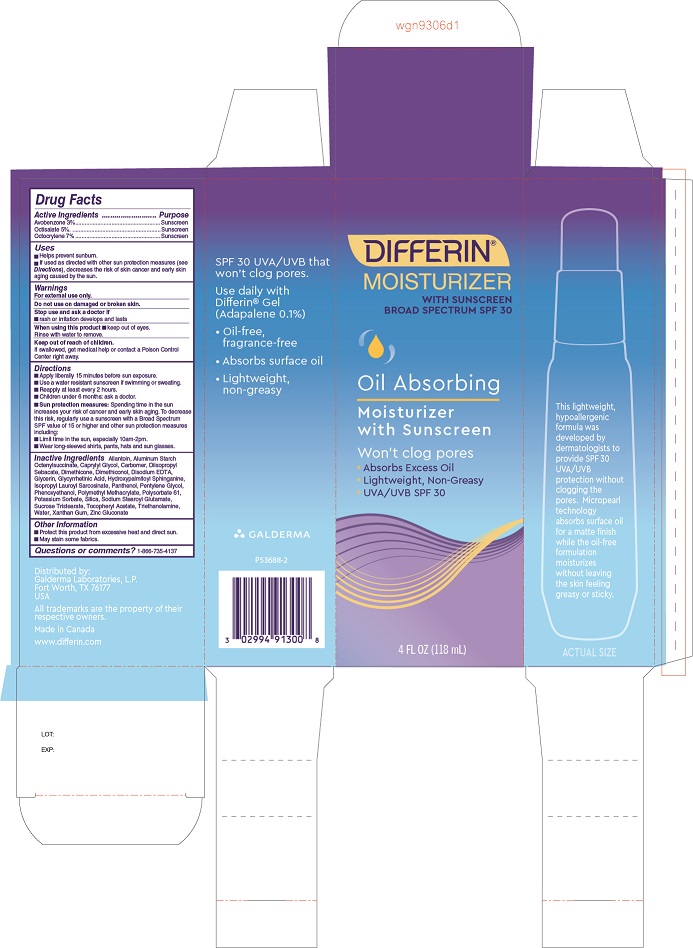
Label: DIFFERIN OIL ABSORBING MOISTURIZER SPF 30- octocrylene, octisalate, avobenzone lotion
- NDC Code(s): 0299-4607-04
- Packager: Galderma Laboratories, L.P.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Uses
- Warnings
- Keep out of reach of children.
-
Directions
• Apply liberally 15 minutes before sun exposure.
• Use a water resistant sunscreen if swimming or sweating.
• Reapply at least every 2 hours.
• Children under 6 months: Ask a doctor.• Sun protection measures: Spending time in the sun increases your risk of cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• Limit time in the sun, especially 10am - 2pm.
• Wear long-sleeved shirts, pants, hats, and sun glasses.
-
Inactive Ingredients
Allantoin, Aluminum Starch Octenylsuccinate, Caprylyl Glycol, Carbomer, Diisopropyl Sebacate, Dimethicone, Dimethiconol, Disodium EDTA, Glycerin, Glycyrrhetinic Acid, Hydroxypalmitoyl Sphinganine, Isopropyl Lauroyl Sarcosinate, Panthenol, Pentylene Glycol, Phenoxyethanol, Polymethyl Methacrylate, Polysorbate 61, Potassium Sorbate, Silica, Sodium Stearoyl Glutamate, Sucrose Tristearate, Tocopheryl Acetate, Triethanolamine, Water, Xanthan Gum, Zinc Gluconate
- Other Information
- Questions or comments?
- QUESTIONS
- Principle Display Panel - 4 FL OZ carton
-
INGREDIENTS AND APPEARANCE
DIFFERIN OIL ABSORBING MOISTURIZER SPF 30
octocrylene, octisalate, avobenzone lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0299-4607 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Octocrylene (UNII: 5A68WGF6WM) (Octocrylene - UNII:5A68WGF6WM) Octocrylene 7 g in 100 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 5 g in 100 mL Avobenzone (UNII: G63QQF2NOX) (Avobenzone - UNII:G63QQF2NOX) Avobenzone 3 g in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Glycerin (UNII: PDC6A3C0OX) Dimethicone (UNII: 92RU3N3Y1O) Diisopropyl Sebacate (UNII: J8T3X564IH) Silicon Dioxide (UNII: ETJ7Z6XBU4) Aluminum Starch Octenylsuccinate (UNII: I9PJ0O6294) Pentylene Glycol (UNII: 50C1307PZG) Sodium Stearoyl Glutamate (UNII: 65A9F4P024) Phenoxyethanol (UNII: HIE492ZZ3T) Caprylyl Glycol (UNII: 00YIU5438U) .alpha.-tocopherol Acetate (UNII: 9E8X80D2L0) Enoxolone (UNII: P540XA09DR) Panthenol (UNII: WV9CM0O67Z) Trolamine (UNII: 9O3K93S3TK) Allantoin (UNII: 344S277G0Z) Potassium Sorbate (UNII: 1VPU26JZZ4) Zinc Gluconate (UNII: U6WSN5SQ1Z) Xanthan Gum (UNII: TTV12P4NEE) Edetate Disodium (UNII: 7FLD91C86K) Hydroxypalmitoyl Sphinganine (UNII: NR33W2353T) Carboxypolymethylene (UNII: 0A5MM307FC) Isopropyl Lauroyl Sarcosinate (UNII: LYR06W430J) Sucrose Tristearate (UNII: 71I93STU5M) Dimethiconol (2000 Cst) (UNII: T74O12AN6Y) Poly(methyl Methacrylate; 450000 Mw) (UNII: Z47NNT4J11) Polysorbate 61 (UNII: X9E1MY2JQG) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0299-4607-04 1 in 1 CARTON 11/01/2017 04/01/2025 1 118 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/01/2017 04/01/2025 Labeler - Galderma Laboratories, L.P. (047350186) Establishment Name Address ID/FEI Business Operations G Production Inc. 251676961 manufacture(0299-4607)