Label: LEUCILLIN- hypochlorous acid liquid
-
NDC Code(s):
86162-050-00,
86162-050-01,
86162-050-15,
86162-050-25, view more86162-050-50, 86162-050-60
- Packager: LYVLEE LIMITED
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Indications
- Directions for use
- Precautions
- Hypochlorous Technology, Ingredients
- Ingredients:
- Storage
-
SPL UNCLASSIFIED SECTION
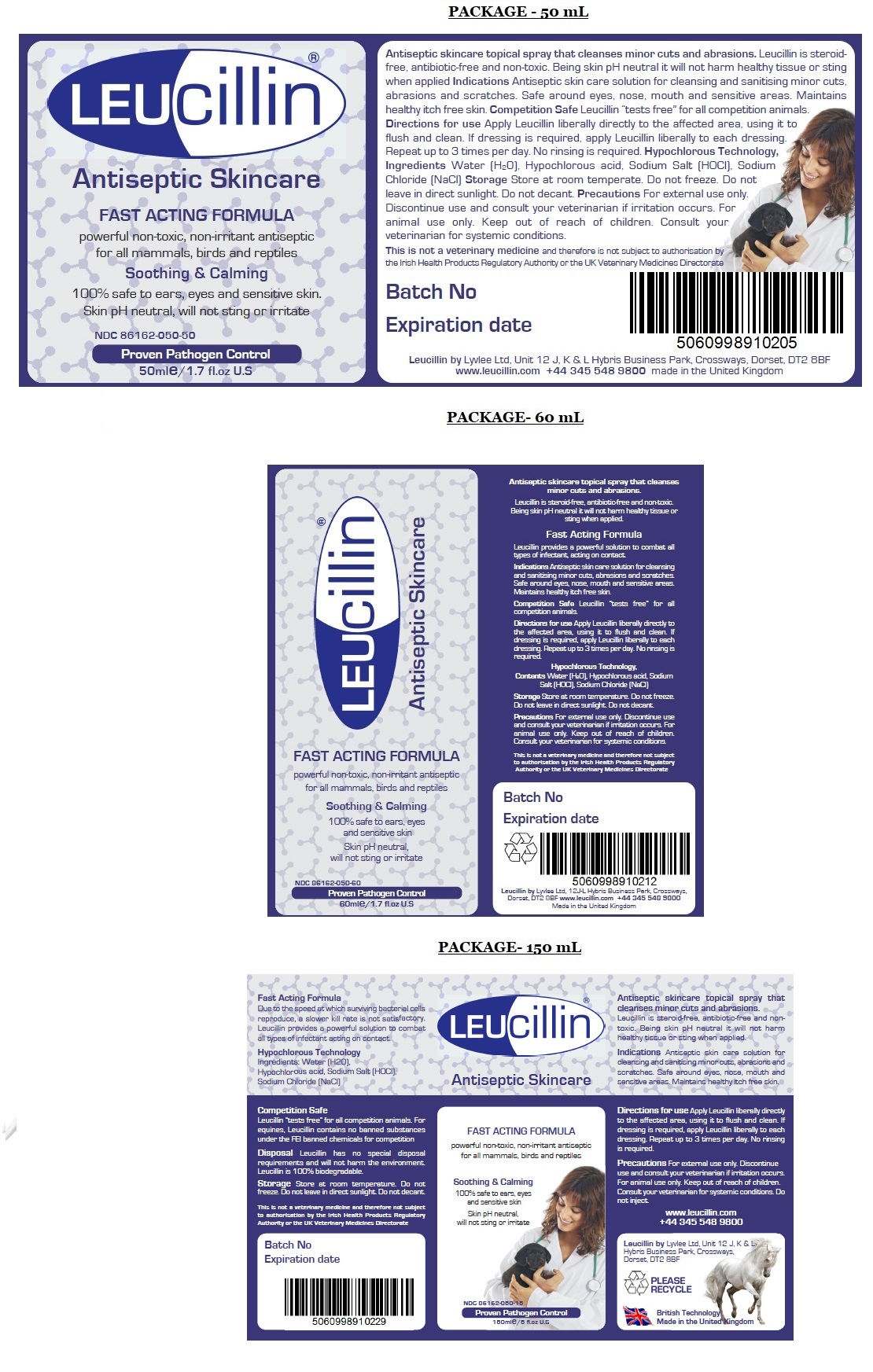
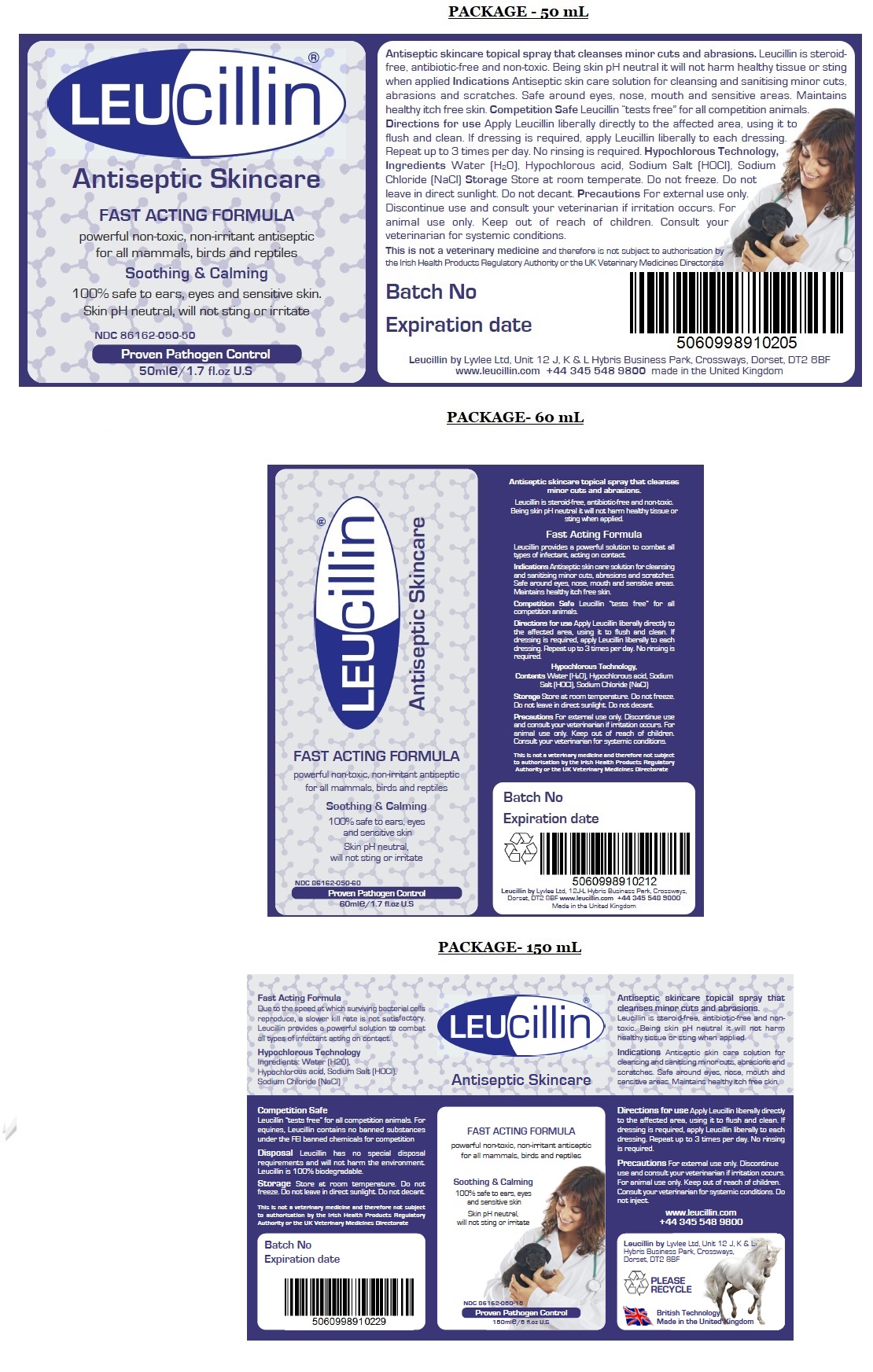
Antiseptic Skincare
FAST ACTING FORMULA
powerful non-toxic, non-irritant antiseptic for all mammals, birds and reptiles
Soothing & Calming
100% safe to ears, eyes, and sensitive skin
Skin pH neutral, will not sting or irritate
Proven Pathogen Control
Leucillin by Lyvlee Ltd, Unit 12 J, K & L Hybris Business Park, Crossways, Dorset, DT2 8BF
www.leucillin.com +44 345 548 9800info@leucillin.co.uk
British Technology
Made in the United KingdomDue to the speed at which surviving bacterial cells reproduce, a slower kill rate is not satisfactory. Leucillin provides a powerful solution to combat all types of infection acting on contact.
Competition Safe
Leucillin “tests free” for all competition animals. For equines, Leucillin contains no banned substances under the FEI banned chemicals for competition list.Disposal Leucillin has no special disposal requirements and will not harm the environment. Leucillin is 100% biodegradable.
This is not a veterinary medicine and therefore not subject to authorisation by the Irish Health Products Regulatory Authority or the UK Veterinary Medicines Directorate
- Packaging
-
INGREDIENTS AND APPEARANCE
LEUCILLIN
hypochlorous acid liquidProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:86162-050 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYPOCHLOROUS ACID (UNII: 712K4CDC10) (HYPOCHLOROUS ACID - UNII:712K4CDC10) HYPOCHLOROUS ACID 0.03 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM HYPOCHLORITE (UNII: DY38VHM5OD) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:86162-050-50 50 mL in 1 BOTTLE, DROPPER 2 NDC:86162-050-60 60 mL in 1 BOTTLE, SPRAY 3 NDC:86162-050-15 150 mL in 1 BOTTLE, SPRAY 4 NDC:86162-050-25 250 mL in 1 BOTTLE, SPRAY 5 NDC:86162-050-00 500 mL in 1 BOTTLE, SPRAY 6 NDC:86162-050-01 5000 mL in 1 CAN Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/17/2022 Labeler - LYVLEE LIMITED (216972894) Establishment Name Address ID/FEI Business Operations LYVLEE LIMITED 216972894 manufacture, api manufacture