Label: ACNE TREATMENT- benzoyl peroxide gel
- NDC Code(s): 76162-229-10
- Packager: Topco associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 23, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Use
- Warnings
- Do not use if you
-
When using this product
- skin irritation and dryness is more likely to occur if you use aother topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time
- avoid unnecessary sun exposure and use a sunscreen
- avoid contact with eyes, lips and mouth
- avoid contact with hair and dyed fabrics, which may be bleached by this product
- skin irritation may occur, characterized by redness, buring, itching, peeling or posible swelling. Irritation may be reduced by using the product less frequently or in a lower concentration
- stop use and ask a doctor if
- Keep out of reach of children
-
Directions
- clean the skin thoroughly before applying this product
- cover the entire affected area with a thin layer one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce daily application to once a day or every other day
- if going outside, apply sunscreen after using this product. If irritation or sensitivity develops stop use of both products and ask a doctor
- inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
DISTRIBUTED BY TOPCO ASSOCIATES LLC
ELK GROVE VILLAGE, IL 60007
TOPCO VIJA0422
QUESTIONS? 1-423-0139
topcare@topco.com
www.topcarebrand.com
Scan here for more information or call 1-888-423-0139
This micronized benzoyl peroxide formula goes to work releasing the medicine deep into the pore where pimples begin
Free from parabens, phthalates, fragrances, alcohol
Quality Guaranteed
This TopCare product is laboratory tested to guarantee its highest quality.
Your total satisfaction is guaranteed.
This product is not manufactured or distributed by Johnson & Johnson Consumer Products Company
distributer of Clean & Clear Persa-Gel 10.
-
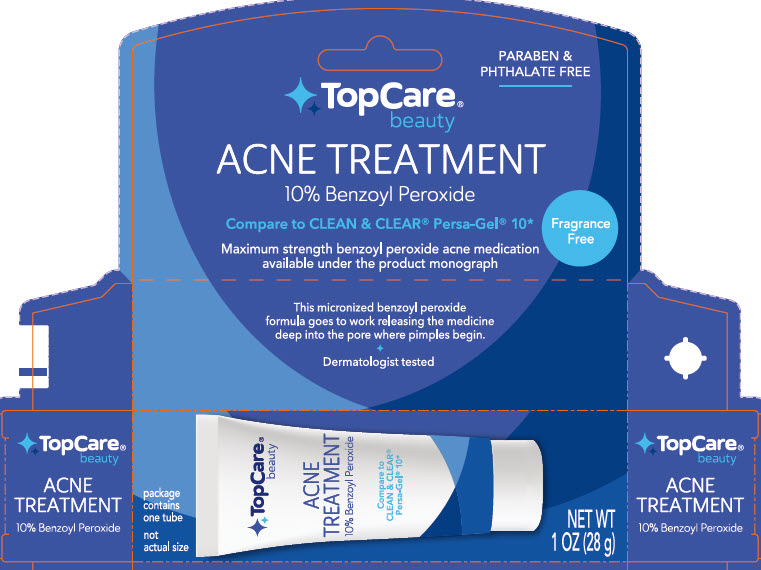
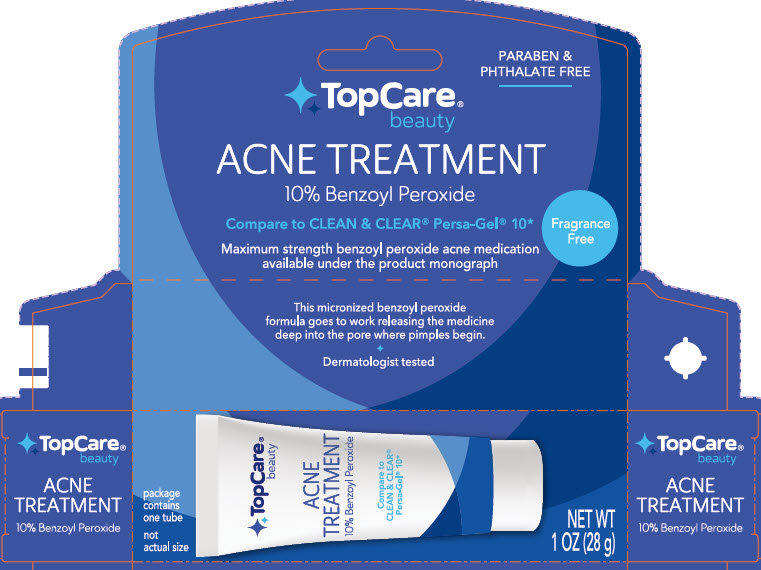
principal display panel
PARABEN & PHTHALATE FREE
TopCare Beauty
ACNE TREATMENT
10% Benzoyl Peroxide
Compare to CLEAN & CLEAR Persa-Gel 10*
This micronized benzoyl peroxide formula goes to work releasing the medicine deep into the pore where pimples begin.
Dermatologist Tested
TopCare Beauty
ACNE TREATMENT
10% Benzoyl Peroxide
Package contains one tube
not actual size
NET WT 1 OZ (28 g)
TopCare Beauty
ACNE TREATMENT
10% Benzoyl Peroxide
-
INGREDIENTS AND APPEARANCE
ACNE TREATMENT
benzoyl peroxide gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76162-229 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZOYL PEROXIDE (UNII: W9WZN9A0GM) (BENZOYL PEROXIDE - UNII:W9WZN9A0GM) BENZOYL PEROXIDE 1 mg in 1 g Inactive Ingredients Ingredient Name Strength CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) EDETATE DISODIUM (UNII: 7FLD91C86K) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LAURETH-4 (UNII: 6HQ855798J) SODIUM HYDROXIDE (UNII: 55X04QC32I) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76162-229-10 1 in 1 PACKAGE 08/23/2022 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 08/23/2022 Labeler - Topco associates LLC (006935977) Registrant - Vi-Jon, LLC (790752542) Establishment Name Address ID/FEI Business Operations Vi-Jon, LLC 790752542 manufacture(76162-229) Establishment Name Address ID/FEI Business Operations Vi-Jon, LLC 088520668 manufacture(76162-229)