Label: ALCOHOL- isopropyl alcohol swab
- NDC Code(s): 50332-0046-0, 50332-0046-5, 50332-0046-7
- Packager: HART Health
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 18, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
- Warnings
- Do not use
- Stop use and ask a doctor if
- Keep out of reach of children
- Directions
- Inactive Ingredient
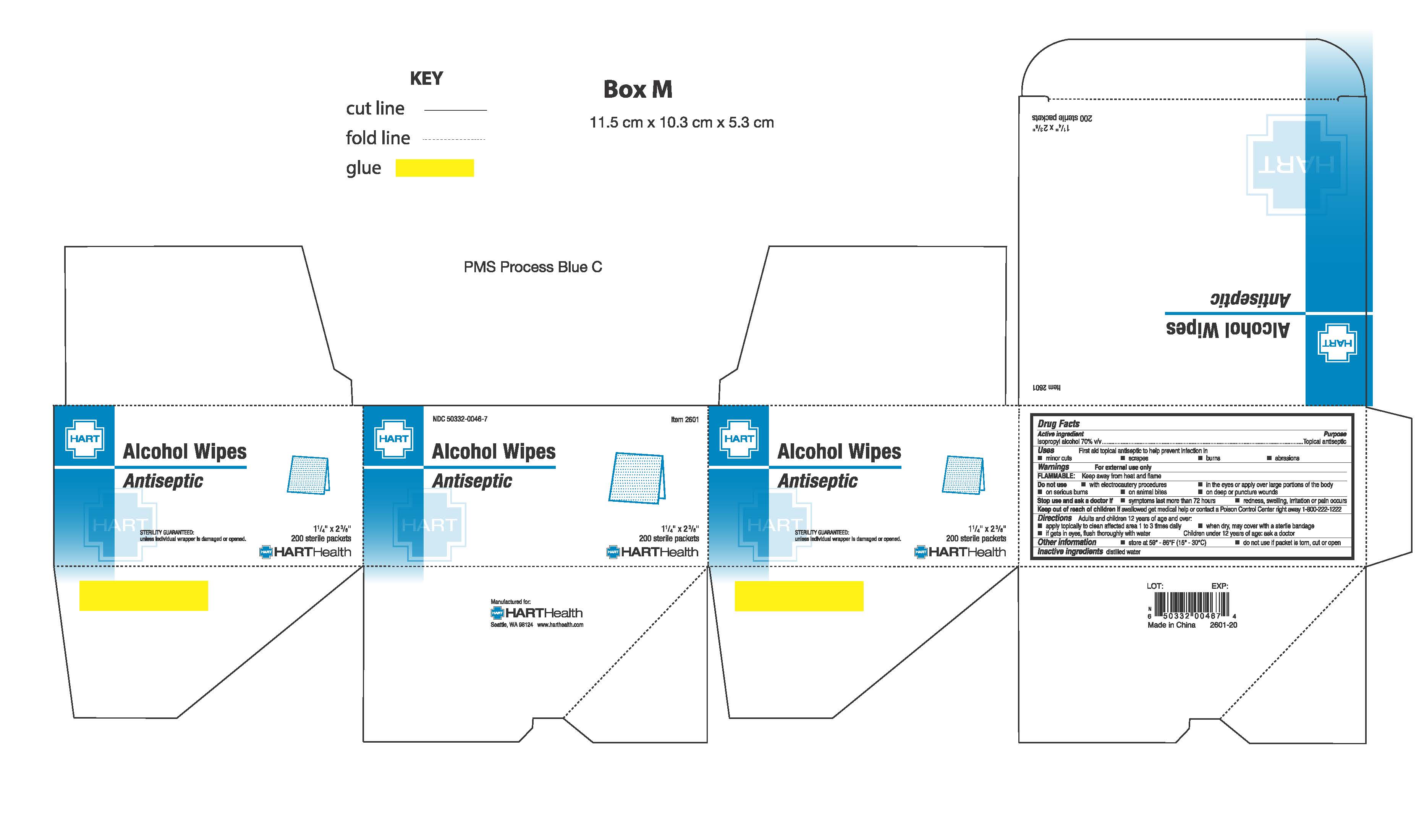
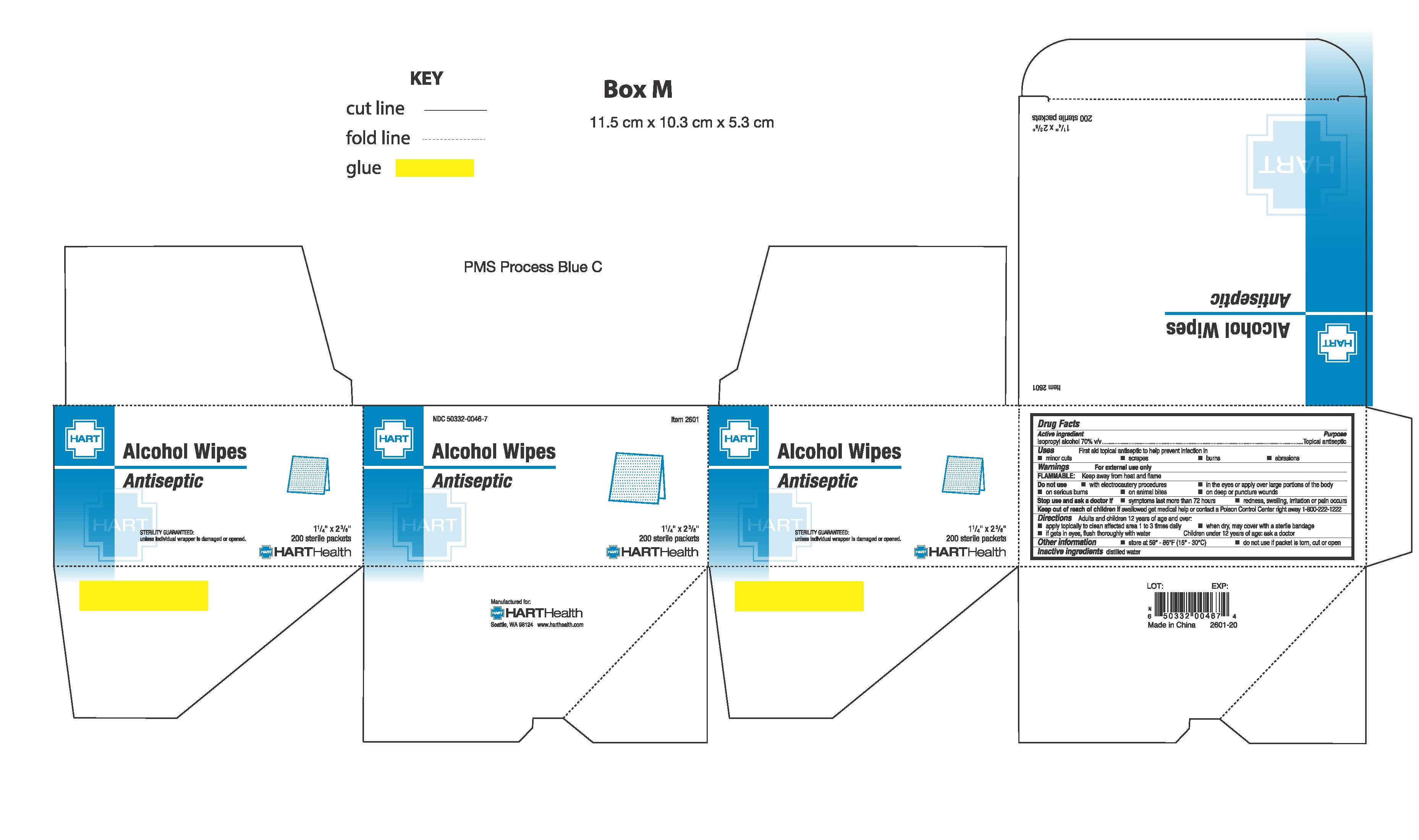
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ALCOHOL
isopropyl alcohol swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50332-0046 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL .70 g in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50332-0046-0 10 in 1 BOX 04/01/1974 1 .35 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:50332-0046-5 50 in 1 BOX 04/01/1974 2 .35 g in 1 PACKET; Type 0: Not a Combination Product 3 NDC:50332-0046-7 200 in 1 BOX 04/20/2016 3 .35 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M 04/01/1974 Labeler - HART Health (069560969)