Label: ACETAMINOPHEN tablet
-
NDC Code(s):
69842-298-05,
69842-298-10,
69842-298-15,
69842-298-21, view more69842-298-24, 69842-298-30, 69842-298-40, 69842-298-60
- Packager: CVS PHARMACY, INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated August 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Liver warning
- Allergy alert
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of the reach of children.
- Directions
- Other information
-
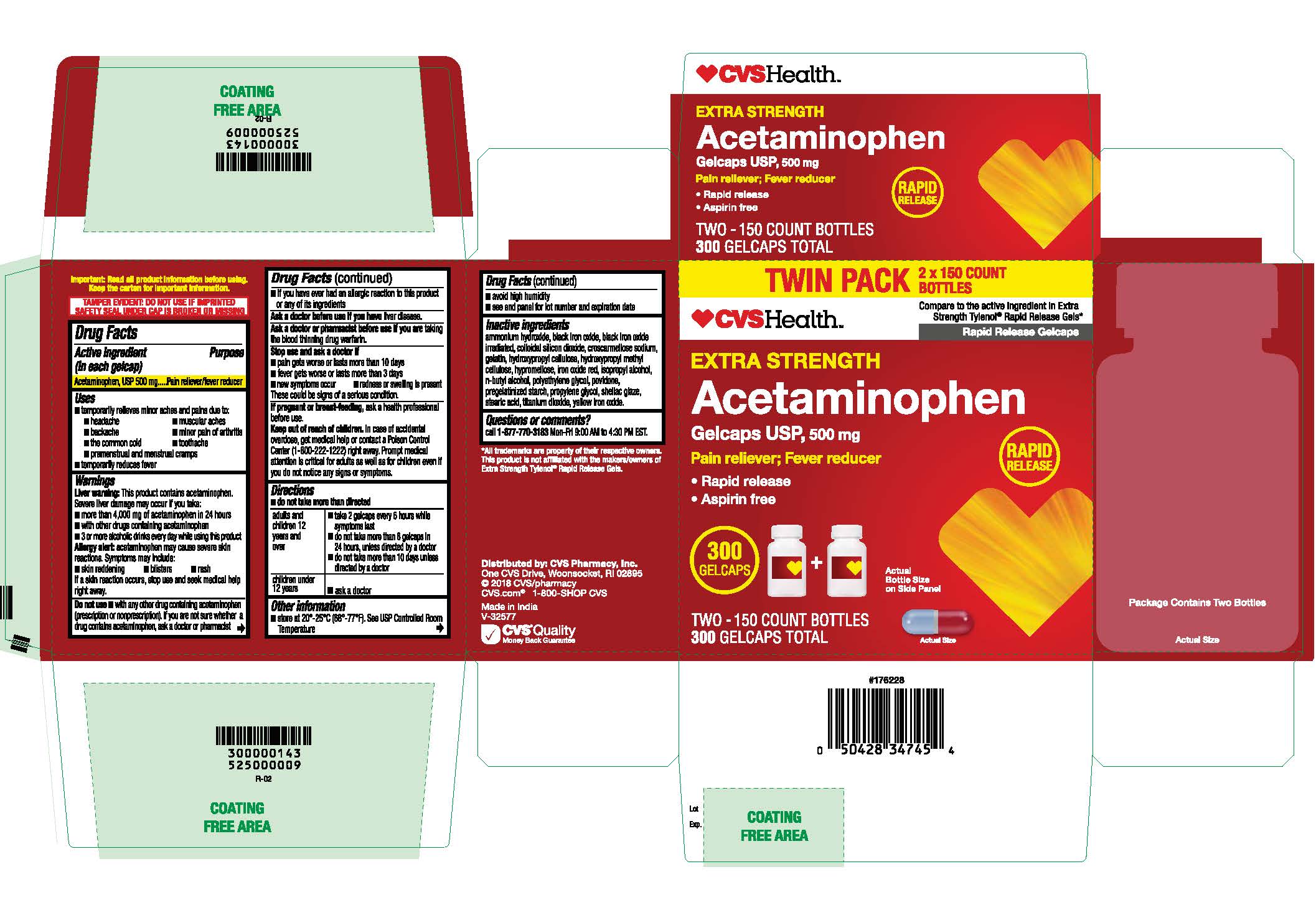
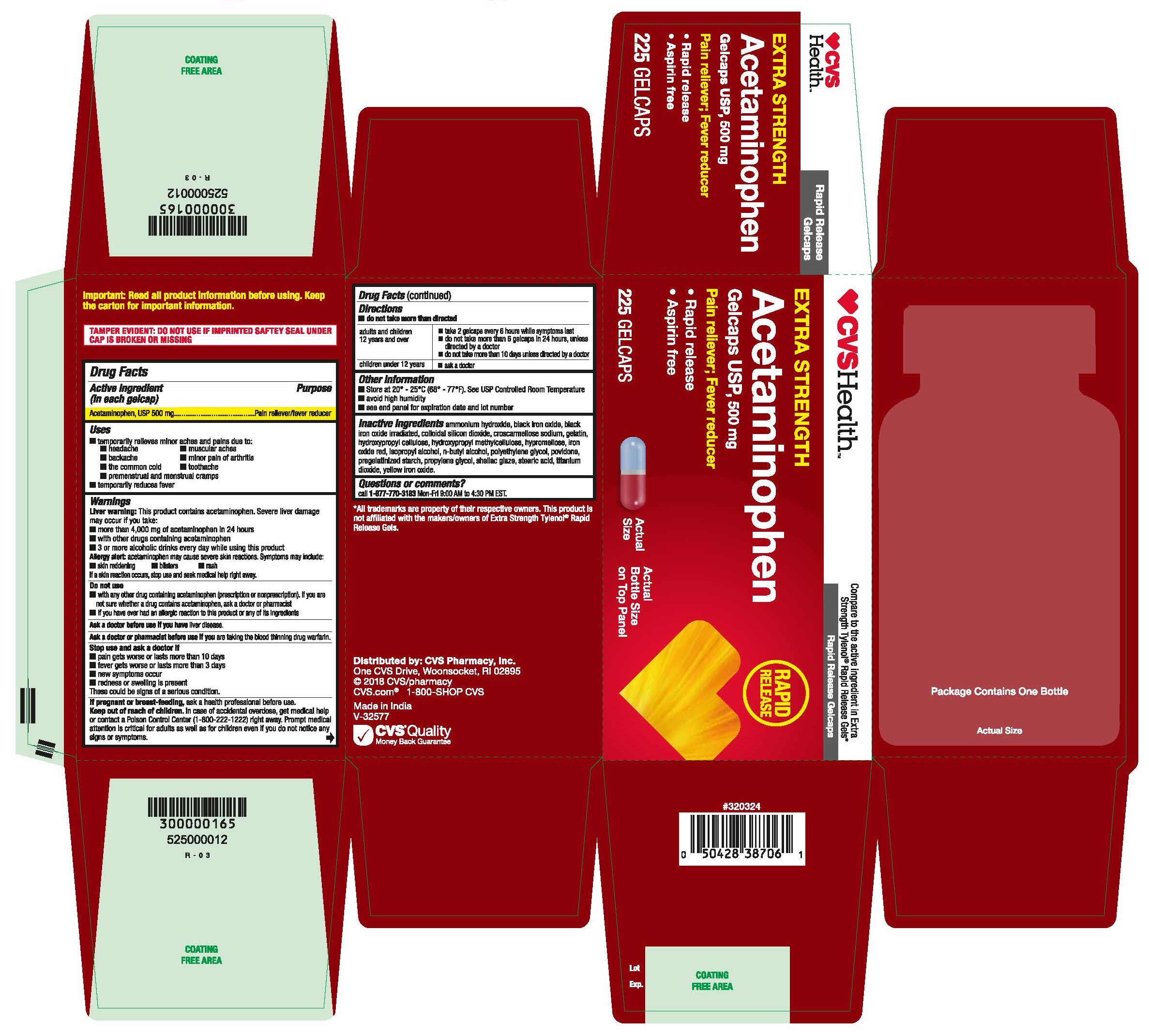
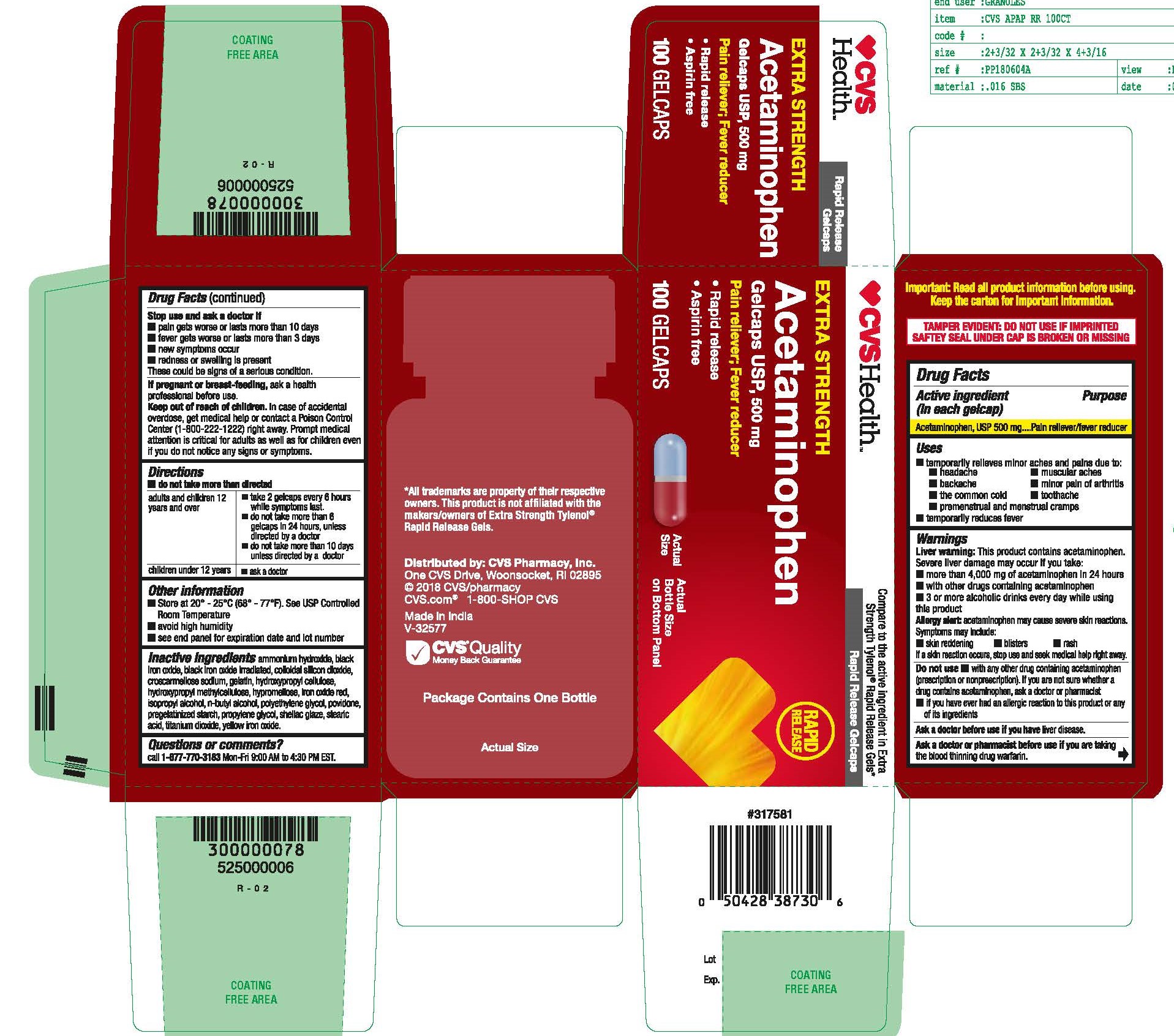
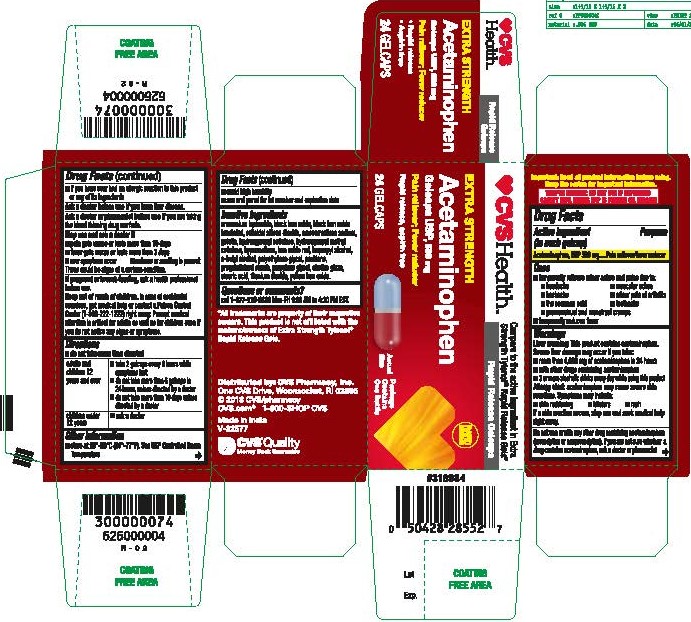
Inactive ingredients
ammonium hydroxide, black iron oxide, black iron oxide irradiated, colloidal silicon dioxide, croscarmellose sodium, gelatin, hydroxypropyl cellulose, hydroxypropyl methyl cellulose, hypromellose, iron oxide red, isopropyl alcohol, n-butyl alcohol, polyethylene glycol, povidone, pregelatinized starch, propylene glycol, shellac glaze, stearic acid, titanium dioxide, yellow iron oxide.
- Questions or comments?
- Acetaminophen, USP 500 mg Rapid Release Gelcaps
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN
acetaminophen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-298 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) FERRIC OXIDE RED (UNII: 1K09F3G675) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) ISOPROPYL ALCOHOL (UNII: ND2M416302) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) FERROSOFERRIC OXIDE (UNII: XM0M87F357) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYPROMELLOSES (UNII: 3NXW29V3WO) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) POVIDONE (UNII: FZ989GH94E) STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) AMMONIA (UNII: 5138Q19F1X) Product Characteristics Color gray (Encapsulated with red opaque and blue gray opaque hard gelatin shells) Score no score Shape OVAL Size 19mm Flavor Imprint Code G1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-298-24 24 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 2 NDC:69842-298-05 50 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 3 NDC:69842-298-10 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 4 NDC:69842-298-21 225 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 5 NDC:69842-298-30 300 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 6 NDC:69842-298-40 400 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 7 NDC:69842-298-60 600 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 8 NDC:69842-298-15 150 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 04/01/2019 Labeler - CVS PHARMACY, INC (062312574)