Label: FEXOFENADINE HYDROCHLORIDE tablet
-
NDC Code(s):
51316-800-08,
51316-800-15,
51316-800-30,
51316-800-45, view more51316-800-70, 51316-800-90
- Packager: CVS PHARMACY, INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 24, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have kidney disease. Your doctor should determine if you need a different dose.
When using this product
- do not take more than directed
- do not take at the same time as aluminum or magnesium antacids
- do not take with fruit juices (see Directions)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
-
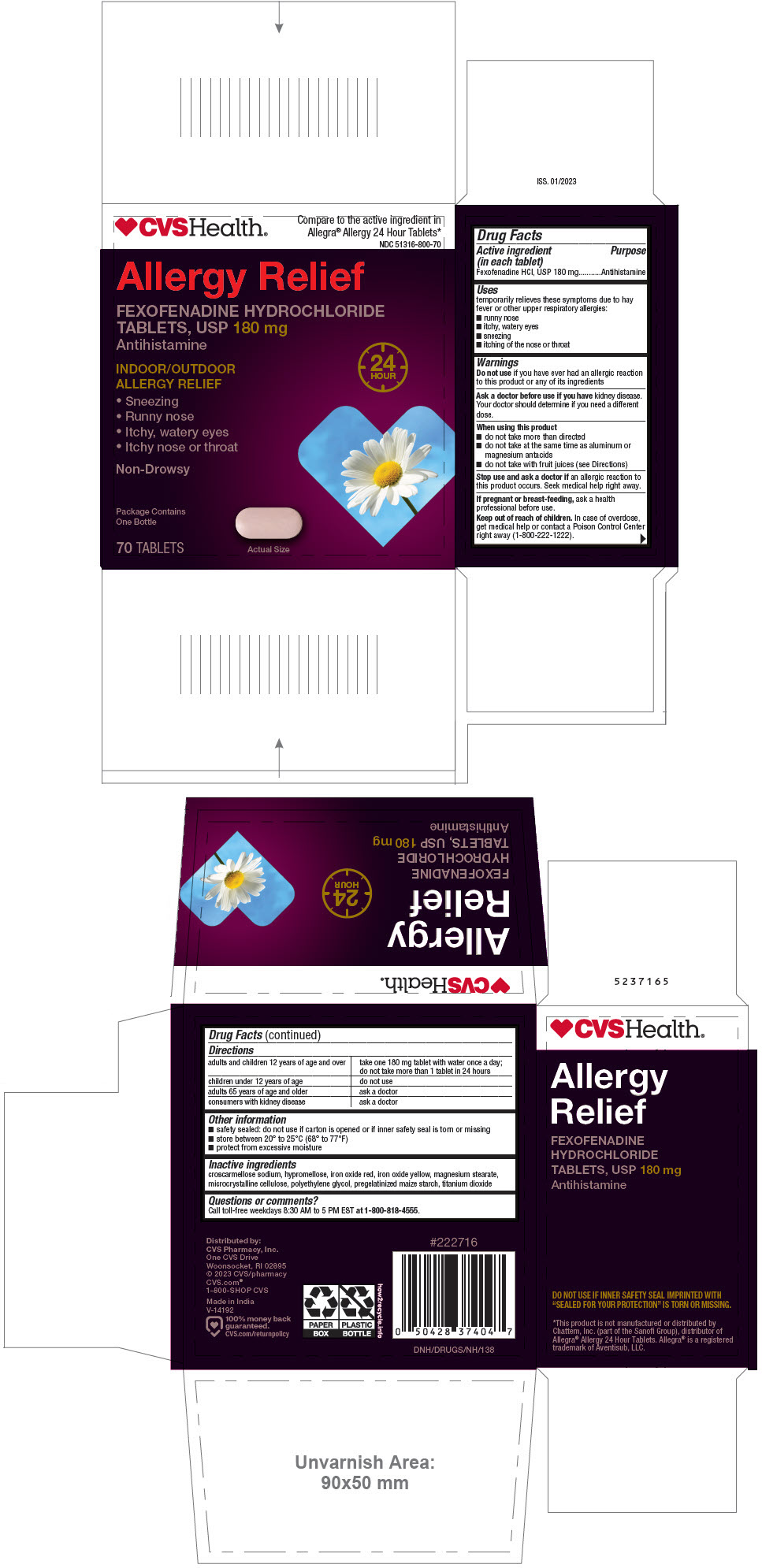
PRINCIPAL DISPLAY PANEL - 180 mg Tablet Bottle Carton
CVS Health®
Compare to the active ingredient
in Allegra® Allergy*NDC 51316-800-70
Non-Drowsy
Allergy Relief
FEXOFENADINE HYDROCHLORIDE
TABLETS, USP 180 mg
Antihistamine24
HOURINDOOR/OUTDOOR
ALLERGY RELIEF- Sneezing
- Runny nose
- Itchy, watery eyes
- Itchy nose or throat
Package Contains
One Bottle70 TABLETS
Actual Size
-
INGREDIENTS AND APPEARANCE
FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51316-800 Route of Administration Oral Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) Product Characteristics Color PINK Score no score Shape OVAL Size 17mm Flavor Imprint Code 545 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51316-800-90 1 in 1 CARTON 09/20/2022 1 90 in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:51316-800-70 1 in 1 CARTON 09/20/2022 2 70 in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:51316-800-45 1 in 1 CARTON 09/20/2022 3 45 in 1 BOTTLE; Type 0: Not a Combination Product 4 NDC:51316-800-30 1 in 1 CARTON 09/20/2022 4 30 in 1 BOTTLE; Type 0: Not a Combination Product 5 NDC:51316-800-15 1 in 1 CARTON 09/20/2022 5 15 in 1 BLISTER PACK; Type 0: Not a Combination Product 6 NDC:51316-800-08 1 in 1 CARTON 09/20/2022 6 180 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091567 09/20/2022 Labeler - CVS PHARMACY, INC (062312574) Establishment Name Address ID/FEI Business Operations Sun Pharmaceutical Industries Limited 650445203 MANUFACTURE(51316-800)

