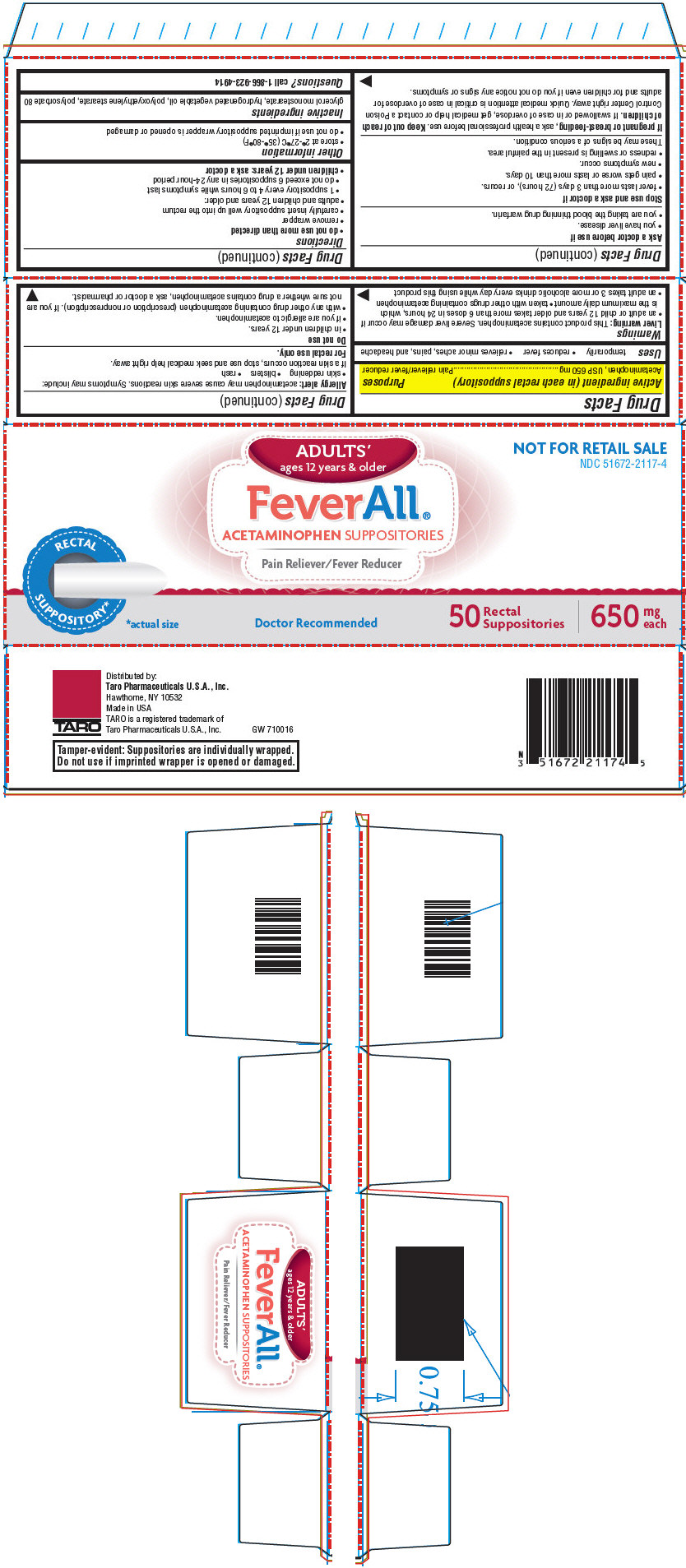
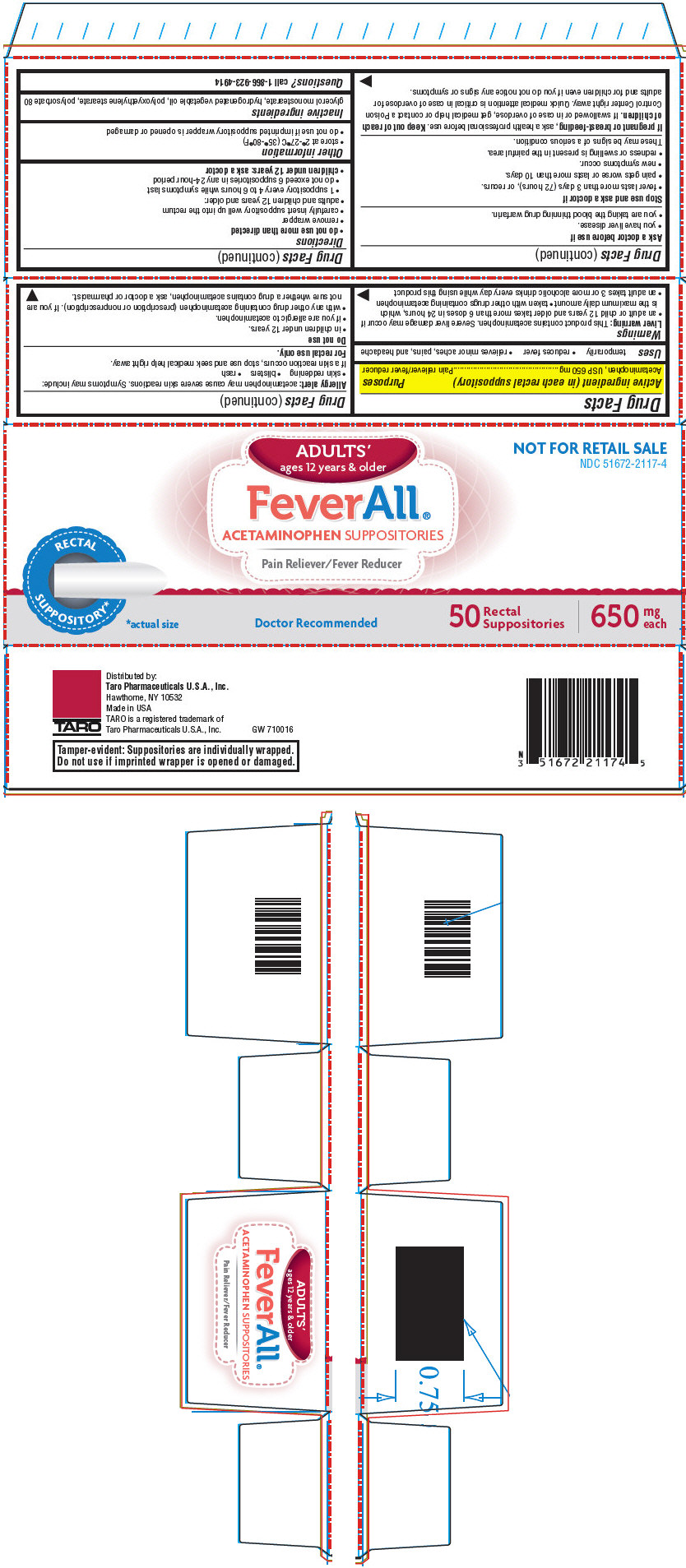
Label: FEVERALL ADULTS- acetaminophen suppository
- NDC Code(s): 51672-2117-0, 51672-2117-4
- Packager: Taro Pharmaceuticals U.S.A. Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated May 19, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each rectal suppository)
- Purposes
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if
- an adult or child 12 years and older takes more than 6 doses in 24 hours, which is the maximum daily amount
- taken with other drugs containing acetaminophen
- an adult takes 3 or more alcoholic drinks every day while using this product
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
Do not use
- in children under 12 years.
- if you are allergic to acetaminophen.
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- Directions
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 50 Suppository Carton
-
INGREDIENTS AND APPEARANCE
FEVERALL ADULTS
acetaminophen suppositoryProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51672-2117 Route of Administration RECTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Acetaminophen (UNII: 362O9ITL9D) (Acetaminophen - UNII:362O9ITL9D) Acetaminophen 650 mg Inactive Ingredients Ingredient Name Strength glyceryl monostearate (UNII: 230OU9XXE4) hydrogenated palm kernel oil (UNII: FM8D1RE2VP) PEG-100 stearate (UNII: YD01N1999R) polysorbate 80 (UNII: 6OZP39ZG8H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51672-2117-4 50 in 1 CARTON 12/12/2013 1 NDC:51672-2117-0 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA018337 12/12/2013 Labeler - Taro Pharmaceuticals U.S.A. Inc. (145186370) Establishment Name Address ID/FEI Business Operations G&W NC Laboratories, Inc. 079419931 MANUFACTURE(51672-2117)