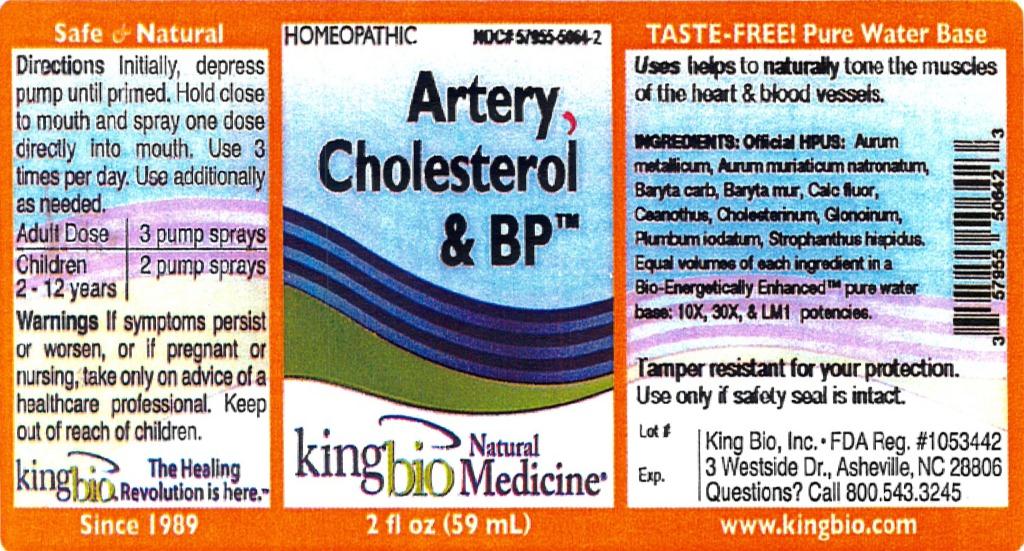
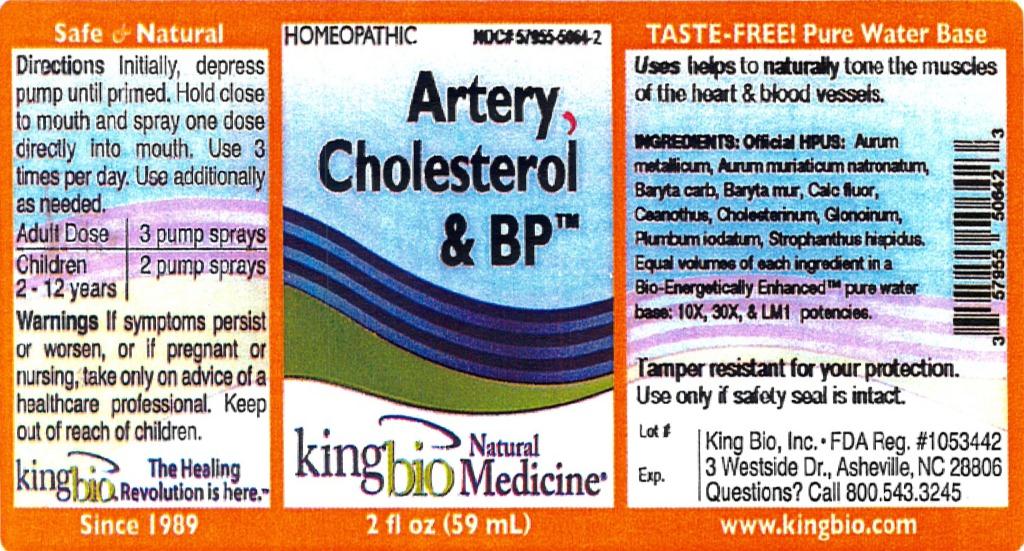
Label: ARTERY CHOLESTEROL AND BP- aurum metallicum, aurum muriaticum natronatum, baryta carbonica, baryta muriatica, calcarea flourica, ceanothus americanus, cholesterinum, glonoinum, plumbum iodatum, strophanthus liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 57955-5064-2 - Packager: King Bio Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 19, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Inactive Ingredient
- Purposes
- Directions
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Uses
- Package Label
-
INGREDIENTS AND APPEARANCE
ARTERY CHOLESTEROL AND BP
aurum metallicum, aurum muriaticum natronatum, baryta carbonica, baryta muriatica, calcarea flourica, ceanothus americanus, cholesterinum, glonoinum, plumbum iodatum, strophanthus liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57955-5064 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GOLD (UNII: 79Y1949PYO) (GOLD - UNII:79Y1949PYO) GOLD 10 [hp_X] in 59 mL SODIUM TETRACHLOROAURATE (UNII: 7FT6QUT299) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM TETRACHLOROAURATE 10 [hp_X] in 59 mL BARIUM CARBONATE (UNII: 6P669D8HQ8) (BARIUM CATION - UNII:V645272HLN) BARIUM CARBONATE 10 [hp_X] in 59 mL BARIUM CHLORIDE DIHYDRATE (UNII: EL5GJ3U77E) (BARIUM CATION - UNII:V645272HLN) BARIUM CHLORIDE DIHYDRATE 10 [hp_X] in 59 mL CALCIUM FLUORIDE (UNII: O3B55K4YKI) (CALCIUM FLUORIDE - UNII:O3B55K4YKI) CALCIUM FLUORIDE 10 [hp_X] in 59 mL CEANOTHUS AMERICANUS LEAF (UNII: 25B1Y14T8N) (CEANOTHUS AMERICANUS LEAF - UNII:25B1Y14T8N) CEANOTHUS AMERICANUS LEAF 10 [hp_X] in 59 mL CHOLESTEROL (UNII: 97C5T2UQ7J) (CHOLESTEROL - UNII:97C5T2UQ7J) CHOLESTEROL 10 [hp_X] in 59 mL NITROGLYCERIN (UNII: G59M7S0WS3) (NITROGLYCERIN - UNII:G59M7S0WS3) NITROGLYCERIN 10 [hp_X] in 59 mL LEAD IODIDE (UNII: OTL90F2GLT) (LEAD IODIDE - UNII:OTL90F2GLT) LEAD IODIDE 10 [hp_X] in 59 mL STROPHANTHUS HISPIDUS SEED (UNII: MO892VI77K) (STROPHANTHUS HISPIDUS SEED - UNII:MO892VI77K) STROPHANTHUS HISPIDUS SEED 10 [hp_X] in 59 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57955-5064-2 59 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 03/19/2012 Labeler - King Bio Inc. (617901350) Registrant - King Bio Inc. (617901350) Establishment Name Address ID/FEI Business Operations King Bio Inc. 617901350 manufacture