Label: SLEEP AID- doxylamine succinate tablet
- NDC Code(s): 49035-968-27
- Packager: Wal-Mart Stores Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
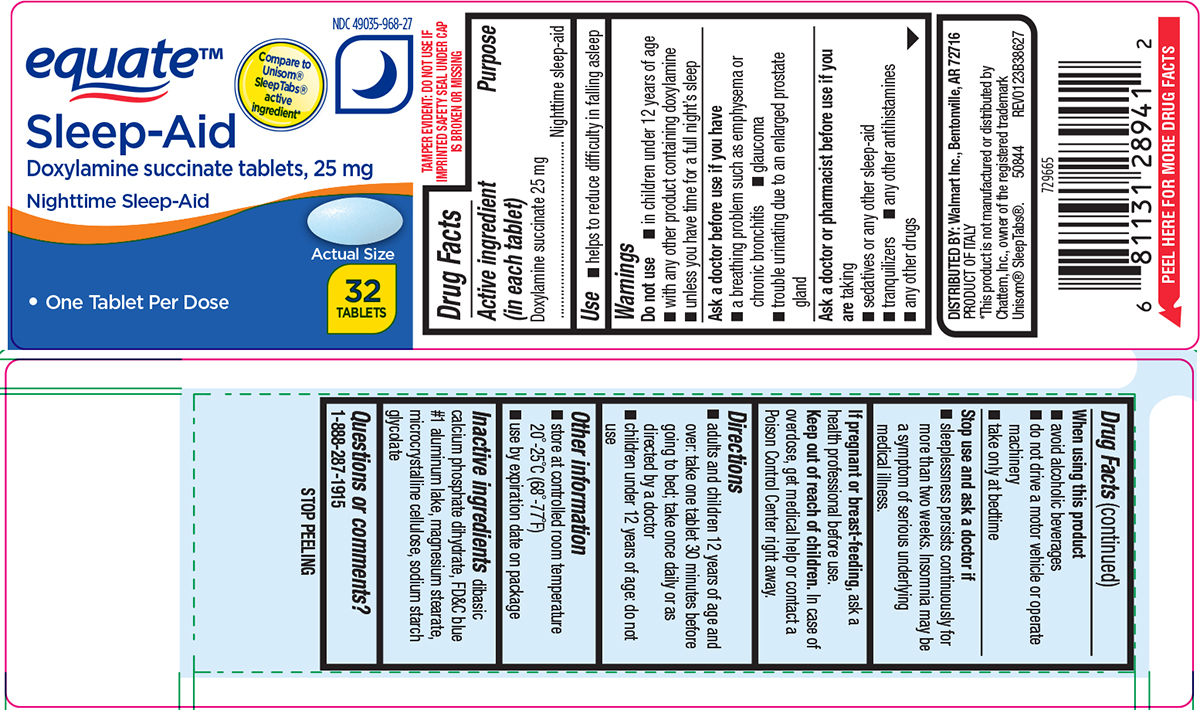
- Active ingredient (in each tablet)
- Purpose
- Use
-
Warnings
Do not use
- in children under 12 years of age
- with any other product containing doxylamine
- unless you have time for a full night’s sleep
Ask a doctor before use if you have
- glaucoma
- a breathing problem such as asthma, emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
Ask a doctor or pharmacist before use if you are
taking
- sedatives or any other sleep-aid
- tranquilizers
- any other antihistamines
- any other drugs
When using this product
- avoid alcoholic beverages
- do not drive a motor vehicle or operate machinery
- take only at bedtime
- in children under 12 years of age
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal display panel
equate™
NDC 49035-968-27
Compare to
Unisom®
SleepTabs®
active
ingredient*Sleep-Aid
Doxylamine succinate 25 mg
Nighttime Sleep-Aid• One Tablet Per Dose
Actual Size
32
TABLETSTAMPER EVIDENT: DO NOT USE IF
IMPRINTED SAFETY SEAL UNDER CAP
IS BROKEN OR MISSINGDISTRIBUTED BY: Walmart Inc., Bentonville, AR 72716
PRODUCT OF ITALY
*This product is not manufactured or distributed by
Chattem, Inc., owner of the registered trademark
Unisom® SleepTabs®. 50844 REV0123B38627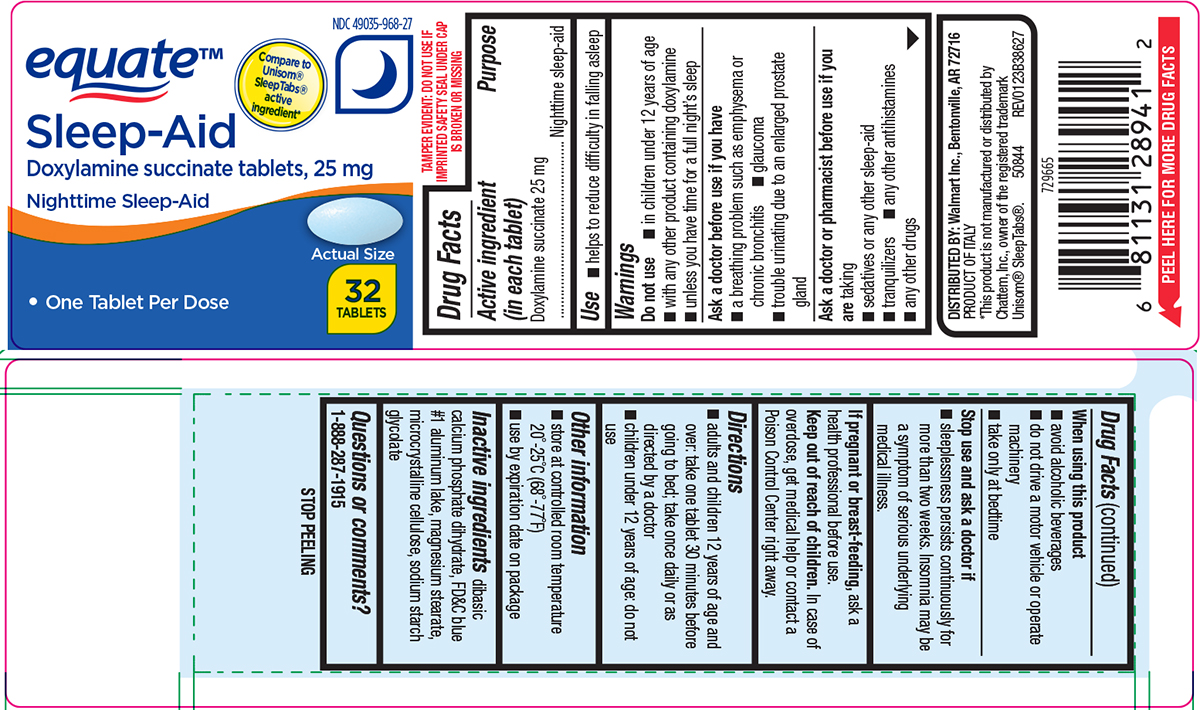
Equate 44-386
-
INGREDIENTS AND APPEARANCE
SLEEP AID
doxylamine succinate tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49035-968 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 25 mg Inactive Ingredients Ingredient Name Strength DIBASIC CALCIUM PHOSPHATE DIHYDRATE (UNII: O7TSZ97GEP) FD&C BLUE NO. 1 ALUMINUM LAKE (UNII: J9EQA3S2JM) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color blue Score no score Shape OVAL Size 10mm Flavor Imprint Code 44;386 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49035-968-27 32 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 07/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040564 07/15/2019 Labeler - Wal-Mart Stores Inc (051957769) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(49035-968) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(49035-968) , pack(49035-968) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(49035-968) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(49035-968)