Label: VETRASEB CK FLUSH- chlorhexidine gluconate, ketoconazole, phytosphingosine salicloyl solution
- NDC Code(s): 13985-736-04, 13985-736-16
- Packager: MWI/VetOne
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 1, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
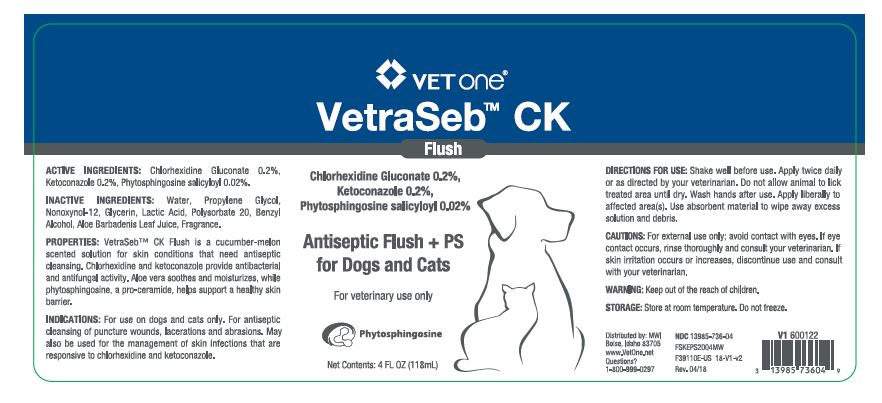
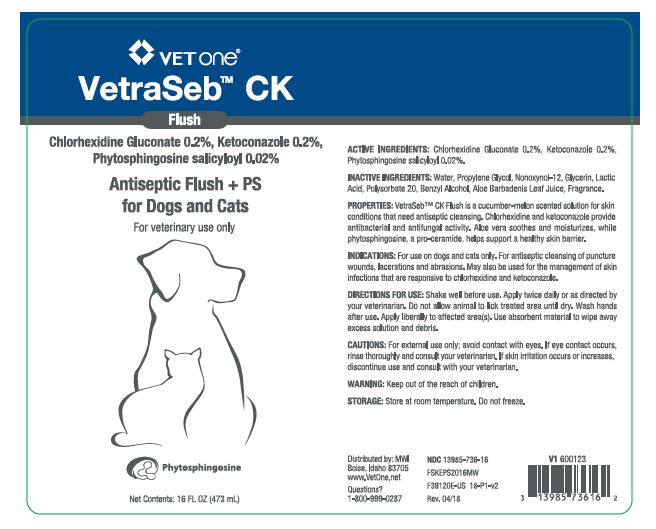
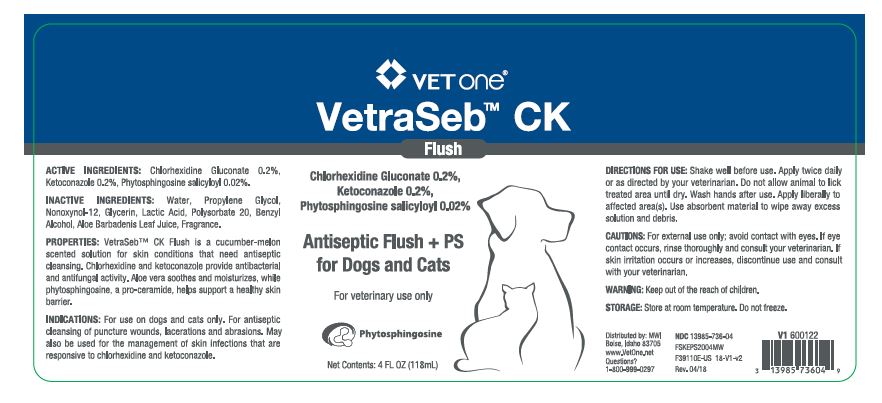
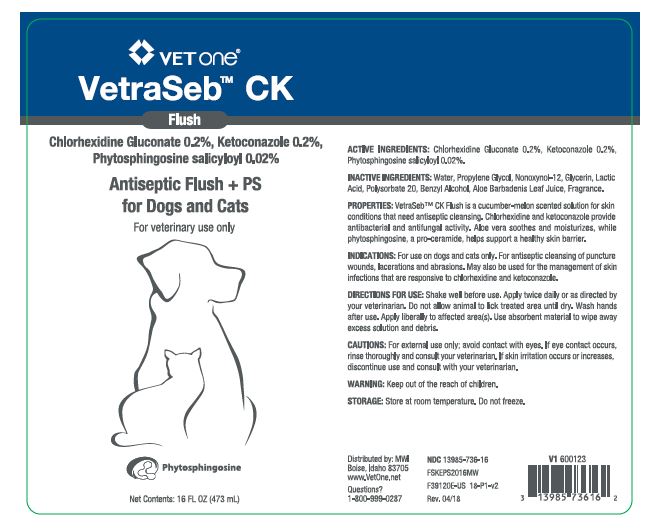
VetraSeb™ CK Flush
Antiseptic Flush + PS for Dogs and Cats
ACTIVE INGREDIENTS: Chlorhexidine Gluconate 0.2%, Ketoconazole 0.2%, Phytosphingosine salicyloyl 0.02%.
INACTIVE INGREDIENTS: Water, Propylene Glycol, Nonoxynol-12, Glycerin, Lactic Acid, Polysorbate 20, Benzyl Alcohol, Aloe Barbadenis Leaf Juice, Fragrance.
PROPERTIES: VetraSeb™ CK Flush is a cucumber-melon scented solution for skin conditions that need antiseptic cleansing. Chlorhexidine and ketoconazole provide antibacterial and antifungal activity. Aloe vera soothes and moisturizes, while phytosphingosine, a pro-ceramide helps support a healthy skin barrier.
-
INDICATIONS & USAGE
INDICATIONS: For use on dogs and cats only. For antiseptic cleansing of puncture wounds, lacerations and abrasions. May also be used for the management of skin infections that are responsive to chlorhexidine and ketoconazole.
DIRECTIONS FOR USE: Shake well before use. Apply twice daily or as directed by your veterinarian. Do not allow animal to lick treated area until dry. Wash hands after use. Apply liberally to affected area(s). Use absorbent material to wipe away excess solution and debris.
- WARNINGS AND PRECAUTIONS
- STORAGE AND HANDLING
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 4 OZ (118 mL) BOTTLE
- PRINCIPAL DISPLAY PANEL - 16 OZ (473 mL) BOTTLE
-
INGREDIENTS AND APPEARANCE
VETRASEB CK FLUSH
chlorhexidine gluconate, ketoconazole, phytosphingosine salicloyl solutionProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:13985-736 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) (CHLORHEXIDINE - UNII:R4KO0DY52L) CHLORHEXIDINE GLUCONATE 2 mg in 1 mL KETOCONAZOLE (UNII: R9400W927I) (KETOCONAZOLE - UNII:R9400W927I) KETOCONAZOLE 2 mg in 1 mL SALICYLOYL PHYTOSPHINGOSINE (UNII: U891J4JCNH) (SALICYLOYL PHYTOSPHINGOSINE - UNII:U891J4JCNH) SALICYLOYL PHYTOSPHINGOSINE 0.2 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) NONOXYNOL-12 (UNII: 6NR43D77O6) GLYCERIN (UNII: PDC6A3C0OX) POLYSORBATE 20 (UNII: 7T1F30V5YH) LACTIC ACID (UNII: 33X04XA5AT) BENZYL ALCOHOL (UNII: LKG8494WBH) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13985-736-04 118 mL in 1 BOTTLE, PLASTIC 2 NDC:13985-736-16 473 mL in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 06/01/2018 Labeler - MWI/VetOne (019926120) Registrant - Ceva Sante Animale (261126049)