Label: TOPCARE TRIPLE ANTIBIOTIC- bacitracin zinc, neomycin, polymyxin b ointment
- NDC Code(s): 36800-416-26, 36800-416-58, 36800-416-64
- Packager: Topco Associates LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 2, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients (in each gram)
- Purpose
- Use
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
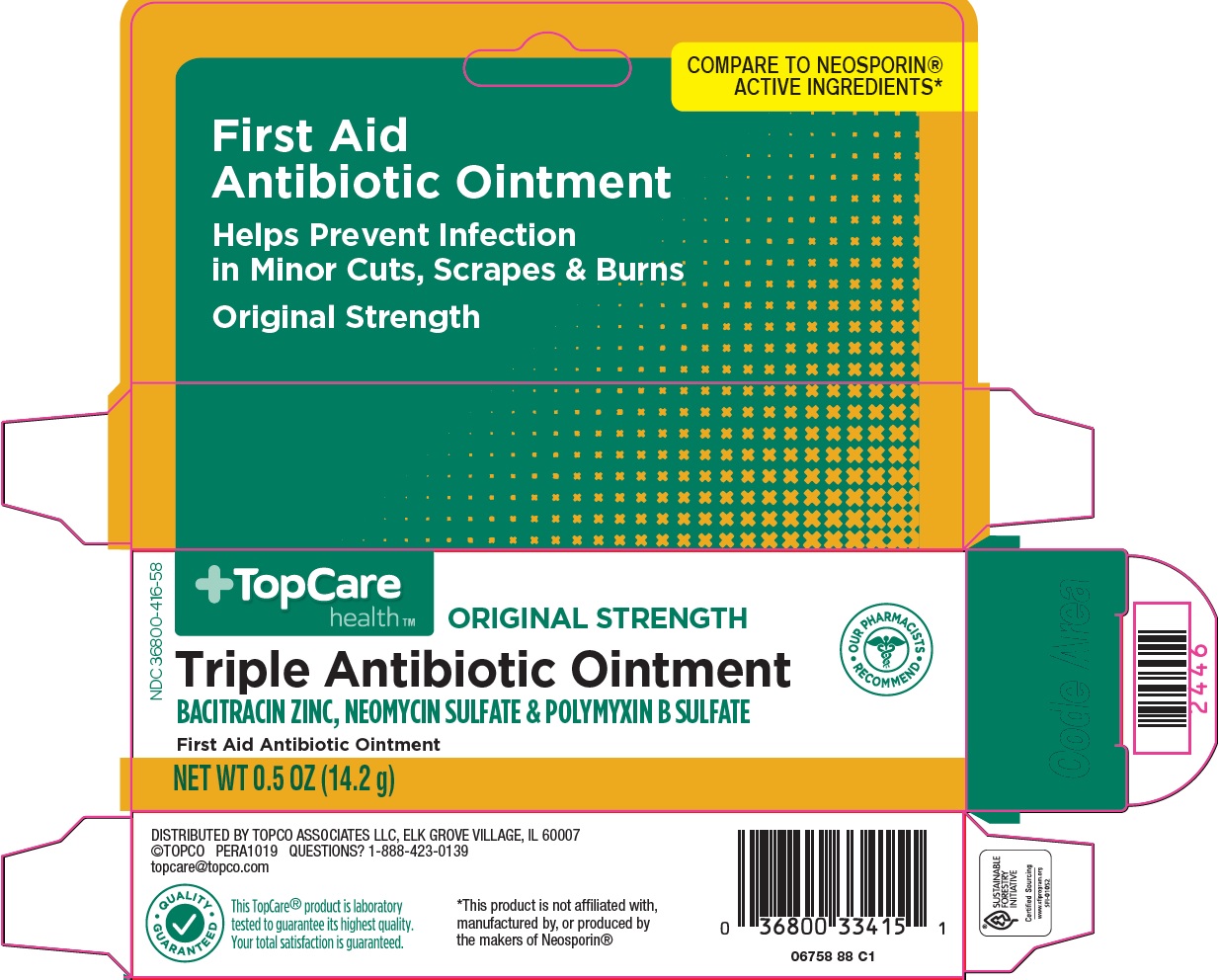
Package/Label Principal Display Panel
COMPARE TO NEOSPORIN® ACTIVE INGREDIENTS
First Aid Antibiotic Ointment
Helps Prevent Infection in Minor Cuts, Scrapes & Burns
Original Strength
ORIGINAL STRENGTH
OUR PHARMACISTS RECOMMEND
Triple Antibiotic Ointment
BACITRACIN ZINC, NEOMYCIN SULFATE & POLYMYXIN B SULFATE
First Aid Antibiotic Ointment
NET WT 0.5 OZ (14.2 g)
-
INGREDIENTS AND APPEARANCE
TOPCARE TRIPLE ANTIBIOTIC
bacitracin zinc, neomycin, polymyxin b ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-416 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [USP'U] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [USP'U] in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) COCOA BUTTER (UNII: 512OYT1CRR) COTTONSEED OIL (UNII: H3E878020N) OLIVE OIL (UNII: 6UYK2W1W1E) SODIUM PYRUVATE (UNII: POD38AIF08) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Product Characteristics Color WHITE (Translucent) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-416-58 1 in 1 CARTON 02/26/2020 1 14.2 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:36800-416-64 1 in 1 CARTON 02/26/2020 2 28 g in 1 TUBE; Type 0: Not a Combination Product 3 NDC:36800-416-26 2 in 1 CARTON 02/26/2020 3 56 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 02/26/2020 Labeler - Topco Associates LLC (006935977)