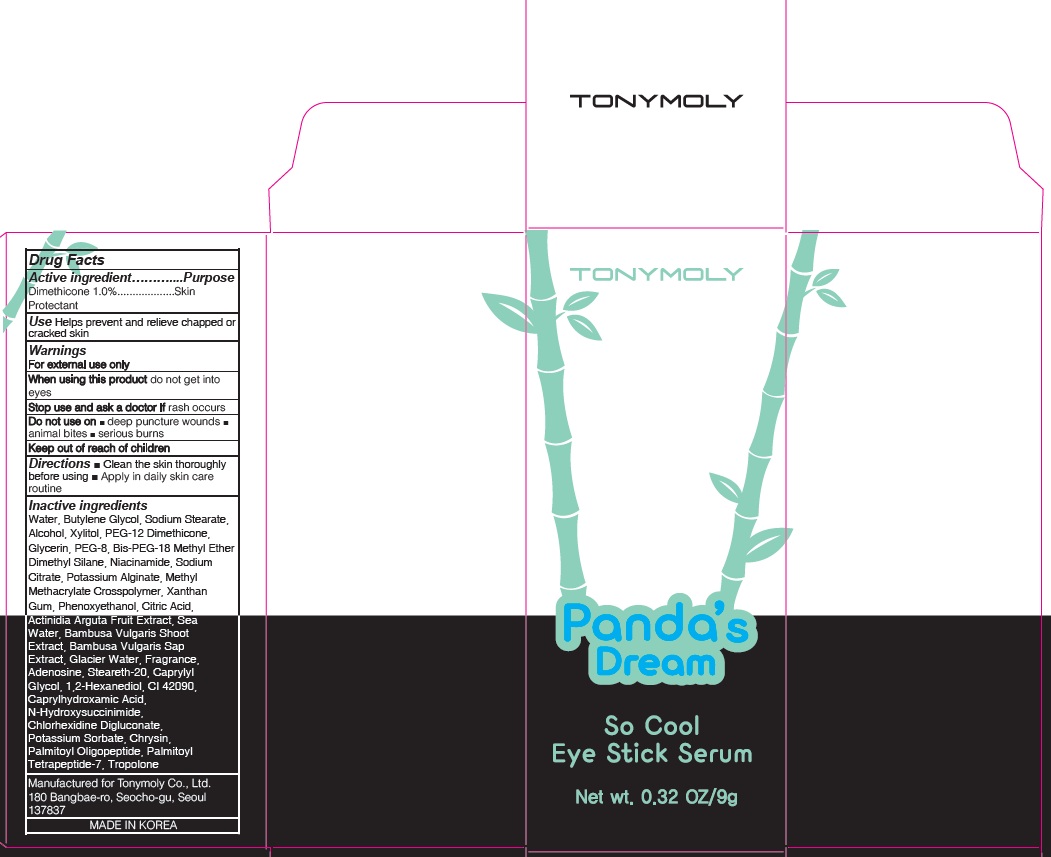
Label: PANDAS DREAM SO COOL EYE SERUM- dimethicone stick
-
Contains inactivated NDC Code(s)
NDC Code(s): 59078-314-01, 59078-314-02 - Packager: TONYMOLY CO.,LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 16, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive Ingredient: Water, Butylene Glycol, Sodium Stearate, Alcohol, Xylitol, PEG-12 Dimethicone, Glycerin, PEG-8, Bis-PEG-18 Methyl Ether Dimethyl Silane, Niacinamide, Sodium Citrate, Potassium Alginate, Methyl Methacrylate Crosspolymer, Xanthan Gum, Phenoxyethanol, Citric Acid, Actinidia Arguta Fruit Extract, Sea Water, Bambusa Vulgaris Shoot Extract, Bambusa Vulgaris Sap Extract, Glacier Water, Fragrance, Adenosine, Steareth-20, Caprylyl Glycol, 1,2-Hexanediol, CI 42090, Caprylhydroxamic Acid, N-Hydroxysuccinimide, Chlorhexidine Digluconate, Potassium Sorbate, Chrysin, Palmitoyl Oligopeptide, Palmitoyl Tetrapeptide-7, Tropolone
- PURPOSE
- WARNINGS
- KEEP OUT OF REACH OF CHILDREN
- Use
- Directions
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PANDAS DREAM SO COOL EYE SERUM
dimethicone stickProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59078-314 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Dimethicone (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) Dimethicone 0.09 g in 9 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Butylene Glycol (UNII: 3XUS85K0RA) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59078-314-02 1 in 1 CARTON 05/01/2016 1 NDC:59078-314-01 9 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 05/01/2016 Labeler - TONYMOLY CO.,LTD (688216798) Registrant - TONYMOLY CO.,LTD (688216798) Establishment Name Address ID/FEI Business Operations TONYMOLY CO.,LTD 688216798 manufacture(59078-314)