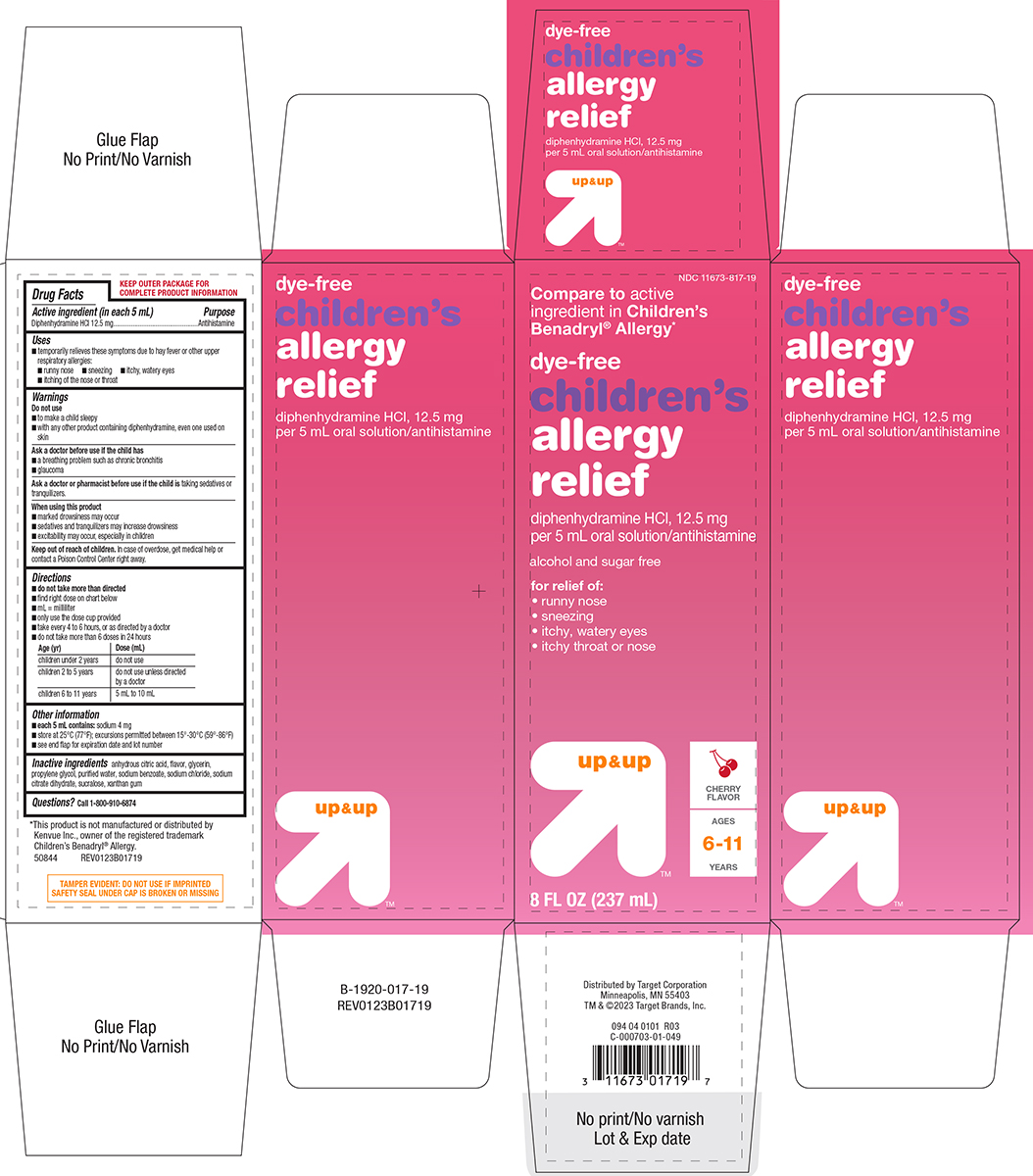
Label: CHILDRENS ALLERGY RELIEF DYE-FREE- diphenhydramine hcl solution
- NDC Code(s): 11673-817-19, 11673-817-36
- Packager: Target Corporation
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 13, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each 5 mL)
- Purpose
- Uses
- Warnings
-
Directions
- do not take more than directed
- find right dose on chart below
- mL = milliliter
- only use the dose cup provided
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 doses in 24 hours
Age (yr) Dose (mL) children under 2 years do not use children 2 to 5 years do not use unless
directed by a doctorchildren 6 to 11 years 5 mL to 10 mL
- Other information
- Inactive ingredients
- Questions?
-
Principal Display Panel
NDC 11673-817-19
Compare to active
ingredient in Children's
Benadryl® Allergy*dye-free
Children's
allergy
reliefdiphenhydramine HCl, 12.5 mg
per 5 mL oral solution/antihistaminealcohol and sugar free
for relief of:
• runny nose
• sneezing
• itchy, watery eyes
• itchy throat or noseup & up™
CHERRY
FLAVORAGES
6-11
YEARS8 FL OZ (237 mL)
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING*This product is not manufactured or distributed by
Kenvue Inc., owner of the registered trademark
Children’s Benadryl® Allergy.
50844 REV0123B01719Distributed by Target Corporation
Minneapolis, MN 55403
TM & ©2023 Target Brands, Inc.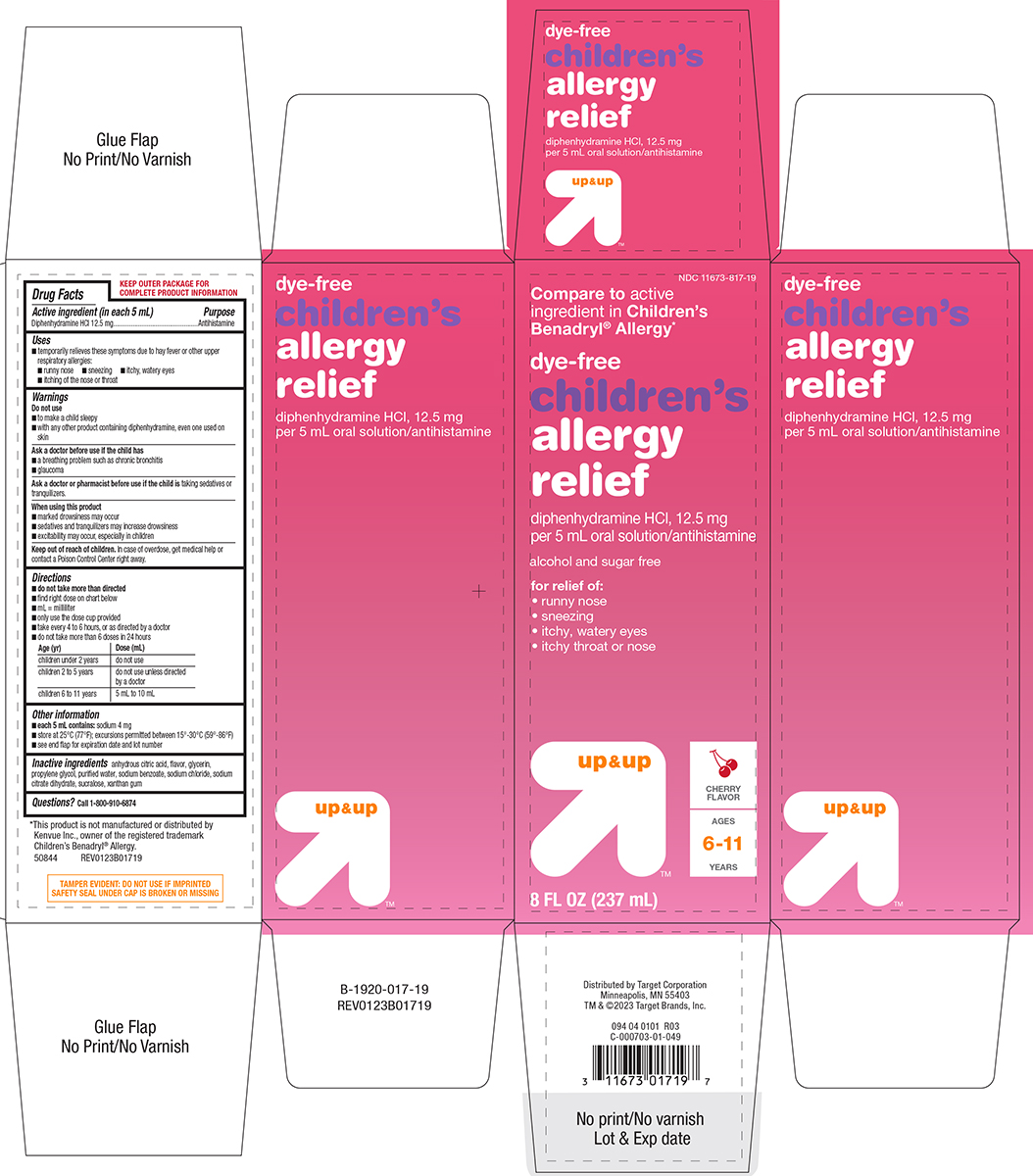
Target 44-017
-
INGREDIENTS AND APPEARANCE
CHILDRENS ALLERGY RELIEF DYE-FREE
diphenhydramine hcl solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11673-817 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 12.5 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM CHLORIDE (UNII: 451W47IQ8X) TRISODIUM CITRATE DIHYDRATE (UNII: B22547B95K) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11673-817-19 1 in 1 CARTON 03/11/2018 1 237 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:11673-817-36 1 in 1 CARTON 03/11/2018 10/08/2021 2 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 03/11/2018 Labeler - Target Corporation (006961700) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 manufacture(11673-817) , pack(11673-817)