Label: DOCOSANOL cream
- NDC Code(s): 21922-023-17, 21922-023-18
- Packager: Encube Ethicals Private Limited
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated October 17, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
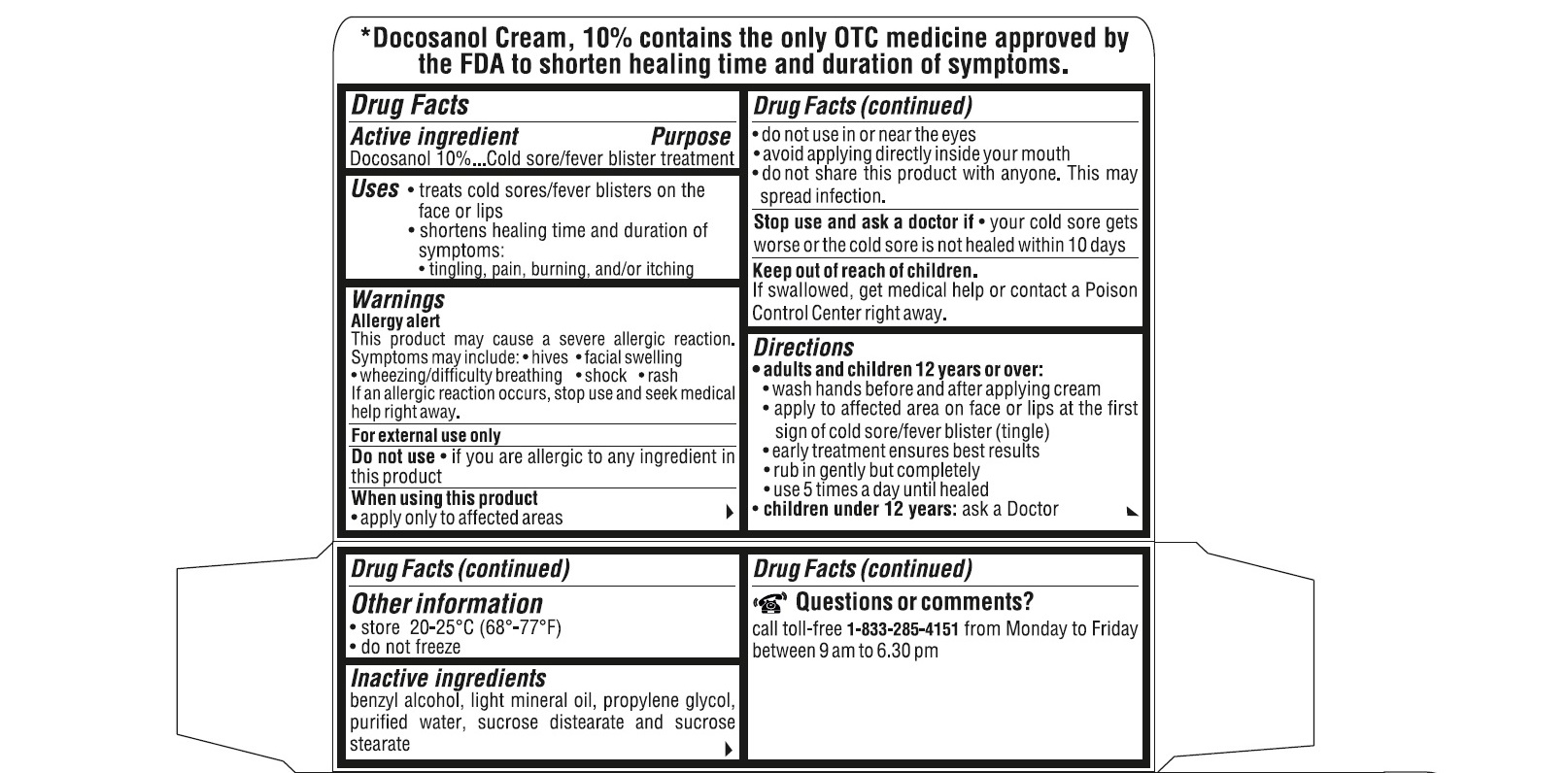
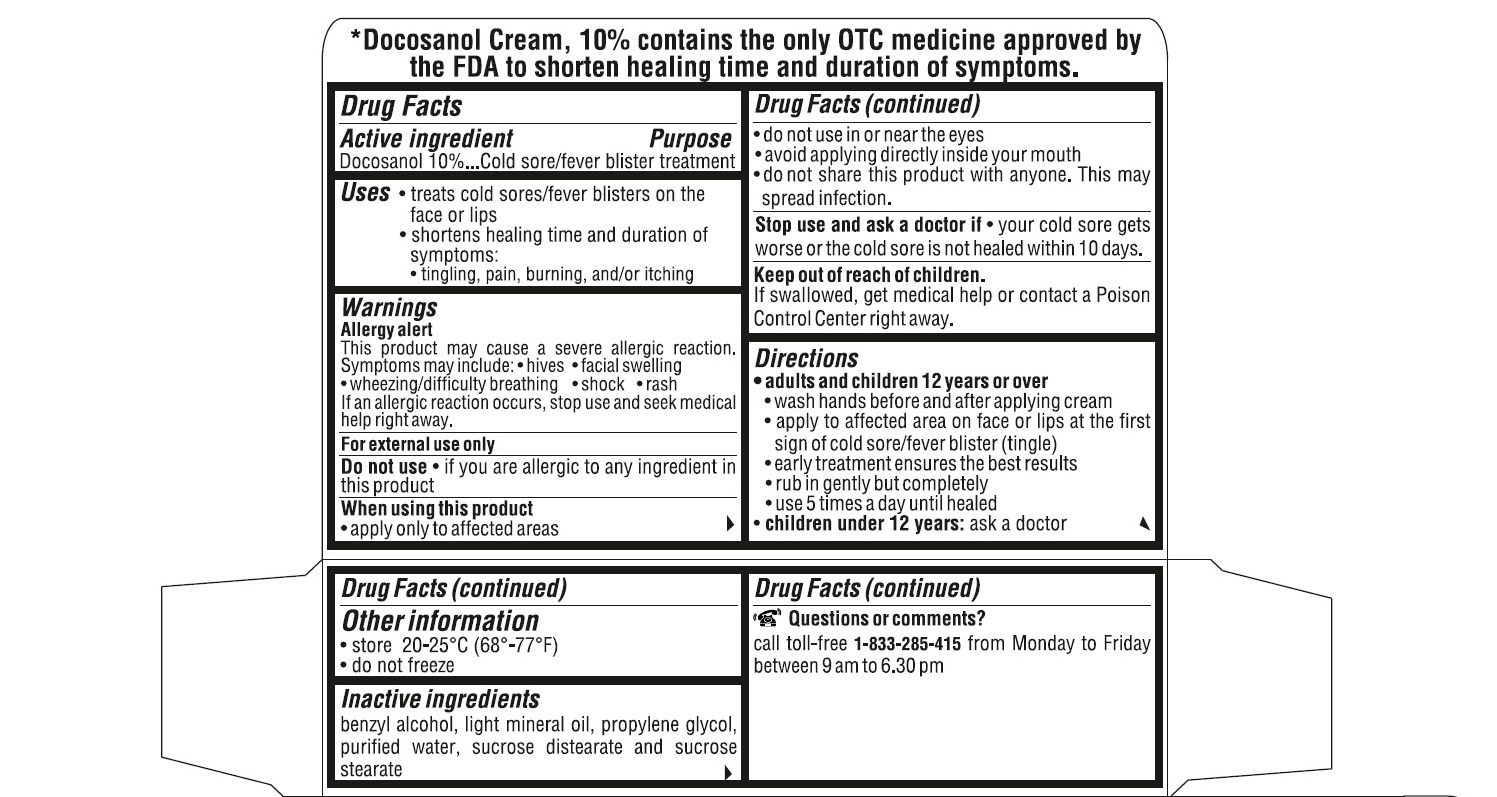
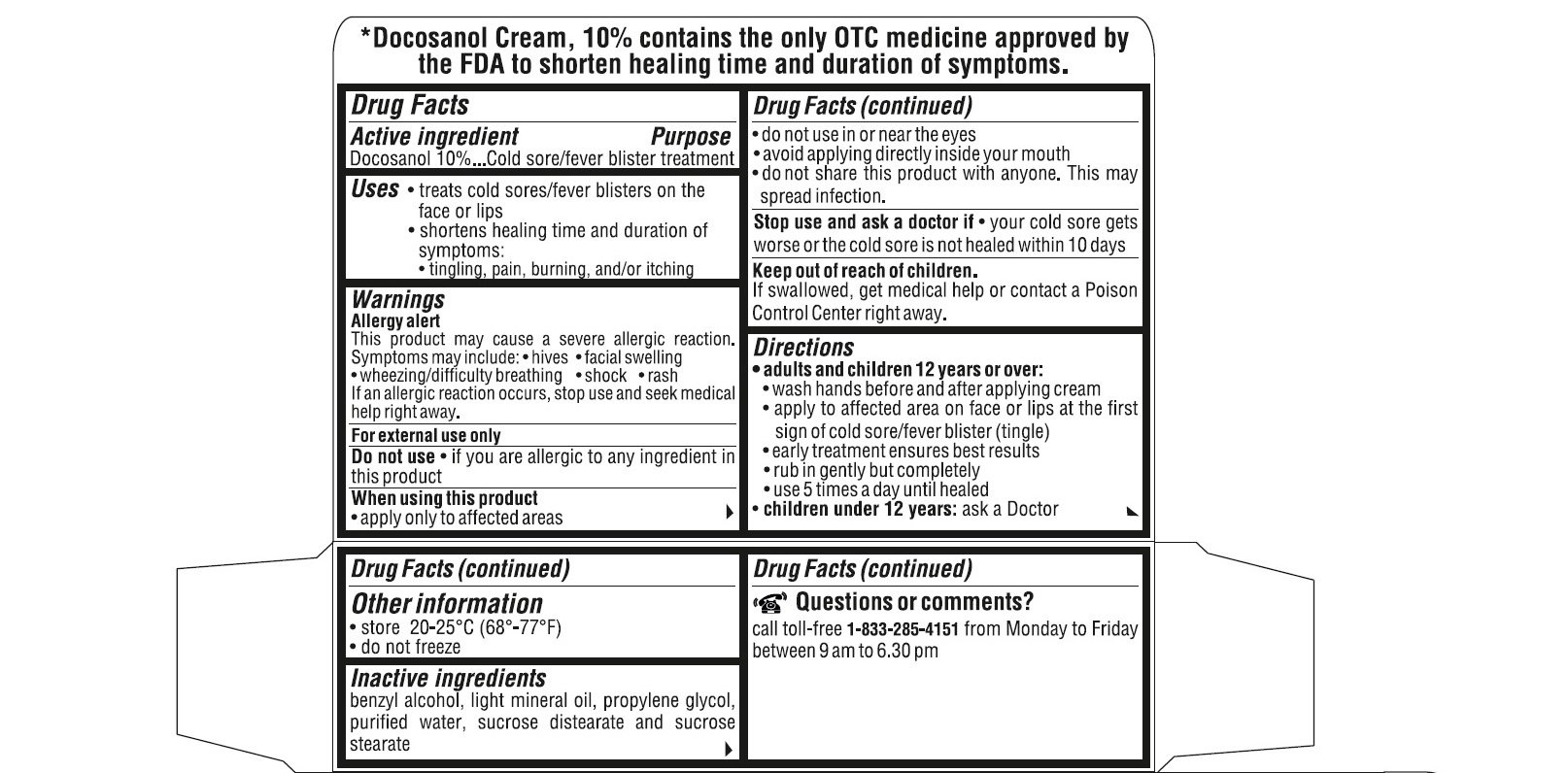
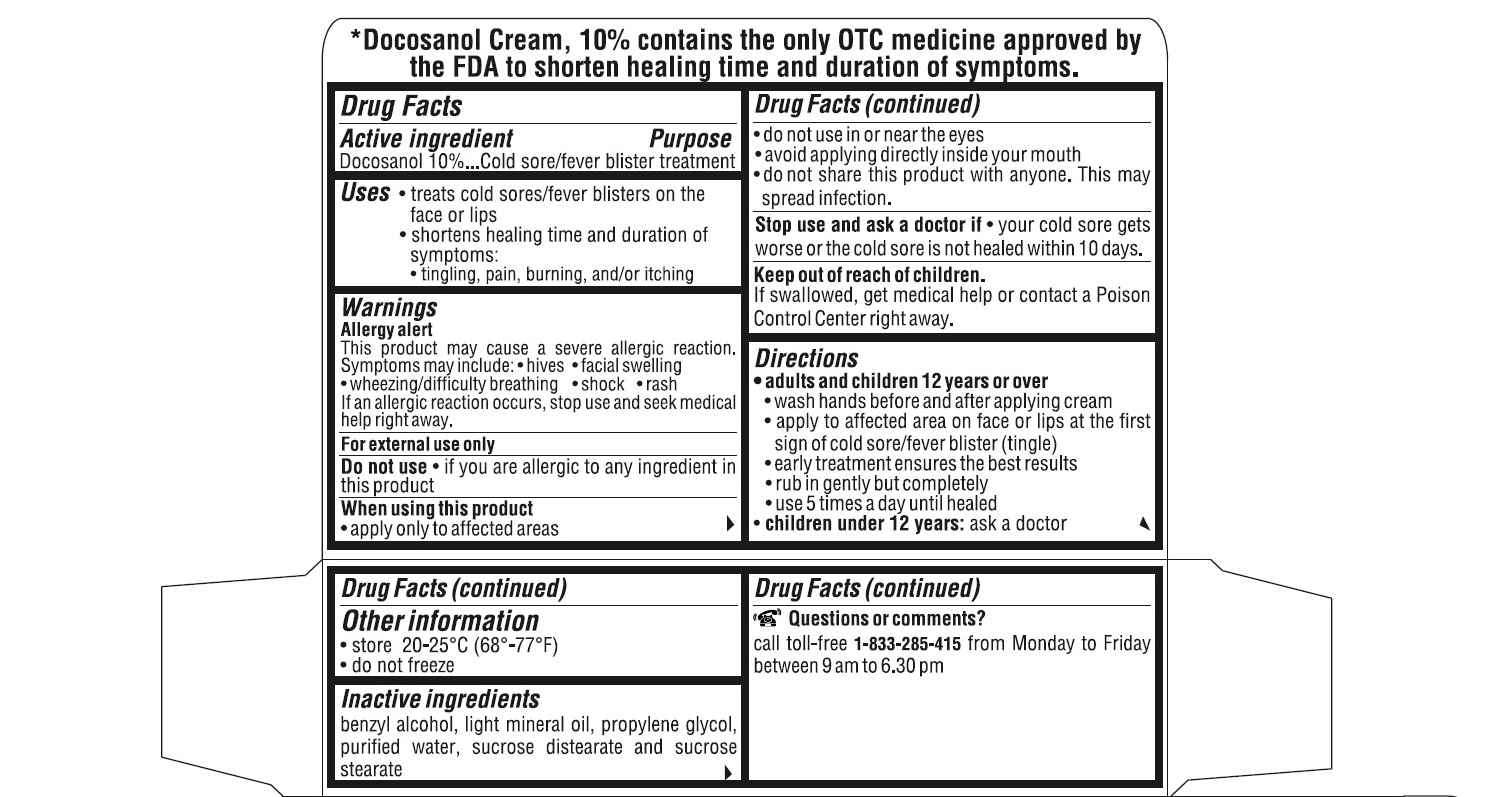
- ACTIVE INGREDIENT(S)
- PURPOSE
- USE(S)
- WARNINGS
- DO NOT USE
- STOP USE AND ASK DOCTOR IF
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
adults and children 12 years or over:
- wash hands before and after applying cream
- apply to affected area on face or lips at the first sign of cold sore/fever blister (tingle).
- early treatment ensures the best results
- rub in gently but completely
- use 5 times a day until healed
children under 12 years : ask a doctor
- STORAGE
- INACTIVE INGREDIENTS
-
QUESTIONS OR COMMENTS?
call toll-free 1-833-285-4151 from Monday to Friday between 9 am to 6.30 pm
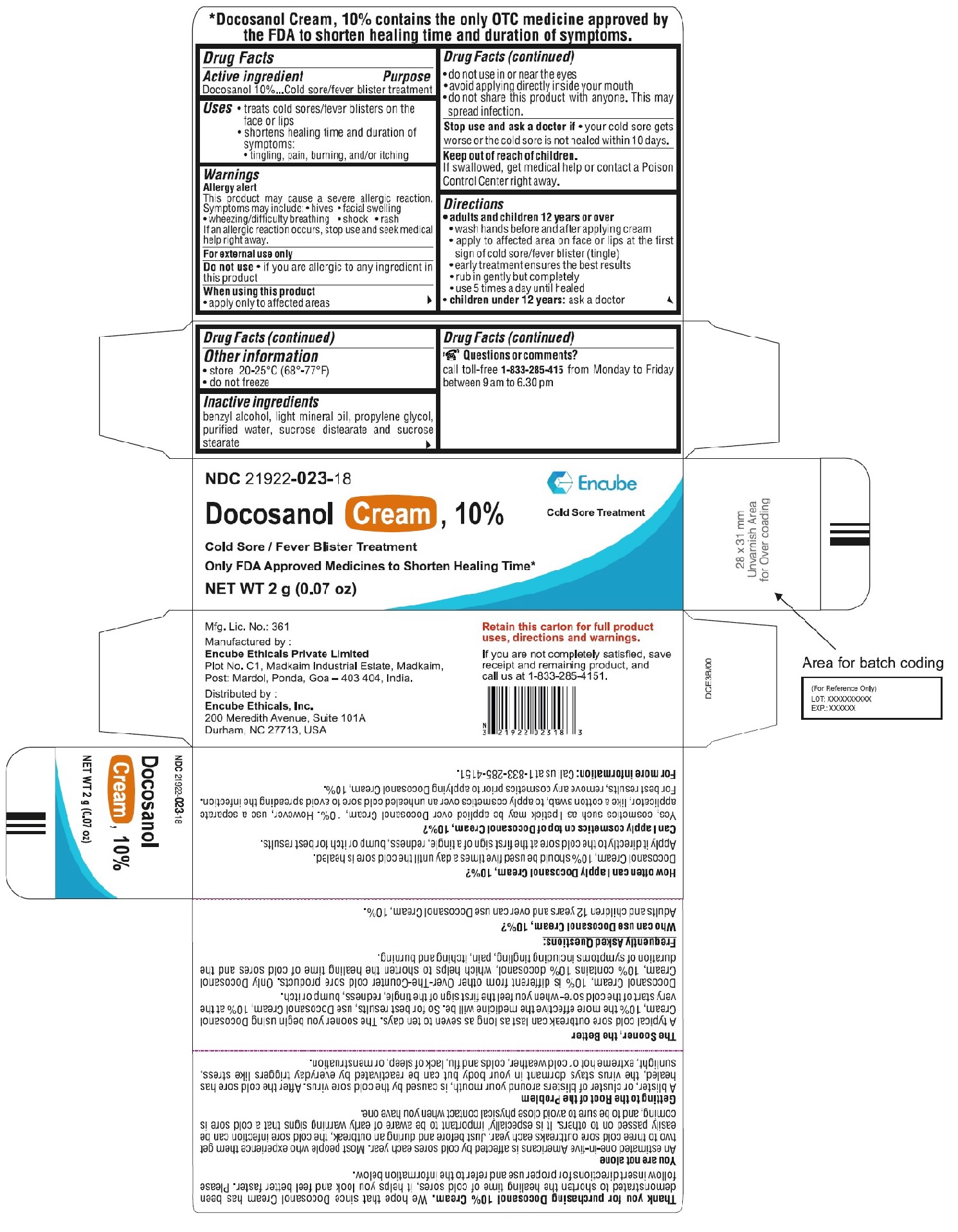
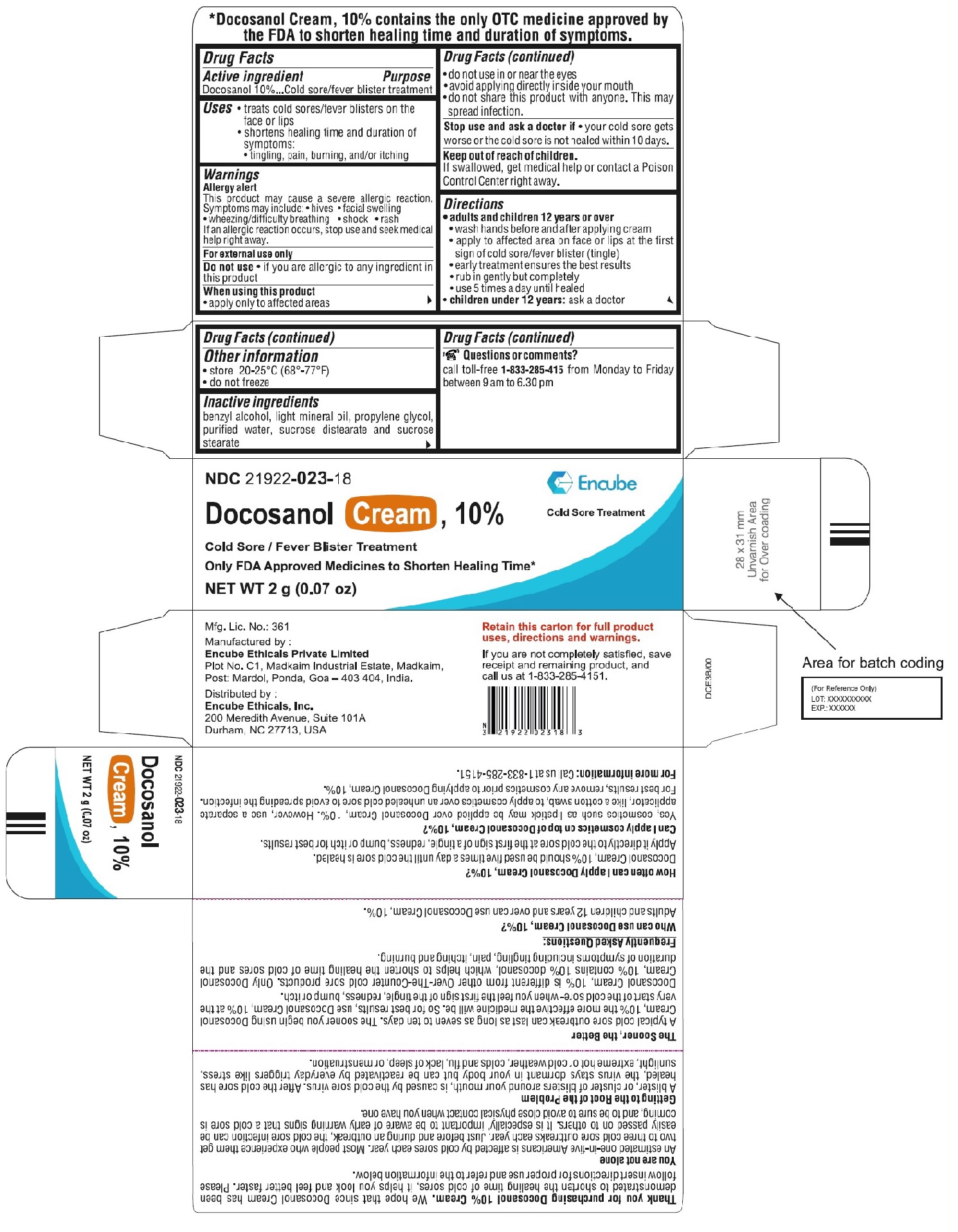
Manufactured by:
Encube Ethicals Private Limited
Plot No. C1,Madkaim Industrial Estate, Madkaim,
Post: Mardol, Ponda, Goa – 403 404, India
Distributed by:
Encube Ethicals, Inc.
200 Meredith Avenue, Suite 101A
Durham, NC 27713, USA
Thank you for purchasingDocosanol Cream, 10%. We hope that since Docosanol Cream shortens the healing time of cold sores, it helps you look and feel better faster. Please follow insert directions for proper use and refer to the information below.
You are not alone
An estimated one-in-five Americans is affected by cold sores each year. Most people who experience them get two to three cold sore outbreaks each year. Just before and during an outbreak, the cold sore infection can be easily passed on to others. It is especially important to be aware of early warning signs that a cold sore is coming, and to be sure to avoid close physical contact when you have one.
Getting to the Root of the Problem
A blister, or cluster of blisters around your mouth, is caused by the cold sore virus. After the cold sore has healed, the virus stays dormant in your body but can be reactivated by everyday triggers like stress, sunlight, extreme hot or cold weather, colds and flu, lack of sleep, or menstruation.
The Sooner, the Better
A typical cold sore outbreak can last as long as seven to ten days. The sooner you begin using Docosanol Cream, 10%, the more effective the medicine will be. So for best results, use Docosanol Cream, 10% at the very start of the cold sore - when you feel the first sign of the tingle, redness, bump or itch.
Docosanol Cream, 10% is different from other over-the-counter cold sore products. Only Docosanol Cream, 10% contains 10% docosanol, which helps to shorten the healing time of cold sores and the duration of symptoms including tingling, pain, itching and burning.
-
FREQUENTLY ASKED QUESTIONS:
For Pump Bottle
How to use your Docosanol Cream, 10% Pump
- Adults and children 12 years and over can use Docosanol Cream, 10%.
- Docosanol Cream, 10% should be used five times a day until the cold sore is healed. Apply it directly to the cold sore at the first sign of a tingle, redness, bump or itch for best results.
- If you wish, you can apply cosmetics such as lipstick over Docosanol Cream, 10%. However, use a separate applicator, like a cotton swab, to apply cosmetics over an unhealed cold sore to avoid spreading the infection. For best results, remove any cosmetics prior to applying Docosanol Cream, 10%.
Press Pump completely about 6-8 times to get first dose.

STEP 1 – Wash your hands before and after treating the cold sore.
STEP 2 – Remove cap and press pump firmly. When first used, expect to press pump completely about 6-8 times before obtaining first dose.
STEP 3 – Rub in gently but completely 5 times a day until healed for maximum of 10 days.
STEP 4 – Place cap back on pump after each use.
For Tube
Who can use Docosanol Cream, 10%?
Adults and children 12 years and over can use Docosanol Cream, 10%.
How often can I apply Docosanol Cream, 10%?
Docosanol Cream, 10% should be used five times a day until the cold sore is healed. Apply it directly to the cold sore at the first sign of a tingle, redness, bump or itch for best results.
Can I apply cosmetics on top of Docosanol Cream, 10%?
Yes, cosmetics such as lipstick may be applied over Docosanol Cream, 10%. However, use a separate applicator, like a cotton swab, to apply cosmetics over an unhealed cold sore to avoid spreading the infection. For best results, remove any cosmetics prior to applying Docosanol Cream, 10%.
-
PRINCIPAL DISPLAY PANEL
For Pump Bottle - Carton Label
NDC 21922-023-17
DOCOSANOL 10% CREAM
Cold Sore/Fever Blister Treatment
Only FDA Approved to Shorten Healing Time*
Cold Sore Treatment
NET WT
2g (0.07oz)



For Pump Bottle - Bottle Label
NDC 21922-023-17
DOCOSANOL 10% CREAM
Cold Sore/Fever Blister Treatment
Only FDA Approved to Shorten Healing Time*
Cold Sore Treatment
NET WT
2g (0.07oz)
For Tube - Carton
NDC 21922-023-18
DOCOSANOL 10% CREAM
Cold Sore/Fever Blister Treatment
Only FDA Approved to Shorten Healing Time*
Cold Sore Treatment
NET WT
2g (0.07 oz)


For Tube
NDC 21922-023-18
DOCOSANOL 10% CREAM
Cold Sore/Fever Blister Treatment
Only FDA Approved to Shorten Healing Time*
Cold Sore Treatment
NET WT
2g (0.07 oz)

-
INGREDIENTS AND APPEARANCE
DOCOSANOL
docosanol creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21922-023 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DOCOSANOL (UNII: 9G1OE216XY) (DOCOSANOL - UNII:9G1OE216XY) DOCOSANOL 100 mg in 1 g Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) LIGHT MINERAL OIL (UNII: N6K5787QVP) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SUCROSE DISTEARATE (UNII: 33X4X4B90S) SUCROSE STEARATE (UNII: 274KW0O50M) Product Characteristics Color WHITE Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21922-023-18 1 in 1 PACKAGE 10/10/2022 1 2 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:21922-023-17 1 in 1 PACKAGE 10/10/2022 2 2 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA212385 10/10/2022 Labeler - Encube Ethicals Private Limited (915834105) Establishment Name Address ID/FEI Business Operations Encube Ethicals Private Limited 725076298 ANALYSIS(21922-023) , LABEL(21922-023) , MANUFACTURE(21922-023) , PACK(21922-023)