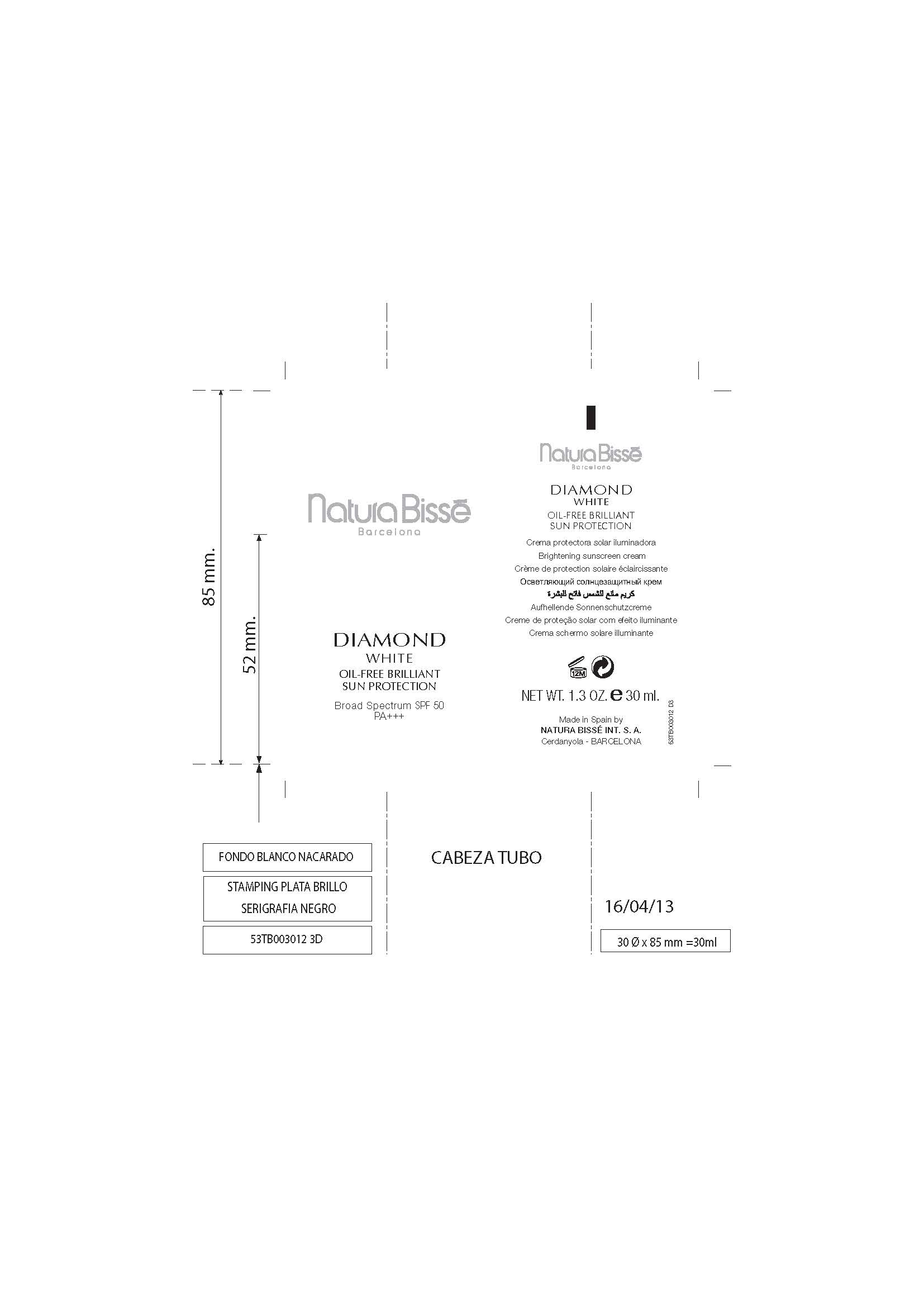
Label: DIAMOND WHITE OIL-FREE BRILLIANT SUN PROTECTION- ensulizole, octinoxate, titanium dioxide, zinc oxide cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 63730-223-01 - Packager: Natura Bisse International SA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 20, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Natura Bisse
Barcelona
DIMAOND
WHITE
OIL-FREE BRILLIANT
SUN PROTECTION
Broad Spectrum SPF 50
PA+++
Crema protectora solar iluminadora
Brightening Sunscreen cream
Creme de protection solaire eclaircissante
Aufhellende Sonnenschutzcreme
Creme de protecao solar com efeito iluminante
Crema schermo solare illuminante
Net Wt. 1.3 oz. e 30 ml
- WARNINGS
-
DOSAGE & ADMINISTRATION
Directions
- apply liberally 15 minutes before skin exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
- children under 6 months: ask a doctor
- Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. - 2 p.m.
- wear long-sleeved shirts, pants, hats, and sun-glasses
-
INACTIVE INGREDIENT
Water (Aqua), Cyclopentasiloxane, Dicaprylyl Carbonate, Glycerin, Cetyl PEG/PPG-10/1 Dimethicone, PEG-10 Dimethicone, Arginine, Stearic Acid, Aluminum Hydroxide, Lactobacillus/Punica Granatum Fruit Ferment Extract, Leuconostoc/Radish Root Ferment Filtrate, Bisabolol, Tocopheryl Acetate, Glyceryl Linoleate, Glyceryl Linolenate, Triethoxycaprylylsilane, Diphenyl Dimethicone/Vinyl Diphenyl Dimethicone/Silsesquioxane Crosspolymer, Aluminum Stearate, Hexyl Laurate, Polyglyceryl-4 Isostearate, Isononyl Isononanoate, Dimethiconol, Dimethicone Crosspolymer, Stearalkonium Hectorite, Disodium EDTA, Ethylhexylglycerin, Propylene Carbonate, Sodium Chloride, Phenoxyethanol, Fragrance (Parfum), Lavandula Hybrida Oil, Linalool, Coumarin, Amyl Cinnamal, Alumina, Iron Oxides (CI 77491, CI 77492, CI 77499).
-
OTHER SAFETY INFORMATION
Made in Spain By
NATURA BISSE INT. S. A.
PQUE TECNOL. del VALLES
Cerdanyola - BARCELONA
Other information protect this product from excessive heat and direct sun.
Questions? 1-800-762-8872
ESTE PROUCTO SOLO PUEDE VENDERSE EN ESTABLECIMIENTOS AUTORIZADOS POR NATURA BISSE
THIS PRODUCT CAN BE SOLD ONLY BY AUTHORIZED NATURA BISSE RETAILERS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIAMOND WHITE OIL-FREE BRILLIANT SUN PROTECTION
ensulizole, octinoxate, titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63730-223 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 2 g in 30 g ENSULIZOLE (UNII: 9YQ9DI1W42) (ENSULIZOLE - UNII:9YQ9DI1W42) ENSULIZOLE 1 g in 30 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 2 g in 30 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 3 g in 30 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) GLYCERIN (UNII: PDC6A3C0OX) CETYL PEG/PPG-10/1 DIMETHICONE (HLB 2) (UNII: V2W71V8T0X) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) ARGININE (UNII: 94ZLA3W45F) STEARIC ACID (UNII: 4ELV7Z65AP) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) PUNICA GRANATUM WHOLE (UNII: O2ZTS50U5E) LEUCONOSTOC/RADISH ROOT FERMENT FILTRATE (UNII: D2QHA03458) LEVOMENOL (UNII: 24WE03BX2T) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLYCERYL LINOLENATE (UNII: SGB6X4G86K) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) ALUMINUM STEARATE (UNII: U6XF9NP8HM) HEXYL LAURATE (UNII: 4CG9F9W01Q) POLYGLYCERYL-4 ISOSTEARATE (UNII: 820DPX33S7) ISONONYL ISONONANOATE (UNII: S4V5BS6GCX) DIMETHICONOL (100000 CST) (UNII: OSA9UP217S) DIMETHICONE (UNII: 92RU3N3Y1O) STEARALKONIUM HECTORITE (UNII: OLX698AH5P) EDETATE DISODIUM (UNII: 7FLD91C86K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) PROPYLENE CARBONATE (UNII: 8D08K3S51E) SODIUM CHLORIDE (UNII: 451W47IQ8X) PHENOXYETHANOL (UNII: HIE492ZZ3T) LAVANDIN OIL (UNII: 9RES347CKG) LINALOOL, (+/-)- (UNII: D81QY6I88E) COUMARIN (UNII: A4VZ22K1WT) .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) ALUMINUM OXIDE (UNII: LMI26O6933) BROWN IRON OXIDE (UNII: 1N032N7MFO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63730-223-01 30 g in 1 BOTTLE; Type 0: Not a Combination Product 09/20/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 04/21/2014 Labeler - Natura Bisse International SA (464431576) Establishment Name Address ID/FEI Business Operations Natura Bisse International SA 464431576 manufacture(63730-223)