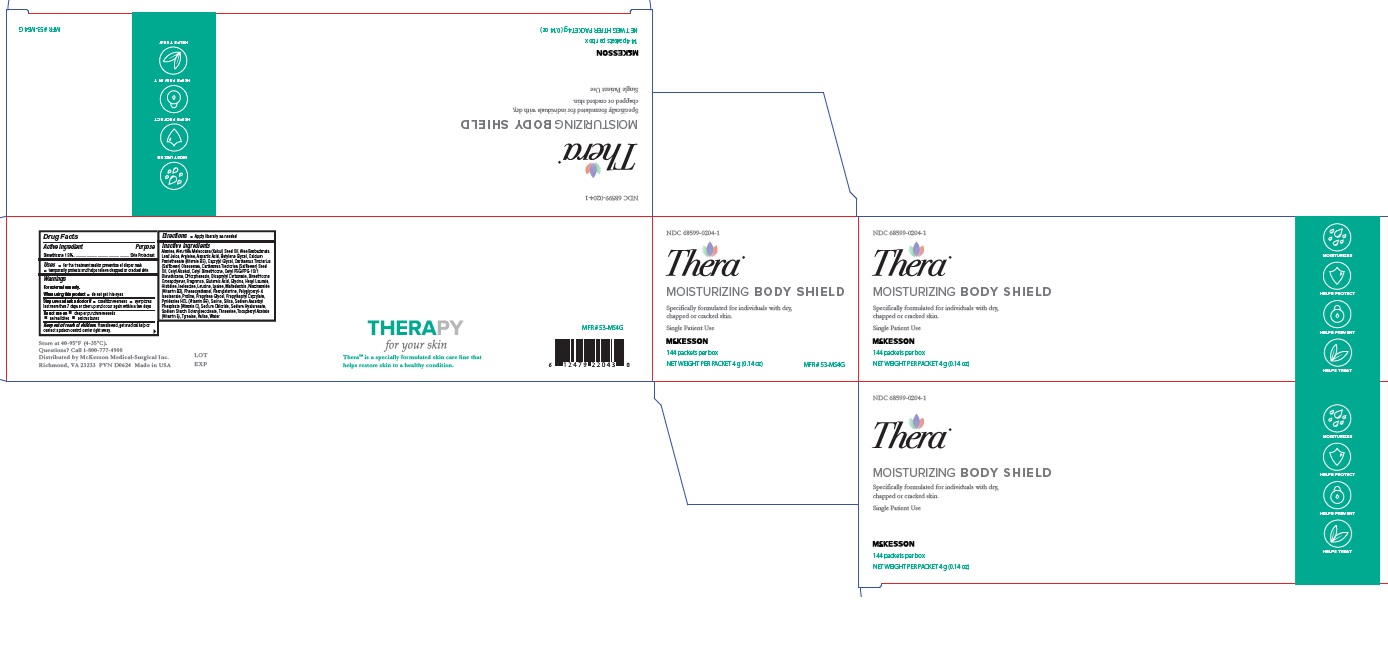
Label: THERA MOISTURIZING BODY SHIELD- dimethicone cream
- NDC Code(s): 68599-0204-1, 68599-0204-4
- Packager: McKesson Medical-Surgical Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 18, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Keep out of reach of children
- Uses
- Warnings
- Directions
-
Inactive ingredients
Inactive ingredients
Alanine, Aleurites Moluccana (Kukui) Seed Oil, Aloe
Barbadensis Leaf Juice, Arginine, Aspartic Acid, Butylene
Glycol, Calcium Pantothenate (Vitamin B5), Caprylyl Glycol,
Carthamus Tinctorius (Safflower) Oleosomes, Carthamus
Tinctorius (Safflower) Seed Oil, Cetyl Alcohol, Cetyl
Dimethicone, Cetyl PEG/PPG-10/1 Dimethicone,
Chlorphenesin, Dicaprylyl Carbonate, Dimethicone
Crosspolymer, Fragrance, Glutamic Acid, Glycine, Hexyl
Laurate, Histidine, Isoleucine, Leucine, Lysine, Maltodextrin,
Niacinamide (Vitamin B3), Phenoxyethanol, Phenylalanine,
Polyglyceryl-4 Isostearate, Proline, Propylene Glycol,
Propylheptyl Caprylate, Pyridoxine HCL (Vitamin B6), Serine,
Silica, Sodium Ascorbyl Phosphate (Vitamin C), Sodium
Chloride, Sodium Hyaluronate, Sodium Starch
Octenylsuccinate, Threonine, Tocopheryl Acetate (Vitamin
E), Tyrosine, Valine, Water - Label (4 oz)
- Label (4g)
-
INGREDIENTS AND APPEARANCE
THERA MOISTURIZING BODY SHIELD
dimethicone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68599-0204 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 15 mg in 1 g Inactive Ingredients Ingredient Name Strength GLUTAMIC ACID (UNII: 3KX376GY7L) THREONINE (UNII: 2ZD004190S) CHLORPHENESIN (UNII: I670DAL4SZ) LYSINE (UNII: K3Z4F929H6) POLYGLYCERYL-4 ISOSTEARATE (UNII: 820DPX33S7) GLYCINE (UNII: TE7660XO1C) SERINE (UNII: 452VLY9402) HEXYL LAURATE (UNII: 4CG9F9W01Q) HISTIDINE (UNII: 4QD397987E) ALANINE (UNII: OF5P57N2ZX) SAFFLOWER OIL (UNII: 65UEH262IS) CETYL PEG/PPG-10/1 DIMETHICONE (HLB 2) (UNII: V2W71V8T0X) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ISOLEUCINE (UNII: 04Y7590D77) TYROSINE (UNII: 42HK56048U) VALINE (UNII: HG18B9YRS7) KUKUI NUT OIL (UNII: TP11QR7B8R) ALOE VERA LEAF (UNII: ZY81Z83H0X) CALCIUM PANTOTHENATE (UNII: 568ET80C3D) MALTODEXTRIN (UNII: 7CVR7L4A2D) NIACINAMIDE (UNII: 25X51I8RD4) PYRIDOXINE HYDROCHLORIDE (UNII: 68Y4CF58BV) SODIUM ASCORBYL PHOSPHATE (UNII: 836SJG51DR) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) ARGININE (UNII: 94ZLA3W45F) CARTHAMUS TINCTORIUS SEED OLEOSOMES (UNII: 9S60Q72309) CAPRYLYL GLYCOL (UNII: 00YIU5438U) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) PHENOXYETHANOL (UNII: HIE492ZZ3T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLHEPTYL CAPRYLATE (UNII: 991Z19V2OD) WATER (UNII: 059QF0KO0R) LEUCINE (UNII: GMW67QNF9C) PHENYLALANINE (UNII: 47E5O17Y3R) SODIUM CHLORIDE (UNII: 451W47IQ8X) PROLINE (UNII: 9DLQ4CIU6V) CETYL ALCOHOL (UNII: 936JST6JCN) ASPARTIC ACID (UNII: 30KYC7MIAI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68599-0204-4 113 g in 1 TUBE; Type 0: Not a Combination Product 09/01/2011 2 NDC:68599-0204-1 144 in 1 CARTON 09/01/2011 2 4 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 09/01/2011 Labeler - McKesson Medical-Surgical Inc. (023904428) Establishment Name Address ID/FEI Business Operations Central Solutions 007118524 manufacture(68599-0204)