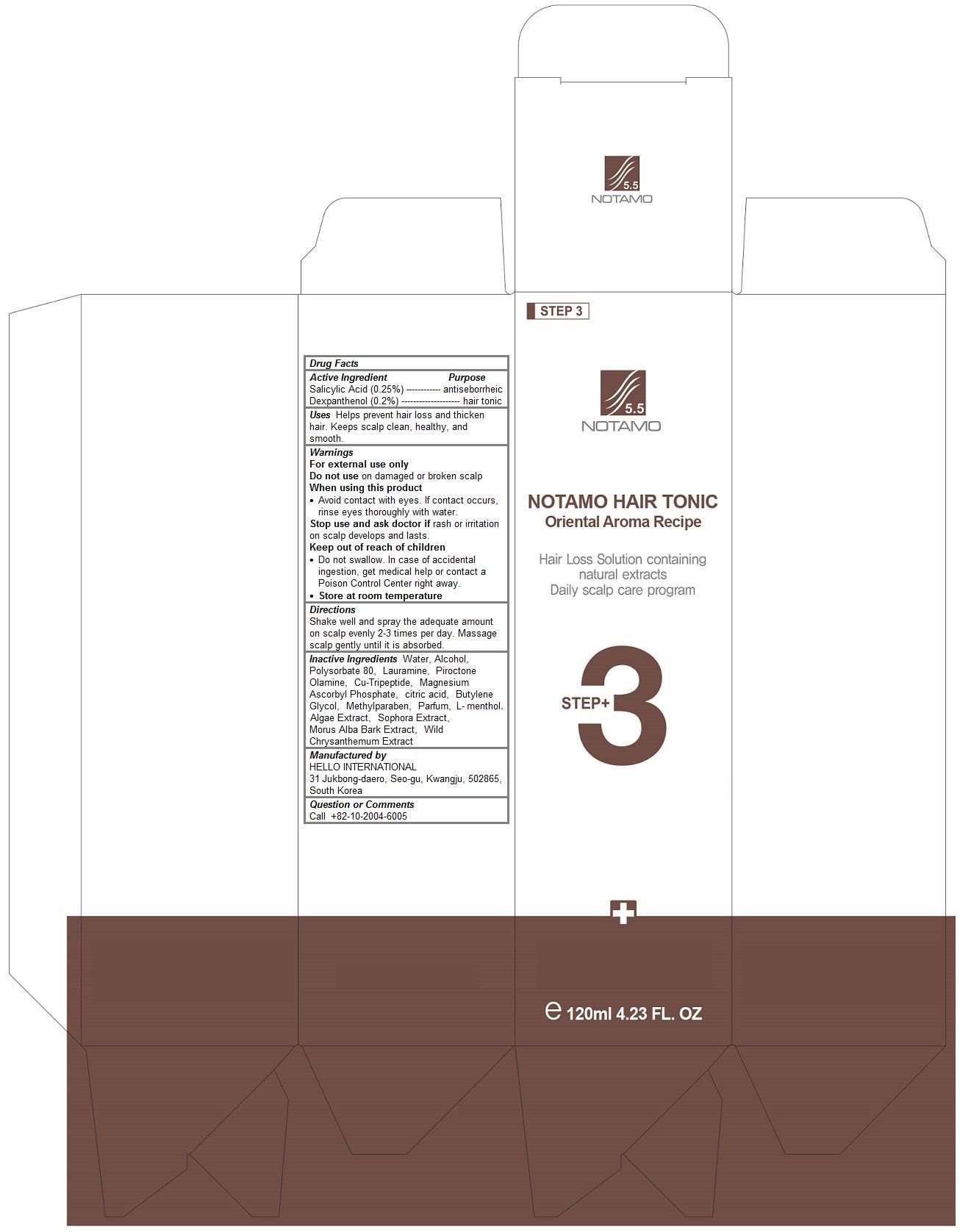
Label: NOTAMO HAIR TONIC- salicylic acid, panthenol liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 69709-102-01, 69709-102-02 - Packager: HELLO INTERNATIONAL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 8, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Notamo Hair Tonic
- Salicylic Acid (0.25%)Dexpanthenol (0.2%)
- antiseborrheichair tonic
- Keep out of reach of children- Do not swallow. In case of accidental ingestion, get medical help or contact a Poison Control Center right away.
- Helps prevent hair loss and thicken hair. Keeps scalp clean, healthy, and smooth.
- WarningsFor external use onlyDo not use on damaged or broken scalpWhen using this product- Avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.Stop use and ask doctor if rash or irritation on scalp develops and lasts.Store at room temperature
- DirectionsShake well and spray the adequate amount on scalp evenly 2-3 times per day. Tap and massage scalp gently using fingertips until it is absorbed.
- Water, Alcohol, Polysorbate 80, Lauramine, Piroctone Olamine, Cu-Tripeptide, Magnesium Ascorbyl Phosphate, citric acid, Butylene Glycol, Methylparaben, Parfum, L-menthol , Algae Extract, Sophora Extract, Morus Alba Bark Extract, Wild Chrysanthemum Extract
-
INGREDIENTS AND APPEARANCE
NOTAMO HAIR TONIC
salicylic acid, panthenol liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69709-102 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXPANTHENOL (UNII: 1O6C93RI7Z) (DEXPANTHENOL - UNII:1O6C93RI7Z) DEXPANTHENOL 0.2 in 120 mL SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 0.25 in 120 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Alcohol (UNII: 3K9958V90M) Polysorbate 80 (UNII: 6OZP39ZG8H) DODECYLAMINE (UNII: YWY9OD6A2K) PIROCTONE OLAMINE (UNII: A4V5C6R9FB) Magnesium Ascorbyl Phosphate (UNII: 0R822556M5) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) Butylene Glycol (UNII: 3XUS85K0RA) Methylparaben (UNII: A2I8C7HI9T) LEVOMENTHOL (UNII: BZ1R15MTK7) SOPHORA FLAVESCENS ROOT (UNII: IYR6K8KQ5K) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69709-102-02 1 in 1 PACKAGE 1 NDC:69709-102-01 120 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/08/2015 Labeler - HELLO INTERNATIONAL (689062090) Registrant - HELLO INTERNATIONAL (689062090) Establishment Name Address ID/FEI Business Operations HELLO INTERNATIONAL 689062090 manufacture(69709-102)