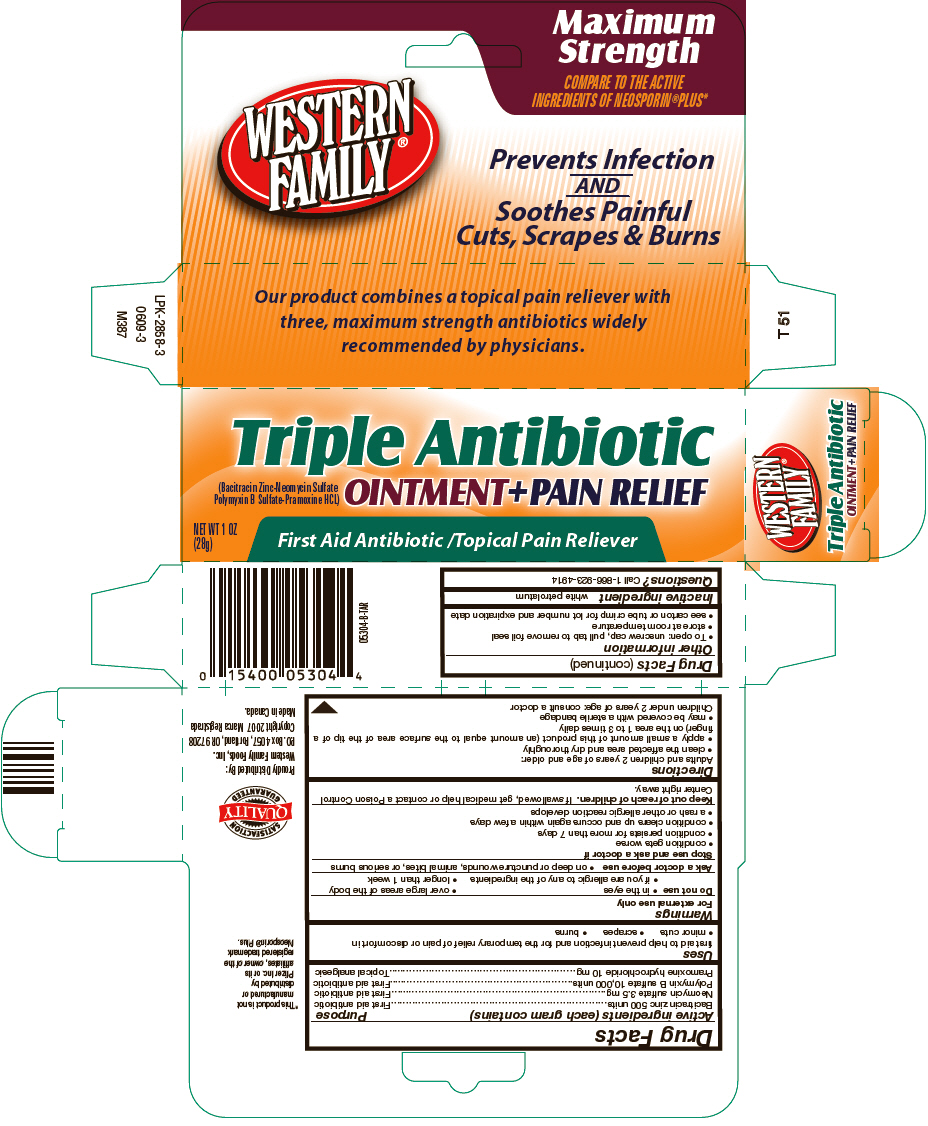
Label: WESTERN FAMILY TRIPLE ANTIBIOTIC PLUS PAIN RELIEF- bacitracin zinc, neomycin sulfate, polymyxin b sulfate, and pramoxine hydrochloride ointment
-
Contains inactivated NDC Code(s)
NDC Code(s): 55312-504-02 - Packager: Western Family Foods Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 15, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
For external use only
Do not use
- in the eyes
- over large areas of the body
- if you are allergic to any of the ingredients
- longer than 1 week
-
Directions
Adults and children 2 years of age and older:
- clean the affected area and dry thoroughly
- apply a small amount of this product (an amount equal to the surface area of the tip of a finger) on the area 1 to 3 times daily
- may be covered with a sterile bandage
Children under 2 years of age: consult a doctor
- Other information
- Inactive ingredient
- Questions?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 28 g Tube Carton
-
INGREDIENTS AND APPEARANCE
WESTERN FAMILY TRIPLE ANTIBIOTIC PLUS PAIN RELIEF
bacitracin zinc, neomycin sulfate, polymyxin b sulfate, and pramoxine hydrochloride ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:55312-504 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Bacitracin zinc (UNII: 89Y4M234ES) (Bacitracin - UNII:58H6RWO52I) Bacitracin 500 [iU] in 1 g Neomycin sulfate (UNII: 057Y626693) (Neomycin - UNII:I16QD7X297) Neomycin 3.5 mg in 1 g Polymyxin B sulfate (UNII: 19371312D4) (Polymyxin B - UNII:J2VZ07J96K) Polymyxin B 10000 [iU] in 1 g Pramoxine hydrochloride (UNII: 88AYB867L5) (Pramoxine - UNII:068X84E056) Pramoxine hydrochloride 10 mg in 1 g Inactive Ingredients Ingredient Name Strength petrolatum (UNII: 4T6H12BN9U) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55312-504-02 1 in 1 CARTON 1 28 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part333B 03/31/2012 Labeler - Western Family Foods Inc (192166072) Registrant - Taro Pharmaceuticals U.S.A., Inc. (145186370) Establishment Name Address ID/FEI Business Operations Taro Pharmaceuticals Inc. 206263295 MANUFACTURE(55312-504)