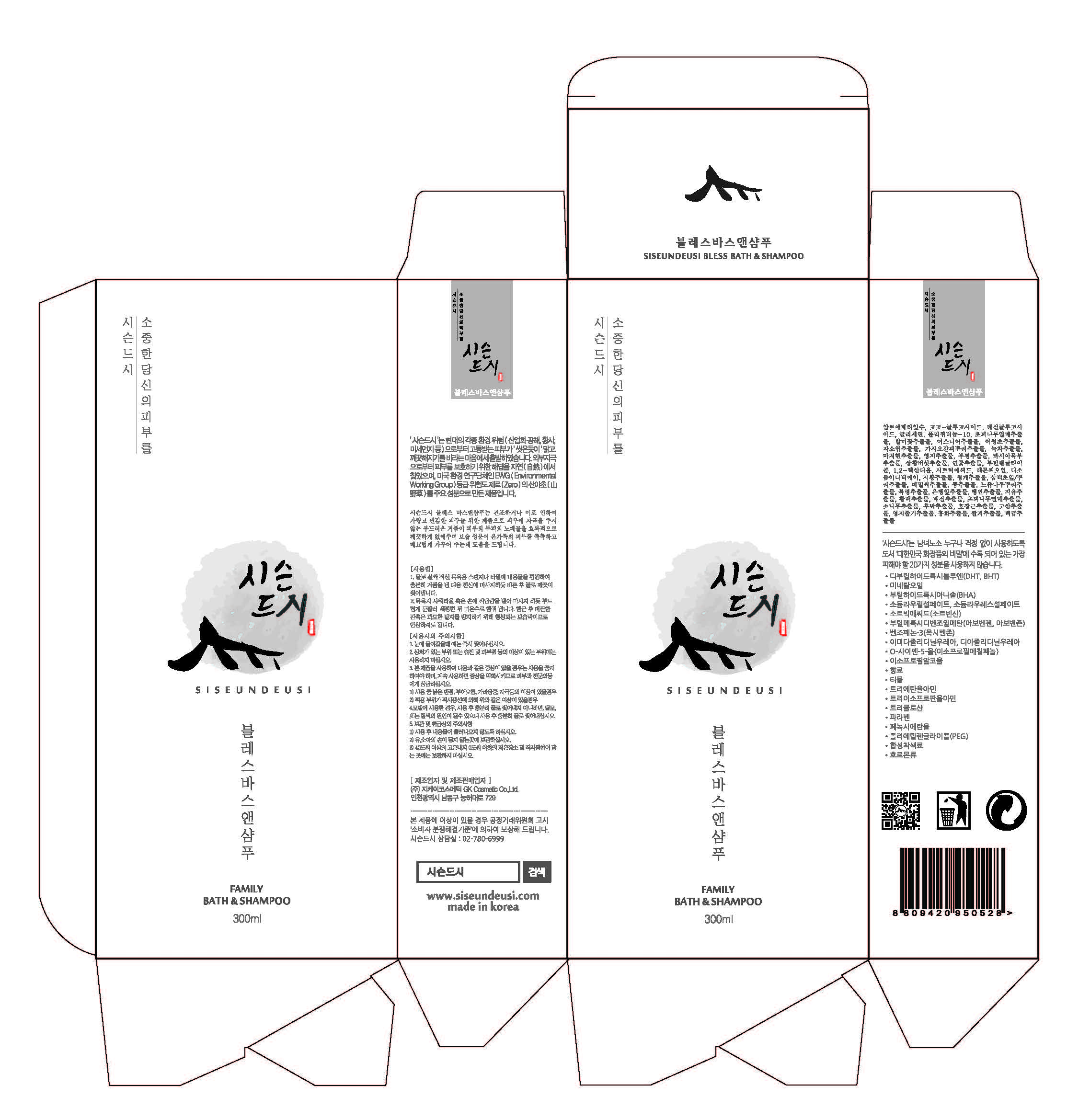
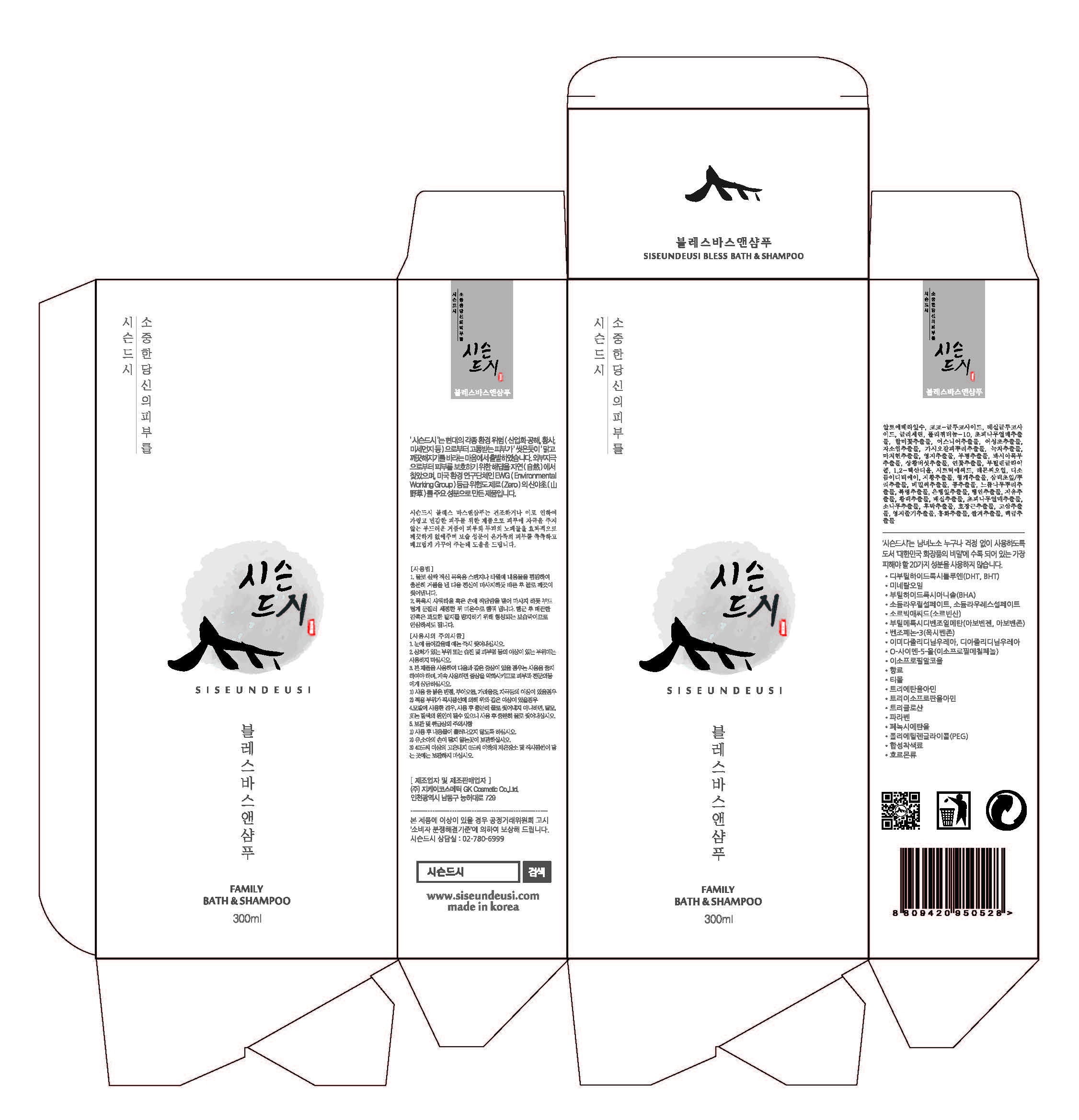
Label: SISEUNDEUSI BLESS BATHANDSHAMPOO- glycerin gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 70098-0007-1 - Packager: GK COSMETIC CO., LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 9, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
- Pump adequate amount of Bless Bath & Shampoo onto a wet sponge or washcloth and lather it with sponge or washcloth. Then gently rub your entire body, and rinse it all off
- Pour adequate amount of Bless Bath & Shampoo on your hand or washcloth in the shower and gently rub your body and then rinse it all off with lukewarm water. Be relieved, smoothness after the wash off is generated moisture layer to avoid too much oil removal from the skin.
- DOSAGE & ADMINISTRATION
-
WARNINGS
1. Do not use in the following cases(Eczema and scalp wounds)
2.Side Effects
1)Due to the use of this druf if rash, irritation, itching and symptopms of hypersnesitivity occur dicontinue use and consult your phamacisr or doctor
3.General Precautions
1)If in contact with the eyes, wash out thoroughty with water If the symptoms are servere, seek medical advice immediately
2)This product is for exeternal use only. Do not use for internal use
4.Storage and handling precautions
1)If possible, avoid direct sunlight and store in cool and area of low humidity
2)In order to maintain the quality of the product and avoid misuse
3)Avoid placing the product near fire and store out in reach of children - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SISEUNDEUSI BLESS BATHANDSHAMPOO
glycerin gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70098-0007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 2.661 g in 100 mL Inactive Ingredients Ingredient Name Strength BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70098-0007-1 300 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 03/09/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part347 03/09/2017 Labeler - GK COSMETIC CO., LTD. (688477309) Registrant - GK COSMETIC CO., LTD. (688477309) Establishment Name Address ID/FEI Business Operations GK COSMETIC CO., LTD. 688477309 manufacture(70098-0007) , label(70098-0007) , pack(70098-0007)