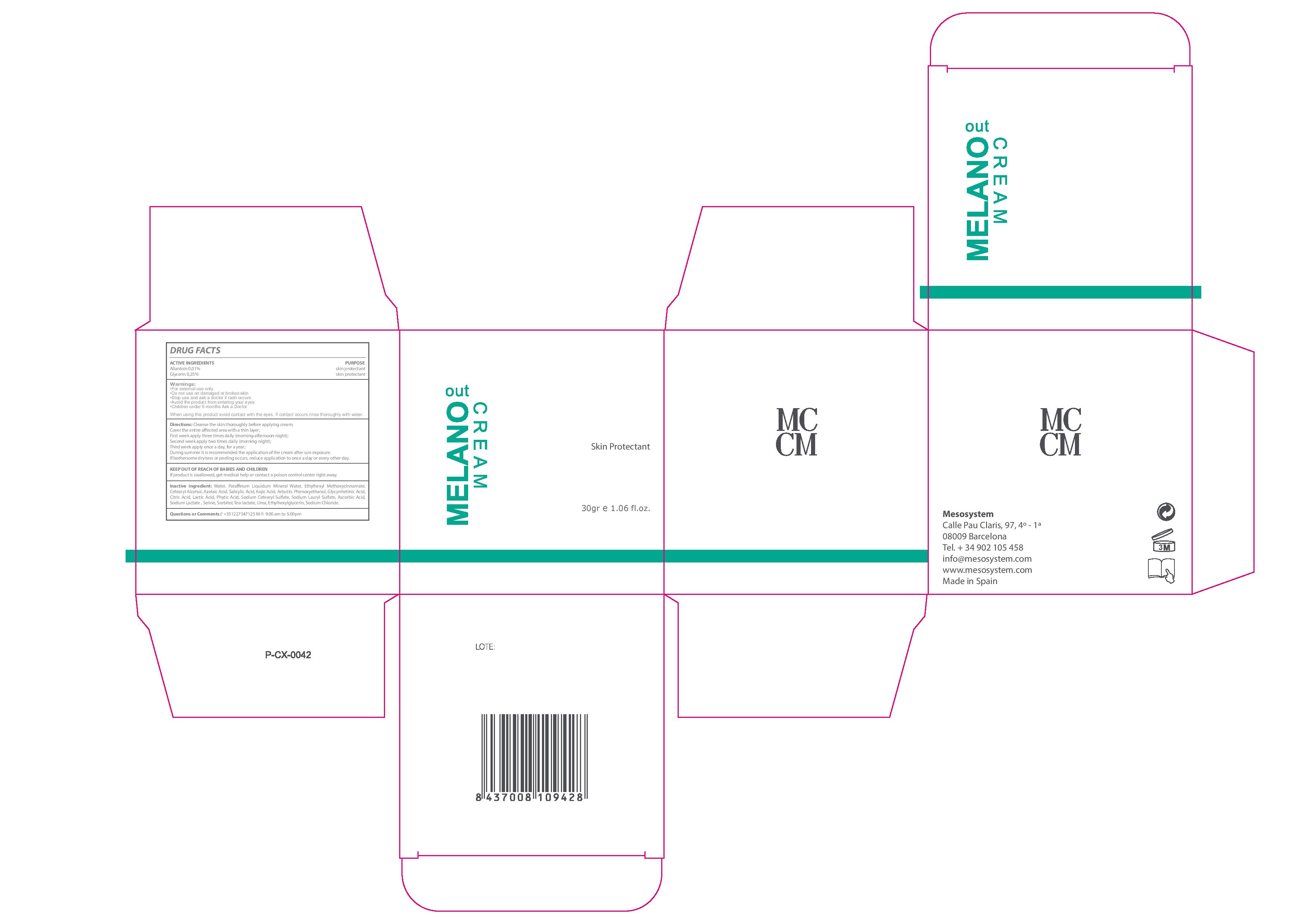
Label: MELANO OUT CREAM- allantoin, glycerin cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 70663-007-01, 70663-007-02 - Packager: MESO SYSTEM S.A.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 17, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Warnings
- WARNINGS
- USES
- KEEP OUT OF REACH OF CHILDREN
-
DIRECTIONS
- Clean the skin thoroughly before applying the cream.
- Cover the entire affected area with a thin layer. First week apply three times daily (morning-afternoon-night). Second week apply two times daily (morning-night). Third week apply once a day for one year.
- During summer it is recommended the application of the cream after sun exposure. If bothersome dryness or peeling occurs, reduce application to once a day or every other day
- QUESTIONS OR COMMENTS ?
- ACTIVE INGREDIENTS
-
Inactive Ingredients
Water, Azelaic Acid, Mineral Oil, Ethylhexyl Methoxycinnamate, Cetearyl Alcohol, Salicylic Acid, Kojic Acid,,Arbutin, Phenoxyethanol, Glycyrrhetinic Acid, Citric Acid, Lactic Acid, Phytic Acid, Sodium Cetearyl Sulfate, Sodium Lauryl Sulfate, Ascorbic Acid, Sodium, Lactate, Serine, Sorbitol, Tea-lactate, Urea, Ethylhexylglycerin, Sodium Chloride

- INDICATIONS & USAGE
- Melano Out Cream
-
INGREDIENTS AND APPEARANCE
MELANO OUT CREAM
allantoin, glycerin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70663-007 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALLANTOIN (UNII: 344S277G0Z) (ALLANTOIN - UNII:344S277G0Z) ALLANTOIN 0.1 mg in 1 mL GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 0.25 mg in 1 mL Inactive Ingredients Ingredient Name Strength SALICYLIC ACID (UNII: O414PZ4LPZ) 1 mg in 1 mL ETHYLHEXYL METHOXYCRYLENE (UNII: S3KFG6Q5X8) 6 mg in 1 mL WATER (UNII: 059QF0KO0R) 52.19 mg in 1 mL AZELAIC ACID (UNII: F2VW3D43YT) 15 mg in 1 mL PHENOXYETHANOL (UNII: HIE492ZZ3T) 0.9 mg in 1 mL ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) 0.1 mg in 1 mL MINERAL OIL (UNII: T5L8T28FGP) 13 mg in 1 mL CETYL ALCOHOL (UNII: 936JST6JCN) 5.4 mg in 1 mL KOJIC ACID (UNII: 6K23F1TT52) 1 mg in 1 mL CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) 0.5 mg in 1 mL LACTIC ACID (UNII: 33X04XA5AT) 0.5 mg in 1 mL SODIUM LACTATE (UNII: TU7HW0W0QT) 0.25 mg in 1 mL SORBITOL, L- (UNII: 01Q0586BG1) 0.25 mg in 1 mL TRIETHANOLAMINE LACTATE (UNII: 064C51N81O) 0.25 mg in 1 mL ARBUTIN (UNII: C5INA23HXF) 0.9 mg in 1 mL ALLANTOIN GLYCYRRHETINIC ACID (UNII: H6FL381368) 0.6 mg in 1 mL FYTIC ACID (UNII: 7IGF0S7R8I) 0.5 mg in 1 mL SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) 0.3 mg in 1 mL SODIUM LAURYL SULFATE (UNII: 368GB5141J) 0.3 mg in 1 mL ASCORBIC ACID (UNII: PQ6CK8PD0R) 0.25 mg in 1 mL SERINE (UNII: 452VLY9402) 0.25 mg in 1 mL HYDROXYETHYL UREA (UNII: MBQ7DDQ7AR) 0.25 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 0.05 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70663-007-02 30 in 1 BOX 12/17/2016 1 NDC:70663-007-01 30 mL in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 12/17/2016 Labeler - MESO SYSTEM S.A. (768263100) Registrant - MESO SYSTEM S.A. (768263100) Establishment Name Address ID/FEI Business Operations MESO SYSTEM S.A. 768263100 manufacture(70663-007)