Label: FIRMING DAY SPF 30- zinc oxide octinoxate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 54272-202-11 - Packager: CEN BEAUTY LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 4, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
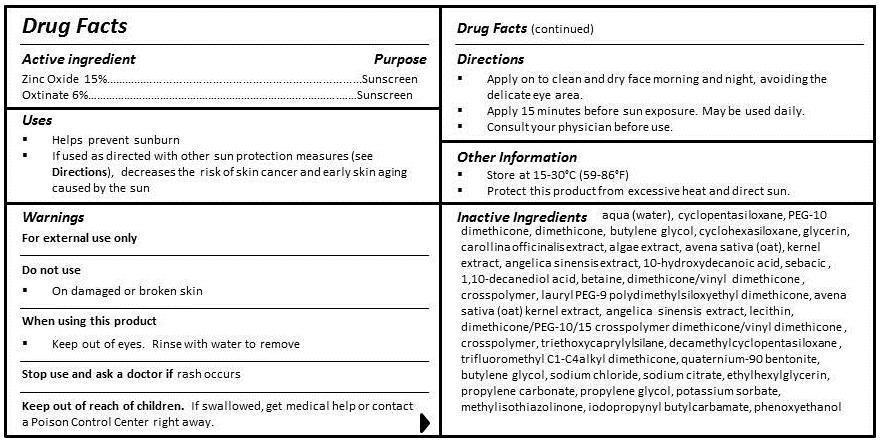
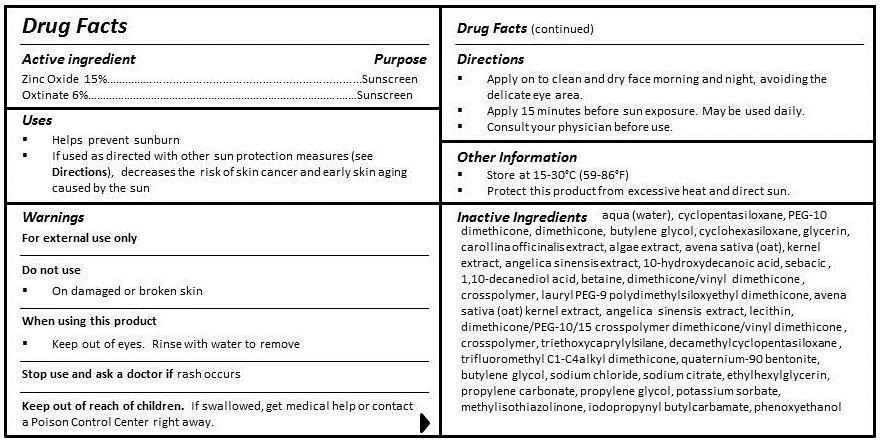
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
INGREDIENTS
aqua (water), cyclopentasiloxane, PEG-10 dimethicone, dimethicone, butylene glycol, cyclohexasiloxane, glycerin, carollina officinalis extract, algae extract, avena sativa (oat) kernel extract, angelica sinensis extract, 10-hydroxydecanoic acid, sebacic acid, 1,10-decanediol acid, betaine, dimethicone/vinyl dimethicone crosspolymer, lauryl PEG-9 polydimethylsiloxyethyl dimethicone, lecithin, dimethicone/PEG-10/15 crosspolymer, triethoxycaprylylsilane, decamethylcyclopentasiloxane, trifluoromethyl C1-C4 alkyl dimethicone, quaternium-90, bentonite, butylene glycol, sodium chloride, sodium citrate, ethylhexylglycerin, propylene carbonate, propylene glycol, potassium sorbate, methylisothiazolinone, iodopropynyl butylcarbamate, phenoxyethanol
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FIRMING DAY SPF 30
zinc oxide octinoxate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54272-202 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 15 g in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 6 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) DIMETHICONE (UNII: 92RU3N3Y1O) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CYCLOMETHICONE 6 (UNII: XHK3U310BA) GLYCERIN (UNII: PDC6A3C0OX) CORALLINA OFFICINALIS (UNII: 4004498D06) PORPHYRA UMBILICALIS (UNII: 14AN0J70WO) OAT (UNII: Z6J799EAJK) ANGELICA SINENSIS WHOLE (UNII: 697D19QDBN) 10-HYDROXYDECANOIC ACID (UNII: NP03XO416B) SEBACIC ACID (UNII: 97AN39ICTC) 1,10-DECANEDIOL (UNII: 5I577UDK52) BETAINE (UNII: 3SCV180C9W) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) PEG-9 POLYDIMETHYLSILOXYETHYL DIMETHICONE (UNII: TYP81E471F) DIMETHICONE PEG-10 PHOSPHATE (UNII: O7Q5NJ7X88) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) DODECAMETHYLPENTASILOXANE (UNII: 0QDQ2VQ5YJ) 2,2,2-TRIFLUOROACETOPHENONE (UNII: 6T7L1UPY09) QUATERNIUM-91 (UNII: 00J8H295NB) BENTONITE (UNII: A3N5ZCN45C) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) SODIUM CHLORIDE (UNII: 451W47IQ8X) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) SODIUM CITRATE (UNII: 1Q73Q2JULR) PROPYLENE CARBONATE (UNII: 8D08K3S51E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) PHENOXYETHANOL (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54272-202-11 30 mL in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 02/21/2013 Labeler - CEN BEAUTY LLC (078664118) Registrant - CEN BEAUTY LLC (078664118)