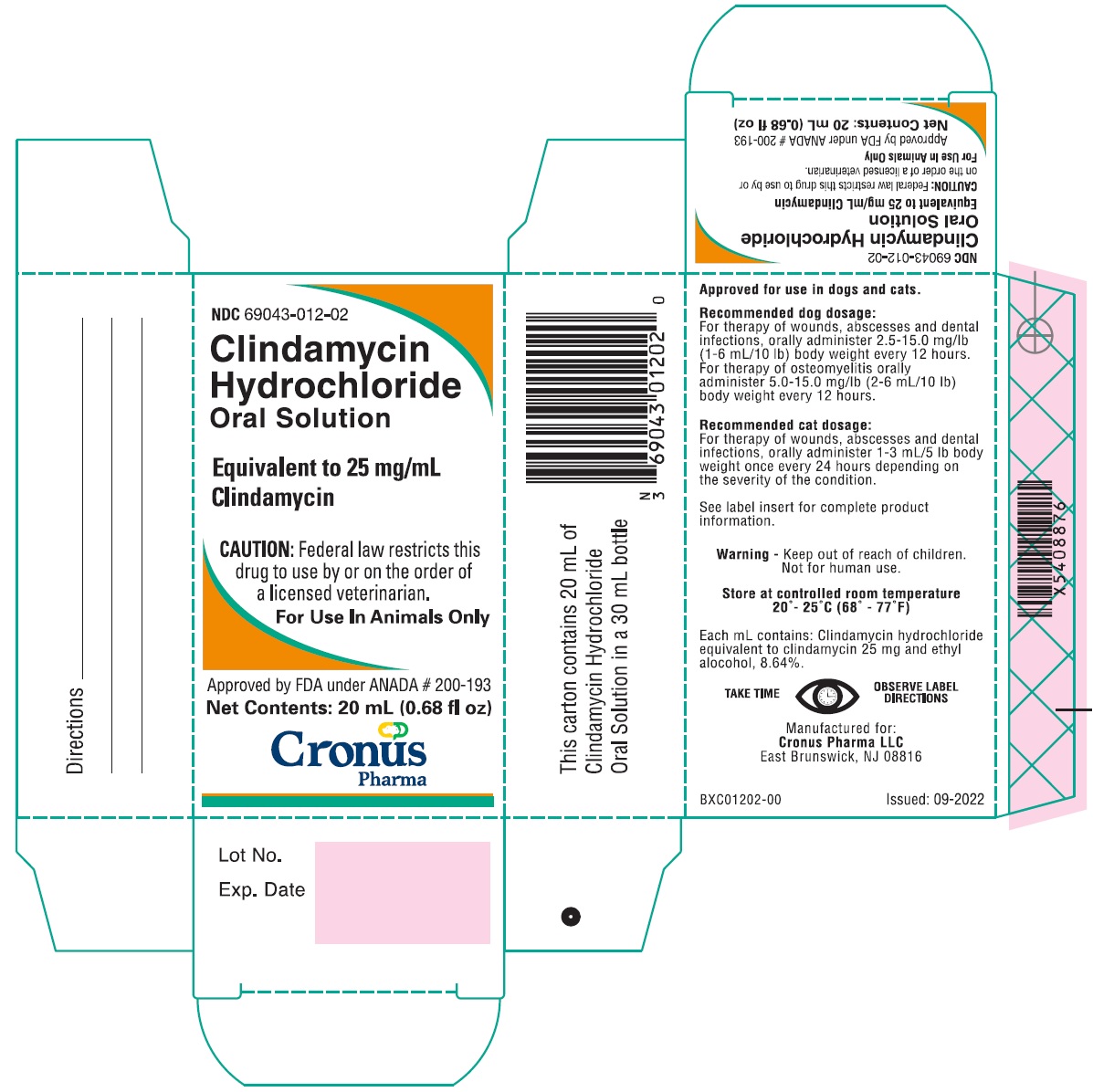
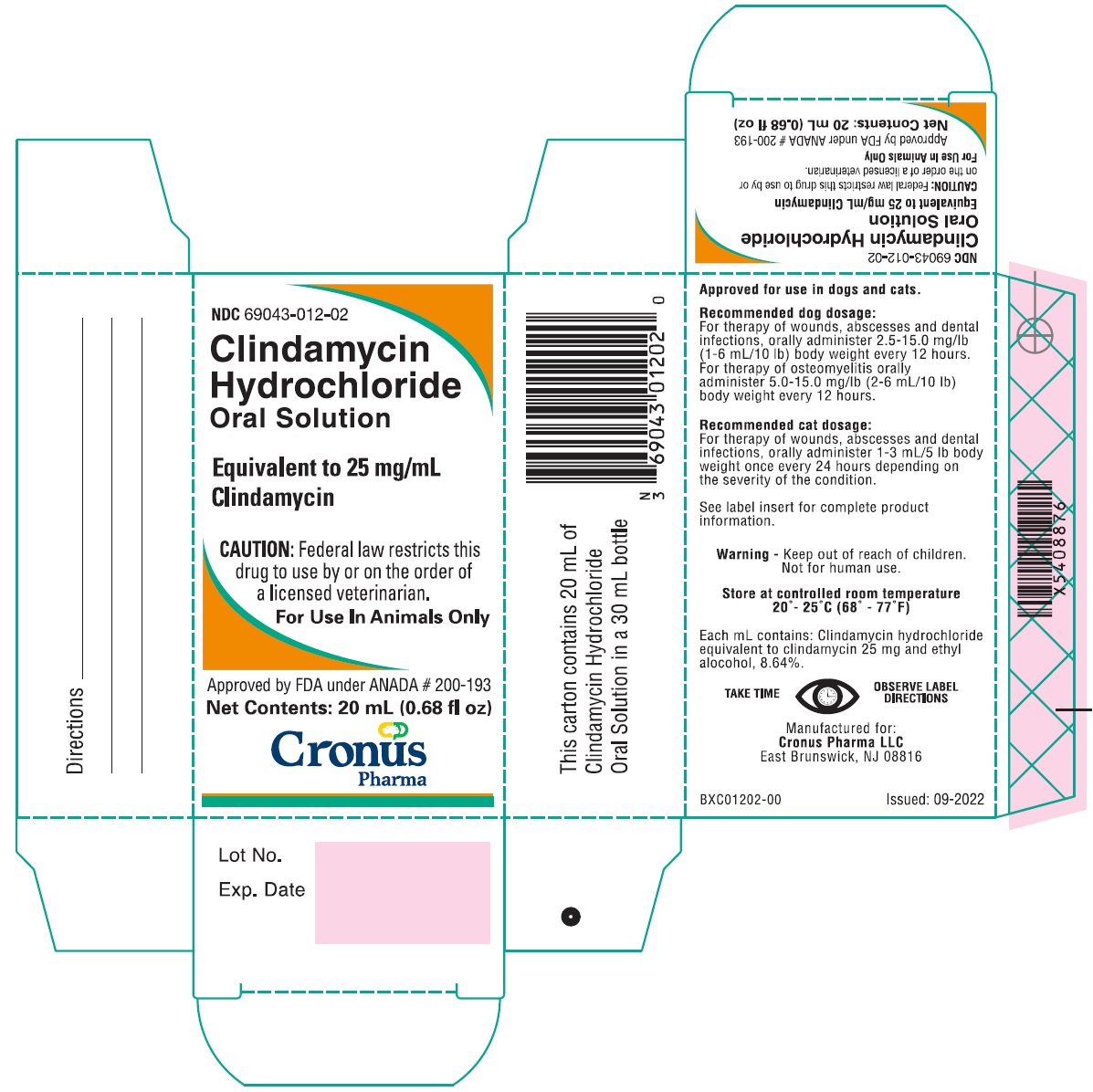
Label: CLINDAMYCIN HYDROCHLORIDE solution
- NDC Code(s): 69043-012-02
- Packager: Cronus Pharma LLC
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated December 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Clindamycin Hydrochloride Oral Solution contains clindamycin hydrochloride which is the hydrated salt of clindamycin. Clindamycin is a semisynthetic antibiotic produced by a 7(S)-chlorosubstitution of the 7(R)- hydroxyl group of a naturally produced antibiotic produced by Streptomyces lincolnensis var. lincolnensis.
Clindamycin Hydrochloride Oral Solution (for use in dogs and cats) is a palatable formulation intended for oral administration. Each mL of Clindamycin Hydrochloride Oral Solution contains clindamycin hydrochloride equivalent to 25 mg clindamycin; and ethyl alcohol, 8.64%.
-
ACTIONS
Site and Mode of Action: Clindamycin is an inhibitor of protein synthesis in the bacterial cell. The site of binding appears to be in the 50S sub-unit of the ribosome. Binding occurs to the soluble RNA fraction of certain ribosomes, thereby inhibiting the binding of amino acids to those ribosomes. Clindamycin differs from cell wall inhibitors in that it causes irreversible modification of the protein-synthesizing subcellular elements at the ribosomal level.
MICROBIOLOGY
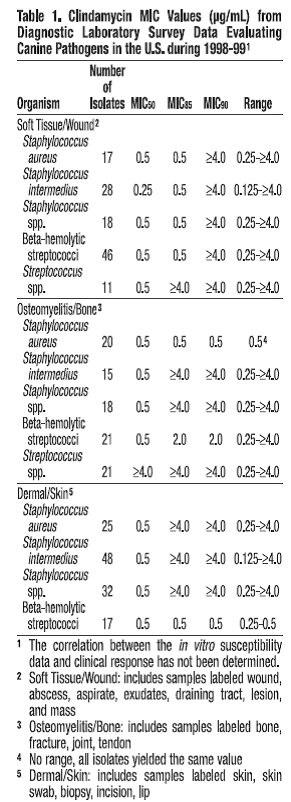
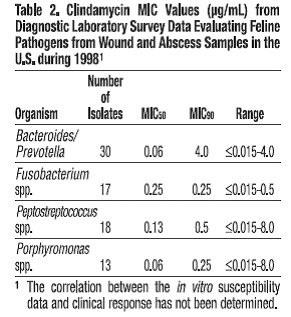
Clindamycin is a lincosaminide antimicrobial agent with activity against a wide variety of aerobic and anaerobic bacterial pathogens. Clindamycin is a bacteriostatic compound that inhibits bacterial protein synthesis by binding to the 50S ribosomal sub-unit. The minimum inhibitory concentrations (MICs) of Gram-positive and obligate anaerobic pathogens isolated from dogs and cats in the United States are presented in Table 1 and Table 2. Bacteria were isolated in 1998-1999. All MICs were performed in accordance with the National Committee for Clinical Laboratory Standards (NCCLS).
-
SPL UNCLASSIFIED SECTION
Absorption: Clindamycin hydrochloride is rapidly absorbed from the canine and feline gastrointestinal tract.
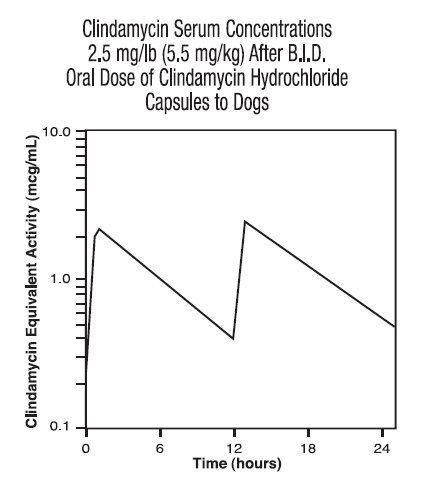
Dog Serum Levels: Serum levels at or above 0.5 μg/mL can be maintained by oral dosing at a rate of 2.5 mg/lb of clindamycin hydrochloride every 12 hours. This same study revealed that average peak serum concentrations of clindamycin occur 1 hour and 15 minutes after oral dosing. The elimination half-life for clindamycin in dog serum was approximately 5 hours. There was no bioactivity accumulation after a regimen of multiple oral doses in healthy dogs.
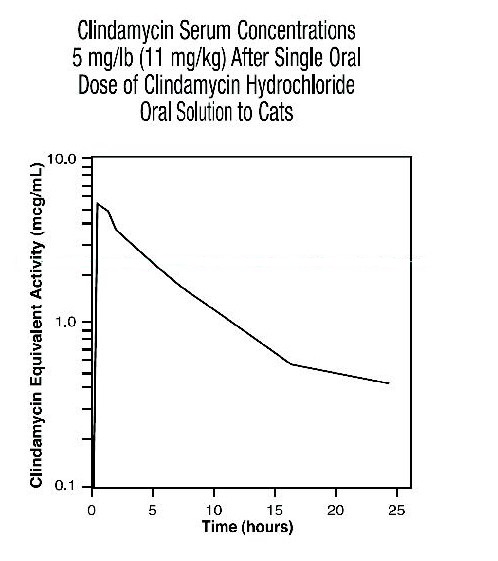
Cat Serum Levels: Serum levels at or above 0.5 μg/mL can be maintained by oral dosing at a rate of 5 mg/lb of clindamycin hydrochloride Solution every 24 hours. The average peak serum concentration of clindamycin occurs approximately 1 hour after oral dosing. The elimination half-life of clindamycin in feline serum is approximately 7.5 hours. In healthy cats, minimal accumulation occurs after multiple oral doses of clindamycin hydrochloride, and steady-state should be achieved by the third dose.
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
-
METABOLISM AND EXCRETION
Extensive studies of the metabolism and excretion of clindamycin hydrochloride administered orally in animals and humans have shown that unchanged drug and bioactive and bioinactive metabolites are excreted in urine and feces. Almost all of the bioactivity detected is serum after clindamycin hydrochloride product administration is due to the parent molecule (clindamycin). Urine bioactivity, however, reflects a mixture of clindamycin and active metabolites, especially N-demethyl clindamycin and clindamycin sulfoxide.
-
ANIMAL SAFETY SUMMARY
Rat and Dog Data: One year Oral toxicity studies in rats and dogs at doses of 30, 100 and 300 mg/kg/day (13.6, 45.5 and 136.4 mg/lb/day) have shown clindamycin hydrochloride to be well tolerated. Differences did not occur in the parameters evaluated to assess toxicity when comparing groups of treated animals with contemporary controls. Rats administered clindamycin hydrochloride at 600 mg/kg/day (272.7 mg/lb/day) for six months tolerated the drug well; however, dogs orally dosed at 600 mg/kg/day (272.7 mg/lb/day) vomited, had anorexia and subsequently lost weight. At necropsy these dogs had erosive gastritis and focal areas of necrosis of the mucosa of the gall bladder.
Safety in gestating bitches or breeding males has not been established.
Cat Data: The recommended daily therapeutic dose range for Clindamycin Hydrochloride Oral Solution is 11 to 33 mg/kg/day (5 to 15 mg/lb/day) depending on the severity of the condition. Clindamycin hydrochloride Solution was tolerated with little evidence of toxicity in domestic shorthair cats when administered orally at 10x the minimum recommended therapeutic daily dose (11 mg/kg 5 mg/lb) for 15 days, and at doses up to 5x the minimum recommended therapeutic dose for 42 days. Gastrointestinal tract upset (soft feces to diarrhea) occurred in control and treated cats with emesis occurring at doses 3x or greater than the minimum recommended therapeutic dose (11 mg/kg/day; 5 mg/lb/day). Lymphocytic inflammation of the gallbladder was noted in a greater number of treated cats at the 110 mg/kg/day (50 mg/lb/day) dose level than for control cats. No other effects were noted. Safety in gestating queens or breeding male cats has not been established.
-
INDICATIONS
Clindamycin Hydrochloride Oral Solution (for use in dogs and cats) is indicated for the treatment of infections caused by susceptible strains of the designated microorganisms in the specific conditions listed below:
Dogs: Skin infections (wounds and abscesses) due to coagulase positive staphylococci (Staphylococcus aureus or Staphylococcus intermedius). Deep wounds and abscesses due to Bacteroides fragilis, Prevotella melaninogenicus, Fusobacterium necrophorum and Clostridium perfringens. Dental infections due to Staphylococcus aureus, Bacteroides fragilis, Prevotella melaninogenicus, Fusobacterium necrophorum and Clostridium perfringens. Osteomyelitis due to Staphylococcus aureus, Bacteroides fragilis, Prevotella melaninogenicus, Fusobacterium necrophorum and Clostridium perfringens.
Cats: Skin infections (wounds and abscesses) due to Staphylococcus aureus, Staphylococcus intermedius, Streptococcus spp. Deep wounds and abscesses due to Clostridium perfringens and Bacteroides fragilis. Dental infections due to Staphylococcus aureus, Staphylococcus intermedius, Streptococcus spp., Clostridium perfringens and Bacteroides fragilis.
-
CONTRAINDICATIONS
Clindamycin Hydrochloride Oral Solution is contraindicated in animals with a history of hypersensitivity to preparations containing clindamycin or lincomycin.
Because of potential adverse gastrointestinal effects, do not administer to rabbits, hamsters, guinea pigs, horses, chinchillas or ruminating animals.
- WARNINGS
-
PRECAUTIONS
During prolonged therapy of one month or greater, periodic liver and kidney function tests and blood counts should be performed.
The use of clindamycin hydrochloride occasionally results in overgrowth of non-susceptible organisms such as clostridia and yeasts. Therefore, the administration of clindamycin hydrochloride should be avoided in those species sensitive to the gastrointestinal effects of clindamycin (see CONTRAINDICATIONS). Should superinfections occur, appropriate measures should be taken as indicated by the clinical situation.
Patients with very severe renal disease and/or very severe hepatic disease accompanied by severe metabolic aberrations should be dosed with caution, and serum clindamycin levels monitored during high dose therapy.
Clindamycin hydrochloride has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, clindamycin hydrochloride should be used with caution in animals receiving such agents.
Safety in gestating bitches and queens or breeding male dogs and cats has not been established.
-
ADVERSE REACTIONS
Side effects occasionally observed in either clinical trials or during clinical use were vomiting and diarrhea.
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Cronus Pharma LLC at 1-844-227-6687 (1-844-2-CRONUS).
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at www.fda.gov/reportanimalae.
-
DOSAGE & ADMINISTRATION
Dogs: Infected Wounds, Abscesses and Dental Infections
Oral: 2.5-15.0 mg/lb body weight every 12 hours.
Duration: Treatment with clindamycin hydrochloride products may be continued up to a maximum of 28 days if clinical judgment indicates. Treatment of acute infections should not be continued for more than three or four days if no response to therapy is seen.
Dosage Schedule:Clindamycin Hydrochloride Oral Solution
Administer 1-6 mL/10 lb body weight every 12 hours.
Dogs: Osteomyelitis
Oral: 5.0-15.0 mg/lb body weight every 12 hours.
Duration: Treatment with Clindamycin Hydrochloride Oral Solution is recommended for a minimum of 28 days. Treatment should not be continued for longer than 28 days if no response to therapy is seen.
Dosage Schedule:
Clindamycin Hydrochloride Oral Solution
Administer 2-6 mL/10 lb body weight every 12 hours.
Cats: Infected Wounds, Abscesses and Dental InfectionsOral: 5.0-15.0 mg/lb body weight once every 24 hours depending on the severity of the condition.
Duration: Treatment with Clindamycin Hydrochloride Oral Solution may be continued up to a maximum of 14 days if clinical judgment indicates. Treatment of acute infections should not be continued for more than three to four days if no clinical response to therapy is seen.
Dosage Schedule:
Clindamycin Hydrochloride Oral Solution, to provide 5.0 mg/lb administer 1 mL/5 lb body weight once every 24 hours; to provide 15.0 mg/lb administer 3 mL/5 lb body weight once every 24 hours.
-
HOW SUPPLIED
Clindamycin Hydrochloride Oral Solution is available as 20 mL filled in 30 mL bottles (25 mg/mL) supplied in packers containing 12 cartoned bottles with direction labels and calibrated dosing droppers.
NDC 69043-012-02
Store at controlled room temperature 20°-25°C (68°-77°F).
Manufactured for:Cronus Pharma LLC
East Brunswick, NJ 08816
ELC01202-00Issued: 07-2021
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CLINDAMYCIN HYDROCHLORIDE
clindamycin hydrochloride solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:69043-012 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLINDAMYCIN HYDROCHLORIDE (UNII: T20OQ1YN1W) (CLINDAMYCIN - UNII:3U02EL437C) CLINDAMYCIN 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69043-012-02 20 mL in 1 BOTTLE, DROPPER Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200193 10/25/2016 Labeler - Cronus Pharma LLC (079421067) Establishment Name Address ID/FEI Business Operations Pegasus Laboratories, Inc. 108454760 ANALYSIS, LABEL, MANUFACTURE Establishment Name Address ID/FEI Business Operations Chongqing Carelife Pharmaceutical Co., Ltd 531132009 API MANUFACTURE