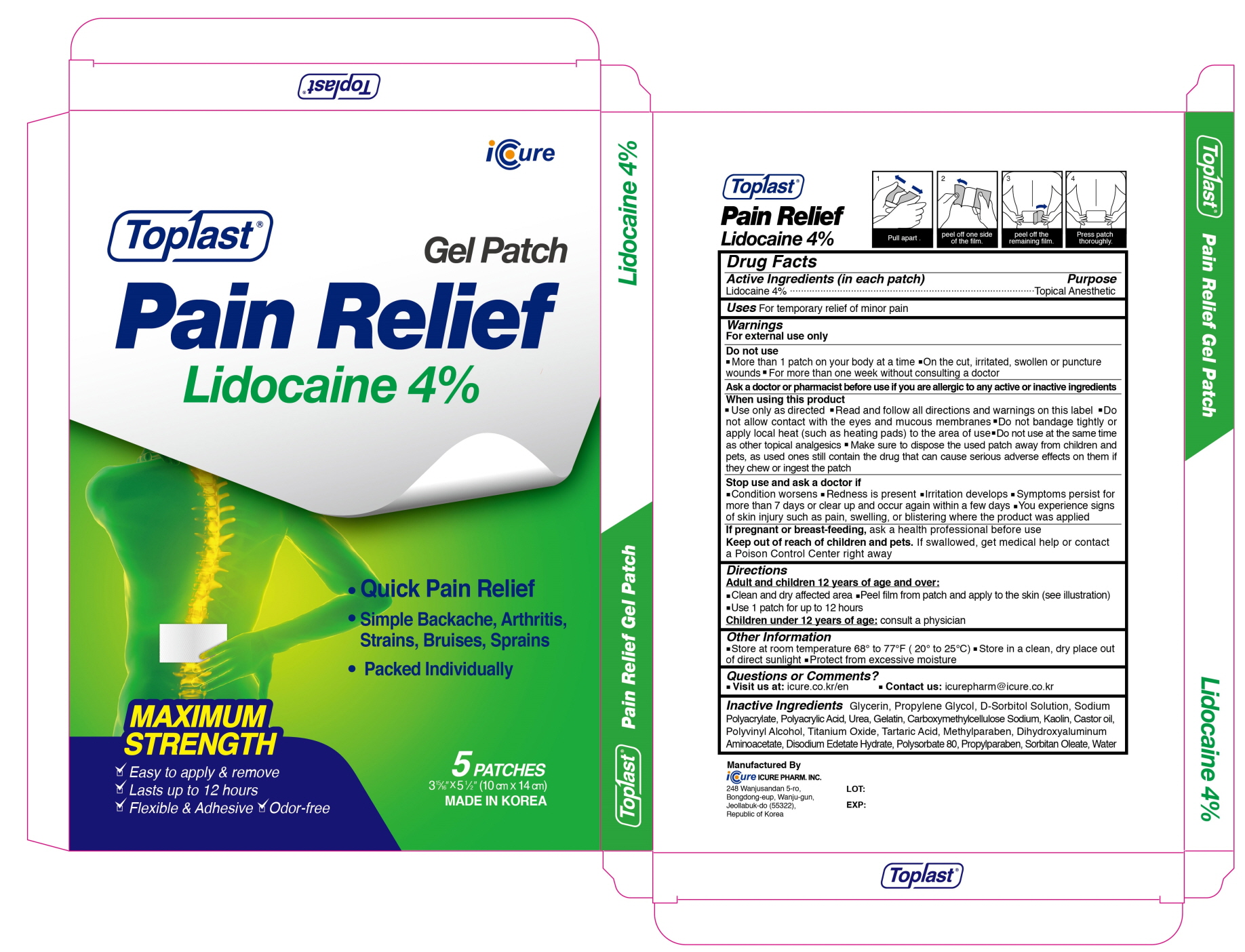
Label: TOPLAST PAIN RELIEF LIDOCAINE 4PERCENT- lidocaine patch
-
Contains inactivated NDC Code(s)
NDC Code(s): 73279-0005-1, 73279-0005-2 - Packager: Icure Pharmaceutical Inc, Wanju Factory
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 16, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
-
INACTIVE INGREDIENTS
Glycerin, Propylene Glycol, D-Sorbitol Solution, Sodium Polyacrylate, Polyacrylic Acid, Urea, Gelatin, Carboxymethylcellulose Sodium, Kaolin, Castor Oil, Polyvinyl Alcohol, Titanium Oxide, Tartaric Acid, Methylparaben, Dihydroxyaluminum Aminoacetate, Disodium Edetate Hydrate, Polysorbate 80, Propylparaben, Sorbitan Oleate, Water
- PURPOSE
-
WARNINGS
For external use only
--------------------------------------------------------------------------------------------------------
Do not use
■ More than 1 patch on your body at a time ■ On the cut, irritated, swollen or puncture wounds ■ For more than one week without consulting a doctor
--------------------------------------------------------------------------------------------------------
Ask a doctor or pharmacist before use if you are allergic to any active or inactive ingredients
--------------------------------------------------------------------------------------------------------
When using this product
■ Use only as directed ■ Read and follow all directions and warnings on this label ■ Do not allow contact with the eyes and mucous membranes ■ Do not bandage tightly or apply local heat (such as heating pads) to the area of use ■ Do not use at the same time as other topical analgesics ■ Make sure to dispose the used patch away from children and pets, as used ones still contain the drug that can cause serious adverse effects on them if they chew or ingest the patch
--------------------------------------------------------------------------------------------------------
Stop use and ask a doctor if
■ Condition worsens ■ Redness is present ■ Irritation develops ■ Symptoms persist for more than 7 days or clear up and occur again within a few days ■ You experience signs of skin injury such as pain, swelling or blistering where the product was applied
--------------------------------------------------------------------------------------------------------
If pregnant or breast-feeding, ask a health professional before use - Keep out of reach of children and pets
- Uses
- Directions
- Other Information
- Questions or Comments
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOPLAST PAIN RELIEF LIDOCAINE 4PERCENT
lidocaine patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73279-0005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Lidocaine (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) Lidocaine 240 mg Inactive Ingredients Ingredient Name Strength Glycerin (UNII: PDC6A3C0OX) Propylene Glycol (UNII: 6DC9Q167V3) SORBITOL (UNII: 506T60A25R) Urea (UNII: 8W8T17847W) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) Gelatin (UNII: 2G86QN327L) Kaolin (UNII: 24H4NWX5CO) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Tartaric Acid (UNII: W4888I119H) Methylparaben (UNII: A2I8C7HI9T) Dihydroxyaluminum Aminoacetate (UNII: DO250MG0W6) EDETATE DISODIUM (UNII: 7FLD91C86K) Polysorbate 80 (UNII: 6OZP39ZG8H) Propylparaben (UNII: Z8IX2SC1OH) SORBITAN MONOOLEATE (UNII: 06XEA2VD56) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73279-0005-2 5 in 1 CARTON 07/01/2020 1 NDC:73279-0005-1 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/01/2020 Labeler - Icure Pharmaceutical Inc, Wanju Factory (695687612) Registrant - Icure Pharmaceutical Inc, Wanju Factory (695687612) Establishment Name Address ID/FEI Business Operations Icure Pharmaceutical Inc, Wanju Factory 695687612 manufacture(73279-0005)