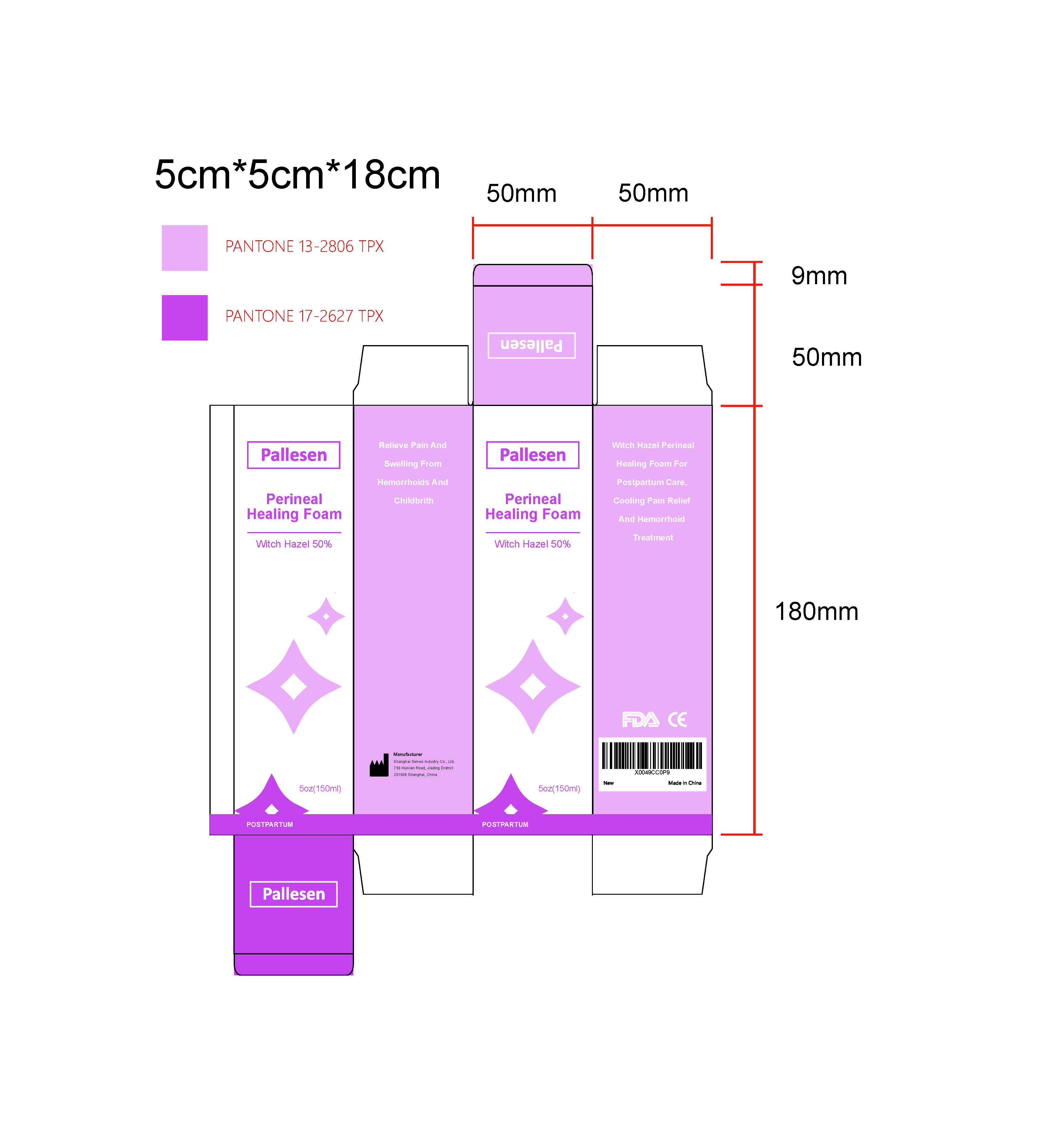
Label: HAZEL PERINEAL HEALING FOAM- witch hazel liquid
- NDC Code(s): 84506-001-01, 84506-001-02
- Packager: SHANGHAI SENWO INDUSTRY CO., LTD.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated July 4, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- WARNINGS
- DOSAGE & ADMINISTRATION
- STORAGE AND HANDLING
-
INACTIVE INGREDIENT
Water
C12-18 ALKYL GLUCOSIDE
SODIUM LAUROYL SARCOSINATE
SODIUM LAUROYL GLUTAMATE
COCAMIDOPROPYL BETAINE
ERYTHRITOL
BUTYLENE GLYCOL
ALCOHOL
HYDROXYACETOPHENONE
1,2-HEXANEDIOL
PHENOXYETHANOL
TOCOPHEROL
SAURURUS CHINENSIS LEAF/ROOT
EXTRACT
TARAXACUM OFFICINALE
(DANDELION) LEAF EXTRACT
STEPHANIA TETRANDRA EXTRACT
MENTHA HAPLOCALYX OIL
CITRIC ACID
PEG-40 HYDROGENATED CASTOR OIL - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HAZEL PERINEAL HEALING FOAM
witch hazel liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84506-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WITCH HAZEL (UNII: 101I4J0U34) (WITCH HAZEL - UNII:101I4J0U34) WITCH HAZEL 50 g in 100 mL Inactive Ingredients Ingredient Name Strength COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) TARAXACUM OFFICINALE (UNII: 39981FM375) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) POLYOXYL 40 HYDROGENATED CASTOR OIL (UNII: 7YC686GQ8F) WATER (UNII: 059QF0KO0R) SODIUM LAUROYL GLUTAMATE (UNII: NCX1UU2D33) ERYTHRITOL (UNII: RA96B954X6) ALCOHOL (UNII: 3K9958V90M) STEPHANIA TETRANDRA WHOLE (UNII: 7S7TV2F4XC) SODIUM LAUROYL SARCOSINATE (UNII: 632GS99618) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PEPPERMINT OIL (UNII: AV092KU4JH) PHENOXYETHANOL (UNII: HIE492ZZ3T) TOCOPHEROL (UNII: R0ZB2556P8) SAURURUS CHINENSIS WHOLE (UNII: 6DRV3D37XS) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84506-001-01 100 mL in 1 BOTTLE; Type 0: Not a Combination Product 07/04/2024 2 NDC:84506-001-02 150 mL in 1 BOTTLE; Type 0: Not a Combination Product 07/04/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M015 07/04/2024 Labeler - SHANGHAI SENWO INDUSTRY CO., LTD. (421273994) Establishment Name Address ID/FEI Business Operations SHANGHAI SENWO INDUSTRY CO., LTD. 421273994 manufacture(84506-001)