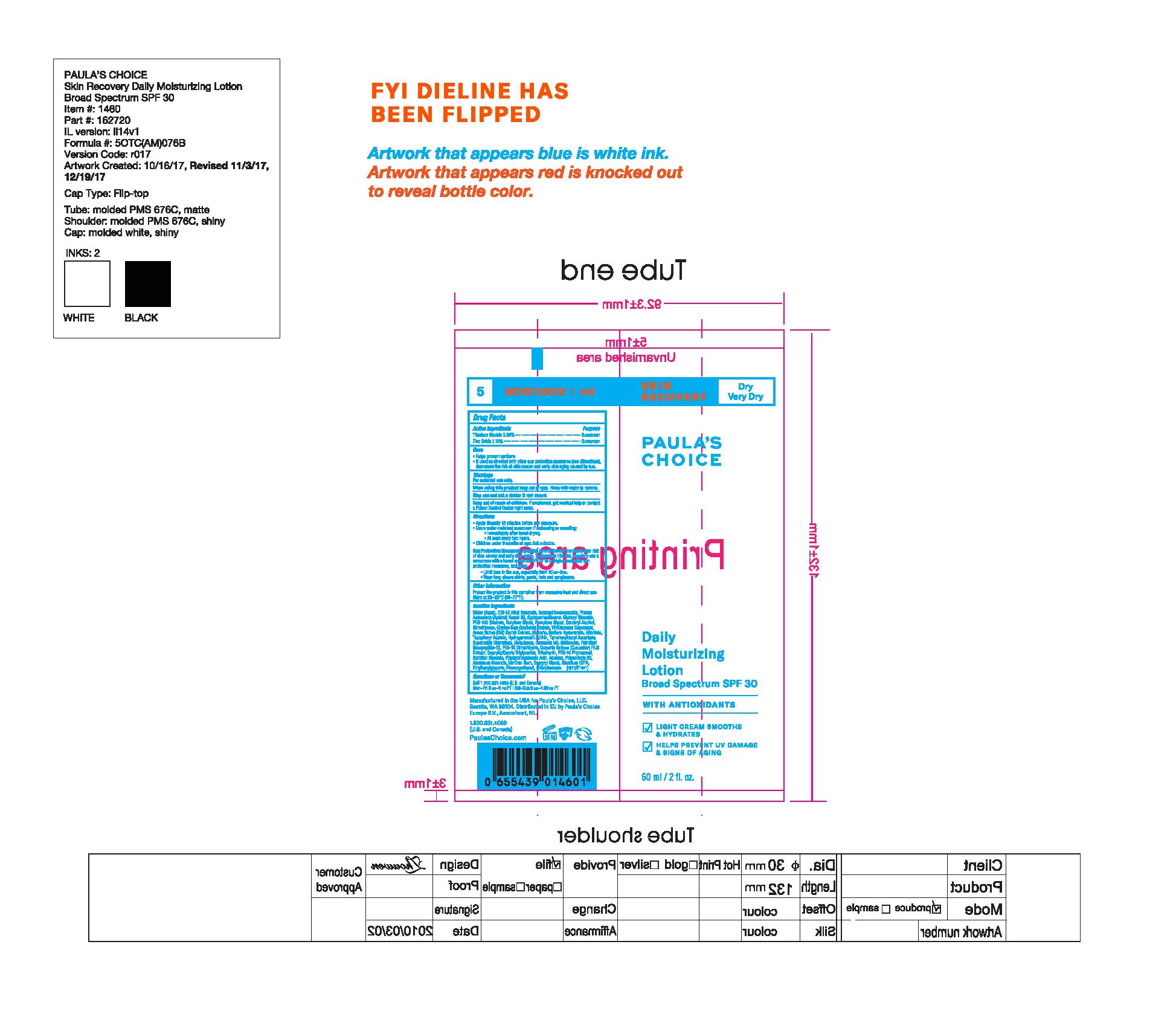
Label: PAULAS CHOICE SKIN RECOVERY DAILY MOISTURIZING SPF 30- titanium dioxide, zinc oxide lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 76144-146-01 - Packager: Paula's Choice
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 6, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PURPOSE
-
INSTRUCTIONS FOR USE
Apply liberally 15 minutes before sun exposure. Use a water-resistant sunscreen if swimming or sweating. Reapply at least every two hours if skin is exposed to sun. Children under 6 months of age: ask a doctor.
Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 25 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m.-2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses. - WARNINGS
- ASK DOCTOR
- WHEN USING
- KEEP OUT OF REACH OF CHILDREN
- STOP USE
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Water, C12-15 Alkyl Benzoate, Isononyl Isononanoate, Prunus Armeniaca (Apricot) Kernel Oil, Cylcopentasiloxane, Glyceryl Stearate, PEG-100 Stearate, Butylene Glycol, Pentylene Glycol, Cetearyl Alcohol, Dimethicone, Glycine Soja (Soybean) Sterols, VP/Eicosene Copolymer, Superoxide Dismutase, Tocopheryl Acetate, Tetrahexyldecyl Ascorbate, Ubiquinone, Ceramide 2, Sodium Hyaluronate, Allantoin, Avena Sativa (Oat) Kernel Extract, PEG-10 Dimethicone, Palmitoyl Oligopeptide, Tribehenin, Hydrogenated Lecithin, Polysorbate 20, Glycerin,PEG-10 Rapeseed Sterol, Caprylic/Capric Triglyceride, Methicone, Cucumis Sativus (Cucumber) Fruit Extract, Alumina, Aluminum Stearate, Polyhydroxystearic Acid, Xanthan Gum, Sorbitan Stearate, Caprylyl Glycol, Disodium EDTA, Ethylhexylglycerin, Phenoxyethanol, Chlorphenesin.
- ACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PAULAS CHOICE SKIN RECOVERY DAILY MOISTURIZING SPF 30
titanium dioxide, zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76144-146 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 38.5 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 31.2 mg in 1 mL Inactive Ingredients Ingredient Name Strength OAT (UNII: Z6J799EAJK) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) APRICOT KERNEL OIL (UNII: 54JB35T06A) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) PENTYLENE GLYCOL (UNII: 50C1307PZG) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) DIMETHICONE (UNII: 92RU3N3Y1O) SUPEROXIDE DISMUTASE (SACCHAROMYCES CEREVISIAE) (UNII: W2T4YRA9AD) .ALPHA.-TOCOPHEROL ACETATE, DL- (UNII: WR1WPI7EW8) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) UBIDECARENONE (UNII: EJ27X76M46) CERAMIDE 2 (UNII: C04977SRJ5) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ALLANTOIN (UNII: 344S277G0Z) PALMITOYL HEXAPEPTIDE-12 (UNII: HO4ZT5S86C) TRIBEHENIN (UNII: 8OC9U7TQZ0) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) POLYSORBATE 20 (UNII: 7T1F30V5YH) GLYCERIN (UNII: PDC6A3C0OX) PEG-10 RAPESEED STEROL (UNII: 258O76T85M) ALUMINUM STEARATE (UNII: U6XF9NP8HM) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) EDETATE DISODIUM (UNII: 7FLD91C86K) CHLORPHENESIN (UNII: I670DAL4SZ) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHICONE (20 CST) (UNII: 6777U11MKT) PEG-100 STEARATE (UNII: YD01N1999R) SOY STEROL (UNII: PL360EPO9J) PEG-10 DIMETHICONE (600 CST) (UNII: 8PR7V1SVM0) WATER (UNII: 059QF0KO0R) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ISONONYL ISONONANOATE (UNII: S4V5BS6GCX) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) CUCUMBER (UNII: YY7C30VXJT) ALUMINUM OXIDE (UNII: LMI26O6933) XANTHAN GUM (UNII: TTV12P4NEE) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) CAPRYLYL GLYCOL (UNII: 00YIU5438U) EICOSYL POVIDONE (UNII: XQQ9MKE2BJ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76144-146-01 60 mL in 1 TUBE; Type 0: Not a Combination Product 06/27/2011 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 06/27/2011 Labeler - Paula's Choice (029583981)