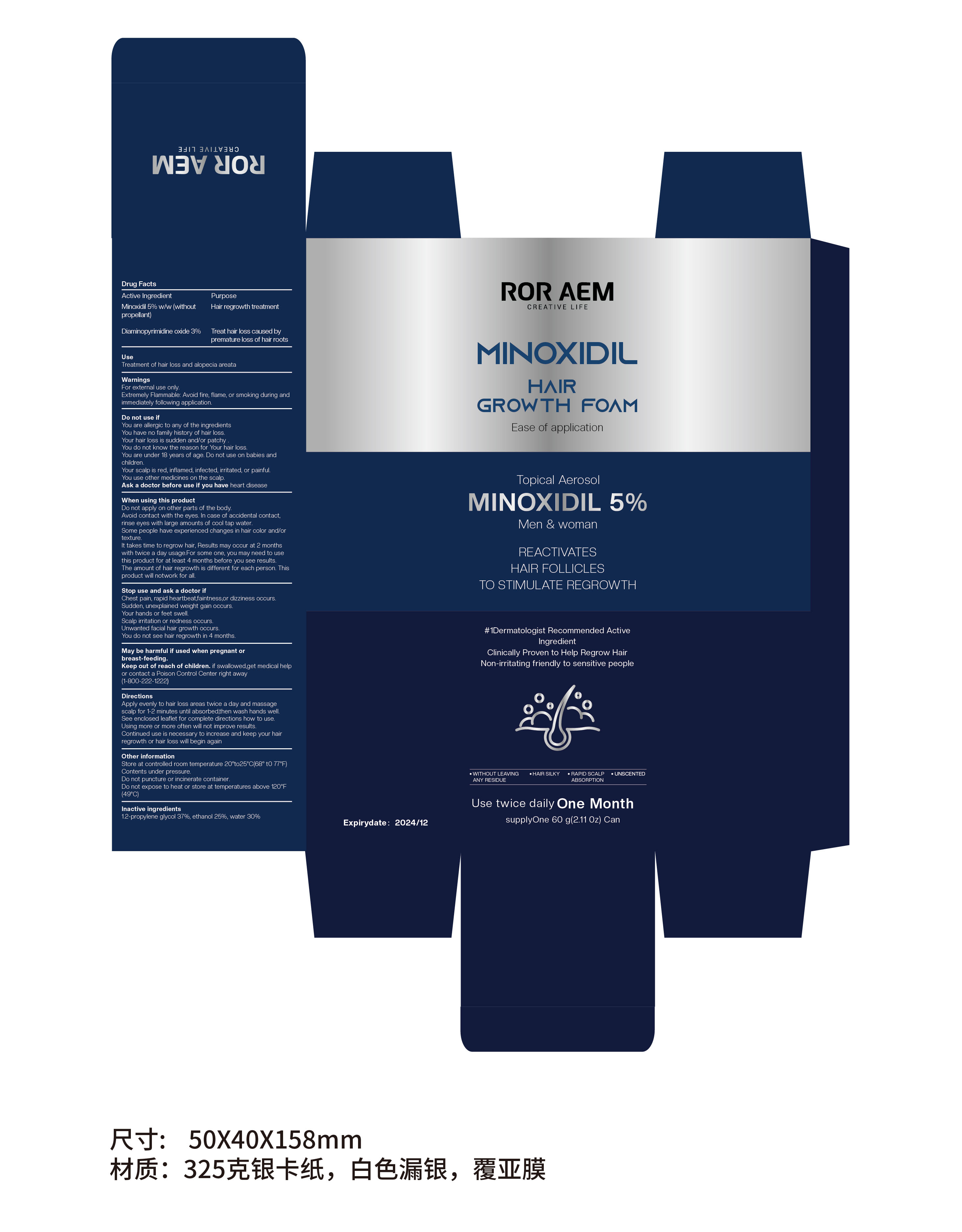
Label: MINOXIDIL HAIR GROWTH FOAM aerosol, foam
- NDC Code(s): 84315-001-01
- Packager: Guangzhou Yibai Technology Sarl
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated May 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
-
DO NOT USE
You are allergic to any of the ingredients
You have no family history of hair loss.
Your hair loss is sudden and/or patchy.
You do not know the reason for Your hair loss.
You are under 18 years of age. Do not use on babies and children.
Your scalp is red, inflamed, infected, irritated, or painful.
You use other medicines on the scalp.
Ask a doctor before use if you have heart disease
-
WHEN USING
Do not apply on other parts of the body.
Avoid contact with the eyes. In case of accidental contact, rinse eyes with large amounts of cool tap water.
Some people have experienced changes in hair color and/or texture.
It takes time to regrow hair, Results may occur at 2 months with twice a day usage.For some one, you may need to use this product for at least 4 months before you see results.The amount of hair regrowth is different for each person. This product will notwork for all.
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Apply evenly to hair loss areas twice a day and massage scalp for 1-2 minutes until absorbed,then wash hands well.See enclosed leaflet for complete directions how to use.Using more or more often will not improve results.Continued use is necessary to increase and keep your hair regrowth or hair loss will begin again
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MINOXIDIL HAIR GROWTH FOAM
minoxidil hair growth foam aerosol, foamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:84315-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIAMINOPYRIMIDINE OXIDE (UNII: 1756681479) (DIAMINOPYRIMIDINE OXIDE - UNII:1756681479) DIAMINOPYRIMIDINE OXIDE 3 g in 100 g MINOXIDIL (UNII: 5965120SH1) (MINOXIDIL - UNII:5965120SH1) MINOXIDIL 5 g in 100 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:84315-001-01 60 g in 1 CANISTER; Type 0: Not a Combination Product 05/09/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA075357 05/09/2024 Labeler - Guangzhou Yibai Technology Sarl (281523247) Establishment Name Address ID/FEI Business Operations Guangzhou Yibai Technology Sarl 281523247 manufacture(84315-001)