Label: HUEY DAILY MINERAL- titanium dioxide, zinc oxide lotion
- NDC Code(s): 83599-391-01, 83599-391-02
- Packager: Huey
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 10, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
- Huey Daily Mineral
-
INGREDIENTS AND APPEARANCE
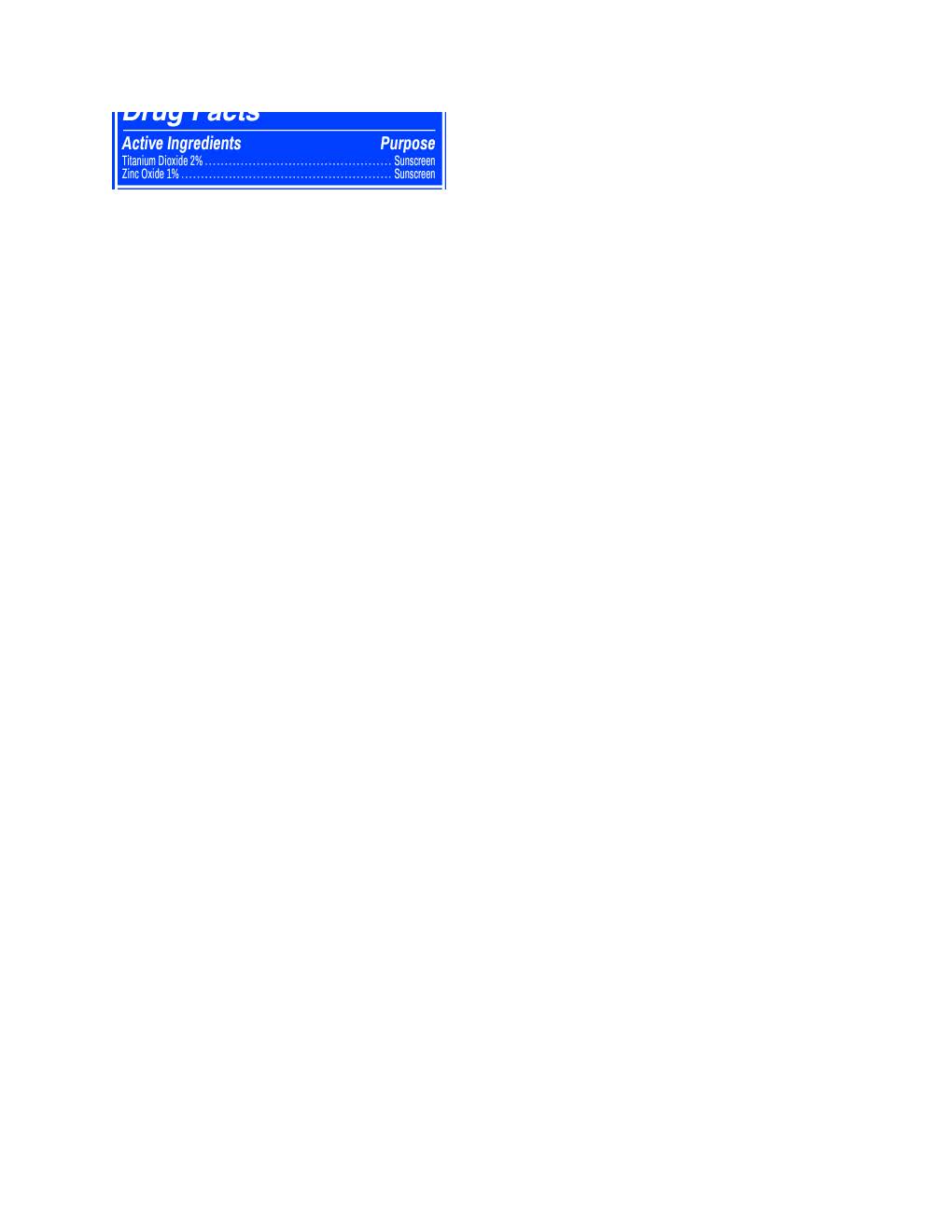
HUEY DAILY MINERAL
titanium dioxide, zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83599-391 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 9.7 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 19.4 mg in 1 mL Inactive Ingredients Ingredient Name Strength BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) POLYESTER-7 (UNII: 0841698D2F) GLYCERIN (UNII: PDC6A3C0OX) NEOPENTYL GLYCOL DIHEPTANOATE (UNII: 5LKW3C543X) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) ALLANTOIN (UNII: 344S277G0Z) CAPRYLYL GLYCOL (UNII: 00YIU5438U) AVENA SATIVA WHOLE (UNII: 5P8D0Z74RG) XANTHAN GUM (UNII: TTV12P4NEE) DICRATERIA ROTUNDA OIL (UNII: Z0DWU9R4GM) DISODIUM EDTA-COPPER (UNII: 6V475AX06U) CAMELLIA SINENSIS WHOLE (UNII: C5M4585ZBZ) TRICETEARETH-4 PHOSPHATE (UNII: 69534Y66NO) PULLULAN (UNII: 8ZQ0AYU1TT) STEARETH-20 (UNII: L0Q8IK9E08) HEXYLENE GLYCOL (UNII: KEH0A3F75J) TRILAURETH-4 PHOSPHATE (UNII: M96W2OLL2V) SODIUM ASCORBATE (UNII: S033EH8359) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) CETYL ALCOHOL (UNII: 936JST6JCN) SQUALANE (UNII: GW89575KF9) PEG-75 STEARATE (UNII: OT38R0N74H) PHENOXYETHANOL (UNII: HIE492ZZ3T) .ALPHA.-BISABOLOL, (+)- (UNII: 105S6I733Z) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) DIMETHICONOL/PROPYLSILSESQUIOXANE/SILICATE CROSSPOLYMER (450000000 MW) (UNII: 9KB5R958PB) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) ETHYLHEXYL ISONONANOATE (UNII: I6KB4GE3K4) CETETH-20 (UNII: I835H2IHHX) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) TOCOPHEROL (UNII: R0ZB2556P8) RUTTNERA LAMELLOSA OIL (UNII: 5XZ38R4SUT) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83599-391-02 1 in 1 CARTON 02/09/2024 1 NDC:83599-391-01 60 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 02/09/2024 Labeler - Huey (118333758) Registrant - Nanophase Technologies Corporation (623502044) Establishment Name Address ID/FEI Business Operations Nanophase Technologies Corporation 118812921 manufacture(83599-391) , pack(83599-391)