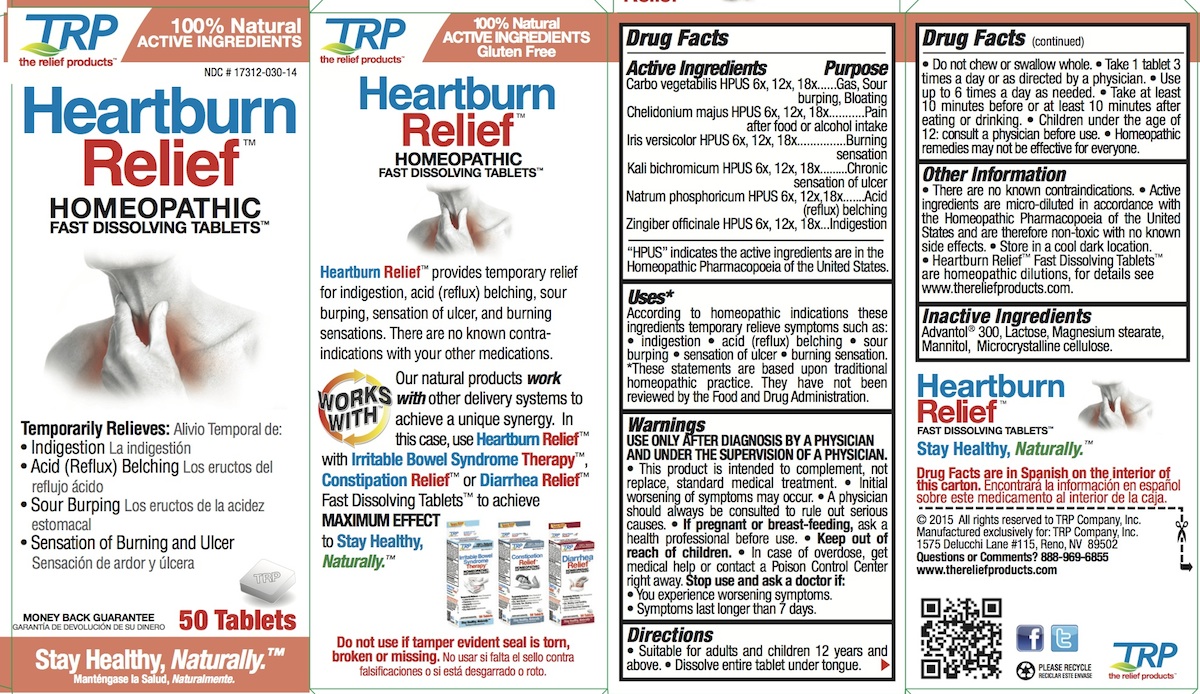
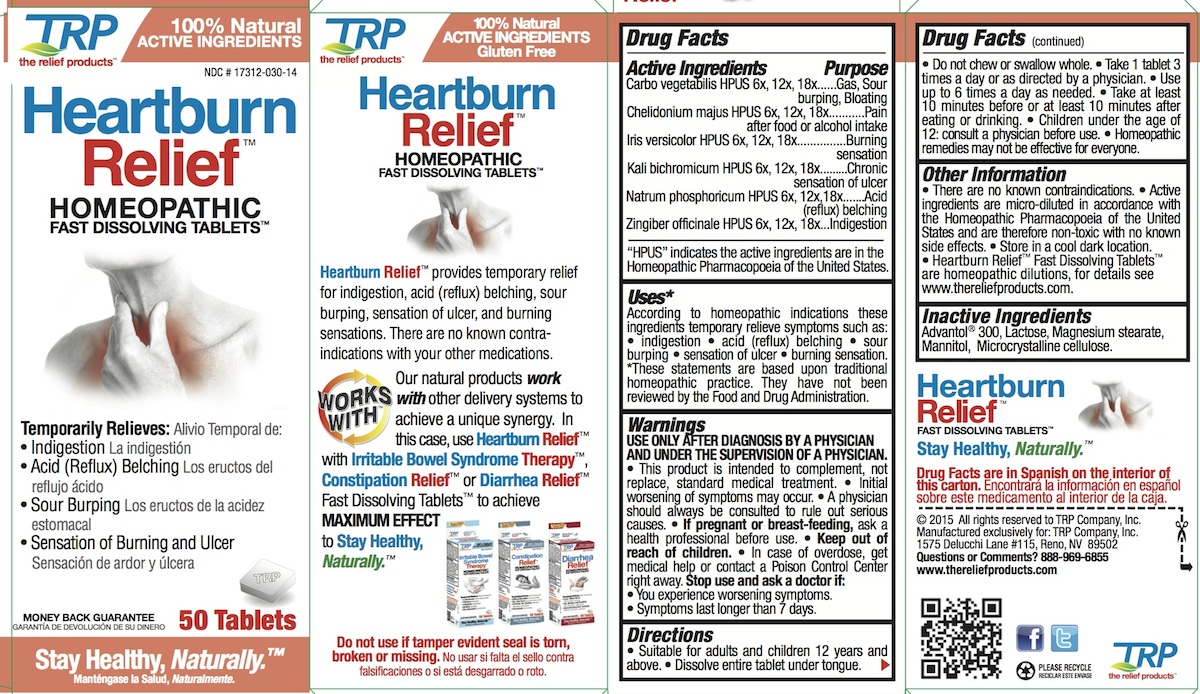
Label: HEARTBURN RELIEF- carbo more tablet, orally disintegrating
-
Contains inactivated NDC Code(s)
NDC Code(s): 17312-030-14 - Packager: TRP Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 11, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PURPOSE
-
INDICATIONS & USAGE
Uses:*
According to homeopathic indications these ingredients temporarily relieve symptoms such as: • indigestion • acid (reflux) belching • sour burping • sensation of ulcer • burning sensation. * These statements are based upon traditional homeopathic practice. They have not been reviewed by the Food and Drug Administration.
- WARNINGS
- PREGNANCY OR BREAST FEEDING
- Keep out of reach of children
- WARNINGS
-
Directions
- Suitable for adults and children 12 years and above.
- Dissolve entire tablet under tongue.
- Do not chew or swallow whole.
- Take 1 tablet 3 times a day or as directed by a physician.
- Use up to 6 times a day as needed.
- Take at least 10 minutes before or at least 10 minutes after eating or drinking.
- Children under the age of 12: consult a physician before use.
Homeopathic remedies may not be effective for everyone.
- Other information
- Inactive Ingredients
- Do not use if tamper evident seal is torn, broken or missing.
- QUESTIONS
-
Active Ingredients
Active Ingredients
Carbo vegetabilis HPUS 6x, 12x, 18x
Chelidonium majus HPUS 6x, 12x, 18x
Iris versicolor HPUS 6x, 12x, 18x
Kali bichromicum HPUS 6x, 12x, 18x
Natrum phosphoricum HPUS 6x, 12x,18x
Zingiber officinale HPUS 6x, 12x, 18x"HPUS" indicates the active ingredients are in the Homeopathic Pharmacopoeia of the United States.
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HEARTBURN RELIEF
carbo more tablet, orally disintegratingProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:17312-030 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACTIVATED CHARCOAL (UNII: 2P3VWU3H10) (ACTIVATED CHARCOAL - UNII:2P3VWU3H10) ACTIVATED CHARCOAL 6 [hp_X] POTASSIUM DICHROMATE (UNII: T4423S18FM) (DICHROMATE ION - UNII:9LKY4BFN2V) POTASSIUM DICHROMATE 6 [hp_X] CHELIDONIUM MAJUS (UNII: 7E889U5RNN) (CHELIDONIUM MAJUS - UNII:7E889U5RNN) CHELIDONIUM MAJUS 6 [hp_X] SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE (UNII: 70WT22SF4B) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM PHOSPHATE, DIBASIC, HEPTAHYDRATE 6 [hp_X] GINGER (UNII: C5529G5JPQ) (GINGER - UNII:C5529G5JPQ) GINGER 6 [hp_X] IRIS VERSICOLOR ROOT (UNII: X43D4L3DQC) (IRIS VERSICOLOR ROOT - UNII:X43D4L3DQC) IRIS VERSICOLOR ROOT 6 [hp_X] Inactive Ingredients Ingredient Name Strength MANNITOL (UNII: 3OWL53L36A) SORBITOL (UNII: 506T60A25R) CROSPOVIDONE (UNII: 68401960MK) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) COPOVIDONE (UNII: D9C330MD8B) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color white Score no score Shape DIAMOND Size 13mm Flavor Imprint Code TRP Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17312-030-14 1 in 1 PACKAGE 04/15/2015 1 50 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 04/15/2015 Labeler - TRP Company (105185719)