Label: NEOMED 325- neomycin sulfate powder, for solution
- NDC Code(s): 61133-5442-2, 61133-5442-3, 61133-5442-4
- Packager: Bimeda, Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated March 28, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
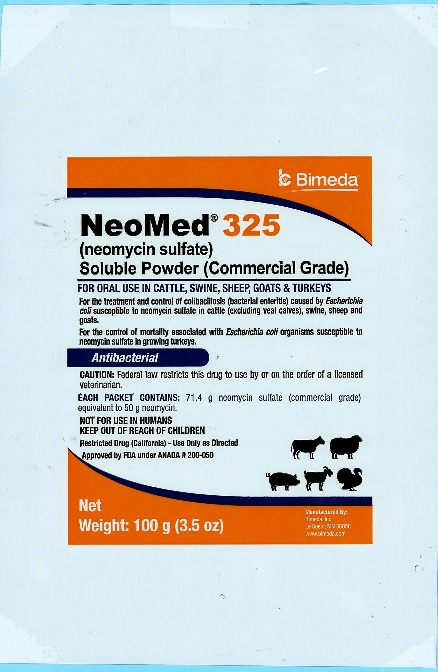
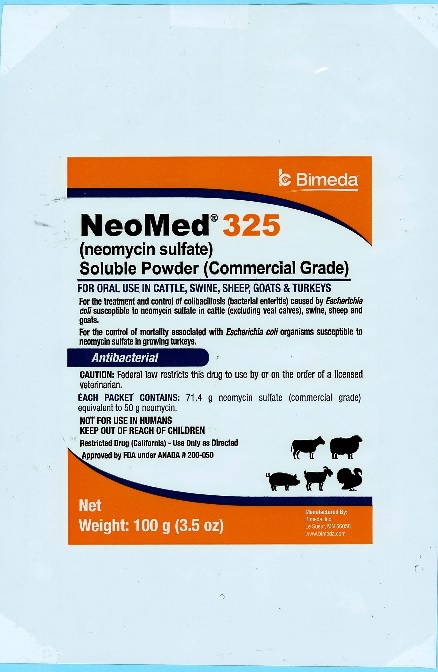
100 g pouch
NeoMed® 325
(neomycin sulfate)
Soluble Powder (Commercial Grade)
FOR ORAL USE IN CATTLE, SWINE, SHEEP, GOATS & TURKEYSFor the treatment and control of colibacillosis (bacterial enteritis) caused by Escherichia coli susceptible to neomycin sulfate in cattle (excluding veal calves), swine, sheep and goats.
For the control of mortality associated with Escherichia coli organisms susceptible to neomycin sulfate in growing turkeys.
AntibacterialCAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Each Packet Contains: 71.4 g neomycin sulfate (commercial grade) equivalent to 50 g neomycin.
NOT FOR USE IN HUMANS
KEEP OUT OF REACH OF CHILDREN
Restricted Drug (California)- Use Only as DirectedApproved by FDA under ANADA # 200-050
NET WEIGHT:
100 g (3.5 oz) -
100 g pouch
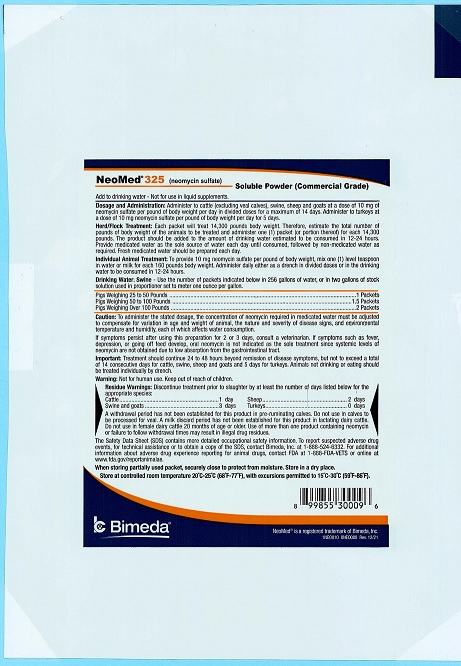
Add to drinking water - Not for use in liquid supplements.
DOSAGE AND ADMINISTRATION: Administer to cattle (excluding veal calves), swine, sheep and goats at a dose of 10 mg of neomycin sulfate per pound of body weight per day in divided doses for a maximum of 14 days. Administer to turkeys at a dose of 10 mg neomycin sulfate per pound of body weight per day for 5 days.
Herd/Flock Treatment: Each packet will treat 7,150 pounds body weight. Therefore, estimate the total number of pounds of body weight of the animals to be treated and administer one (1) packet (or portion thereof) for each 7,150 pounds. The product should be added to the amount of drinking water estimated to be consumed in 12-24 hours. Provide medicated water as the sole source of water each day until consumed, followed by non-medicated water as required. Fresh medicated water should be prepared each day.
Individual Animal Treatment: To provide 10 mg neomycin sulfate per pound of body weight, mix one (1) level teaspoon in water or milk for each 160 pounds of body weight. Administer daily either as a drench in divided doses or in the drinking water to be consumed in 12-24 hours.
Drinking Water: Swine Use the number of packets indicated below in 256 gallons of water, or in two gallons of stock solution used in proportioner set to meter one ounce per gallon.
Pigs Weighing 25 to 50 Pounds 2 Packets Pigs Weighing 50 to 100 Pounds 3 Packets Pigs Weighing Over 100 Pounds 4 Packets -
200 g Pouch
NeoMed® 325
(neomycin sulfate)
Soluble Powder (Commercial Grade)
FOR ORAL USE IN CATTLE, SWINE, SHEEP, GOATS & TURKEYSFor the treatment and control of colibacillosis (bacterial enteritis) caused by Escherichia coli susceptible to neomycin sulfate in cattle (excluding veal calves), swine, sheep and goats.
For the control of mortality associated with Escherichia coli organisms susceptible to neomycin sulfate in growing turkeys.
AntibacterialCAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Each Packet Contains: 142.8 g neomycin sulfate (commercial grade) equivalent to 100 g neomycin.
NOT FOR USE IN HUMANS
KEEP OUT OF REACH OF CHILDREN
Restricted Drug (California)- Use Only as DirectedApproved by FDA under ANADA # 200-050
NET WEIGHT:
200 g (7 oz) -
200 g Pouch
Add to drinking water - Not for use in liquid supplements.
DOSAGE AND ADMINISTRATION: Administer to cattle (excluding veal calves), swine, sheep and goats at a dose of 10 mg of neomycin sulfate per pound of body weight per day in divided doses for a maximum of 14 days. Administer to turkeys at a dose of 10 mg neomycin sulfate per pound of body weight per day for 5 days.
Herd/Flock Treatment: Each packet will treat 14,300 pounds body weight. Therefore, estimate the total number of pounds of body weight of the animals to be treated and administer one (1) packet (or portion thereof) for each 14,300 pounds. The product should be added to the amount of drinking water estimated to be consumed in 12-24 hours. Provide medicated water as the sole source of water each day until consumed, followed by non-medicated water as required. Fresh medicated water should be prepared each day.
Individual Animal Treatment: To provide 10 mg neomycin sulfate per pound of body weight, mix one (1) level teaspoon in water or milk for each 160 pounds of body weight. Administer daily either as a drench in divided doses or in the drinking water to be consumed in 12-24 hours.
Drinking Water: Swine Use the number of packets indicated below in 256 gallons of water, or in two gallons of stock solution used in proportioner set to meter one ounce per gallon.Pigs Weighing 25-50 Pounds 1 Packet Pigs Weighing 50 to 100 Pounds 1.5 Packets Pigs Weighing Over 100 Pounds 2 Packets -
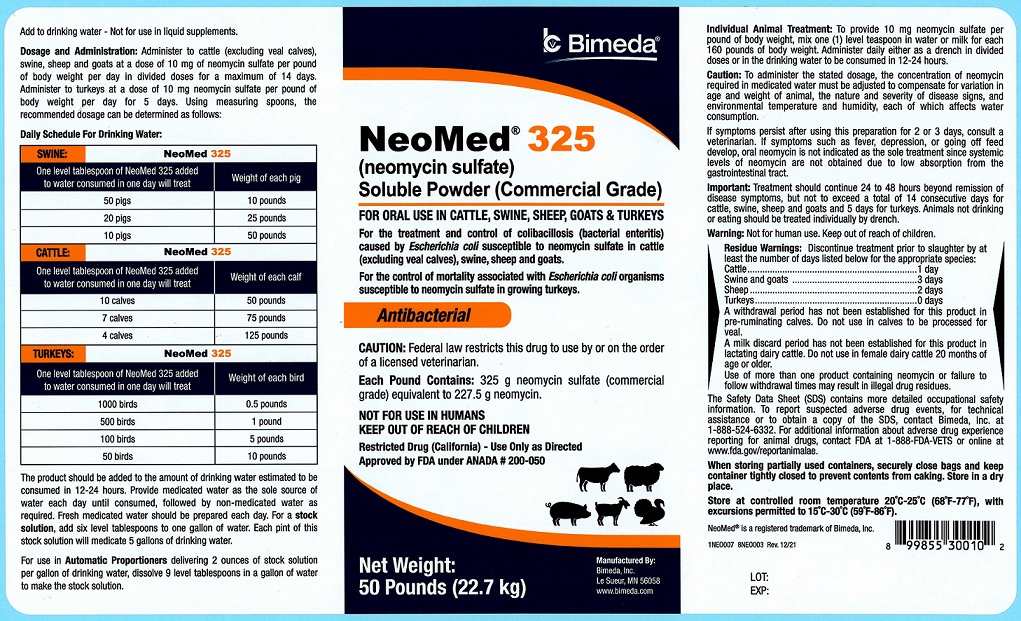
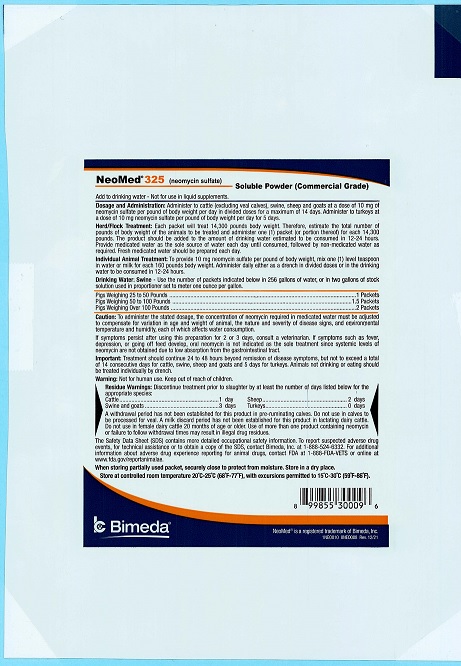
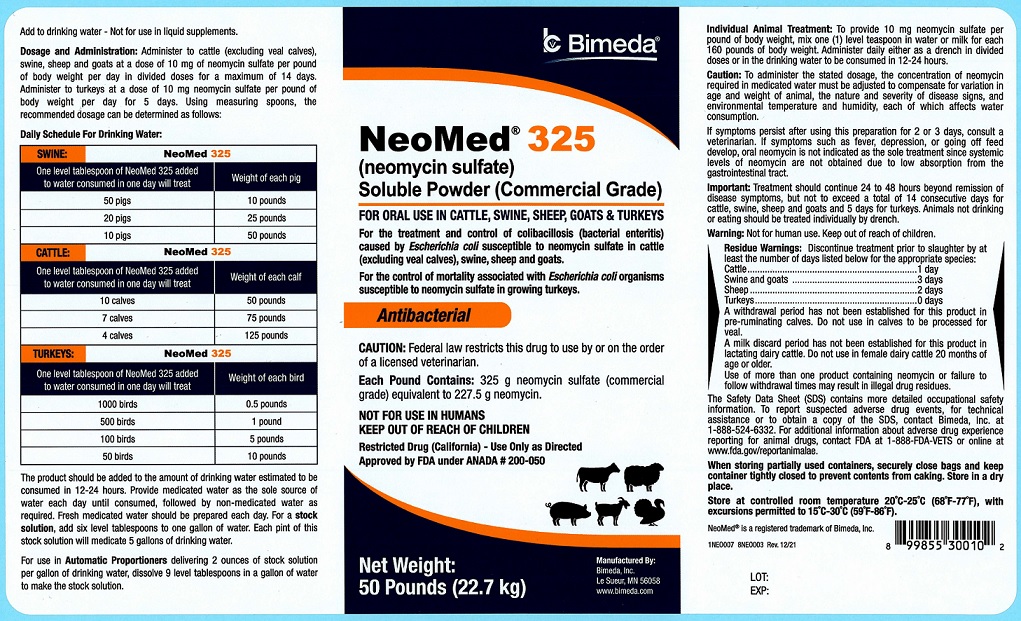
50 Pounds
NeoMed® 325
(neomycin sulfate)
Soluble Powder (Commercial Grade)
FOR ORAL USE IN CATTLE, SWINE, SHEEP, GOATS & TURKEYSFor the treatment and control of colibacillosis (bacterial enteritis) caused by Escherichia coli susceptible to neomycin sulfate in cattle (excluding veal calves), swine, sheep and goats.
For the control of mortality associated with Escherichia coli organisms susceptible to neomycin sulfate in growing turkeys.
AntibacterialCAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Each Pound Contains: 325 g neomycin sulfate (commercial grade) equivalent to 227.5 g neomycin.
NOT FOR USE IN HUMANS
KEEP OUT OF REACH OF CHILDREN
Restricted Drug (California)- Use Only as DirectedApproved by FDA under ANADA # 200-050
NET WEIGHT:
50 Pounds (22.7 kg) -
50 Pounds
Add to drinking water - Not for use in liquid supplements.
Dosage and Administration: Administer to cattle (excluding veal calves), swine, sheep and goats at a dose of 10 mg of neomycin sulfate per pound of body weight per day in divided doses for a maximum of 14 days. Administer to turkeys at a dose of 10 mg neomycin sulfate per pound of body weight per day for 5 days. Using measuring spoons, the recommended dosage can be determined as follows:Daily Schedule For Drinking Water:
SWINE One level tablespoon of NeoMed 325 added to water consumed in one day will treat Weight of each pig 50 pigs 10 pounds 20 pigs 25 pounds 10 pigs 50 pounds CATTLE One level tablespoon of NeoMed 325 added to water consumed in one day will treat Weight of each calf 10 calves 50 pounds 7 calves 75 pounds 4 calves 125 pounds TURKEYS One level tablespoon of NeoMed 325 added to water consumed in one day will treat Weight of each bird 1000 birds 0.5 pounds 500 birds 1 pounds 100 birds 5 pounds 50 birds 10 pounds The product should be added to the amount of drinking water estimated to be consumed in 12-24 hours. Provide medicated water as the sole source of water each day until consumed, followed by non-medicated water as required. Fresh medicated water should be prepared each day. For a stock solution, add six level tablespoons to one gallon of water. Each pint of this stock solution will medicate 5 gallons of drinking water.
For use in Automatic Proportioners delivering 2 ounces of stock solution per gallon of drinking water, dissolve 9 level tablespoons in a gallon of water to make the stock solution.Individual Animal Treatment: To provide 10 mg neomycin sulfate per pound of body weight, mix one (1) level teaspoon in water or milk for each 160 pounds of body weight. Administer daily either as a drench in divided doses or in the drinking water to be consumed in 12-24 hours.
-
WARNINGS AND PRECAUTIONS
Caution: To administer the stated dosage, the concentration of neomycin required in medicated water must be adjusted to compensate for variation in age and weight of animal, the nature and severity of disease signs, and environmental temperature and humidity, each of which affects water consumption.
If symptoms persist after using this preparation for 2 or 3 days, consult a veterinarian. If symptoms such as fever, depression, or going off feed develop, oral neomycin is not indicated as the sole treatment since systemic levels of neomycin are not obtained due to low absorption from the gastrointestinal tract.
Important: Treatment should continue 24 to 48 hours beyond remission of disease symptoms, but not to exceed a total of 14 consecutive days for cattle, swine, sheep and goats and 5 days for turkeys. Animals not drinking or eating should be treated individually by drench.
Warning: Not for human use. Keep out of reach of children.
-
RESIDUE WARNING
Residue Warnings: Discontinue treatment prior to slaughter by at least the number of days listed below for appropriate species:
Cattle 1 day Swine and Goats 3 days Sheep 2 days Turkeys 0 days A withdrawal period has not been established for this product in pre-ruminating calves. Do not use in calves to be processed for veal. A milk discard period has not been established for this product in lactating dairy cattle. Do not use in female dairy cattle 20 months of age or older. Use of more than one product containing neomycin or failure to follow withdrawal times may result in illegal drug residues.
The Safety Data Sheet (SDS) contains more detailed occupational safety information. To report suspected adverse drug events, for technical assistance or to obtain a copy of the SDS, contact Bimeda, Inc. at 1-888-524-6332. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or online at www.fda.gov/reportanimalae.
- 100 g and 200 g Pouches
- 50 Pounds
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NEOMED 325
neomycin sulfate powder, for solutionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:61133-5442 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NEOMYCIN SULFATE (UNII: 057Y626693) (Neomycin - UNII:I16QD7X297) Neomycin 227.5 g in 0.454 kg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:61133-5442-2 22.7 kg in 1 PAIL 2 NDC:61133-5442-3 0.2 kg in 1 POUCH 3 NDC:61133-5442-4 0.1 kg in 1 POUCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200050 01/12/2010 Labeler - Bimeda, Inc. (060492923) Registrant - Bimeda, Inc. (060492923) Establishment Name Address ID/FEI Business Operations Bimeda, Inc. 060492923 manufacture